Optimizing Protein Cross-Linker Production: A Technical Deep Dive into Novel Maleimide Ester Synthesis
The landscape of biochemical reagent manufacturing is undergoing a significant transformation driven by the need for greener, more efficient synthetic pathways. A pivotal development in this sector is detailed in patent CN113087650A, which outlines a robust preparation method for 2-maleimidoacetic acid N-hydroxysuccinimide ester. This compound serves as a critical protein cross-linking agent, playing an indispensable role in elucidating protein conformation, subunit composition, and structural interactions, while also acting as a key building block for functional polymer materials. The disclosed technology represents a paradigm shift from traditional, hazardous synthesis routes to a streamlined, high-yield process that leverages readily available starting materials like maleic anhydride and glycine. For R&D directors and procurement strategists, understanding this technological leap is essential for securing a reliable supply of high-purity intermediates that meet stringent regulatory and environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-maleimidoacetic acid N-hydroxysuccinimide ester has been plagued by significant operational and environmental challenges that hinder scalable production. Traditional methodologies typically rely on a multi-step sequence initiating with acetic anhydride and sodium acetate to generate 2-maleimidoacetic acid, followed by a condensation reaction using DIC (diisopropylcarbodiimide). This legacy approach is characterized by a notoriously long reaction route that accumulates impurities at every stage, drastically reducing overall throughput. Furthermore, the use of acetic anhydride introduces severe safety hazards due to its corrosive nature and pungent odor, creating a hostile working environment and necessitating expensive containment infrastructure. The reliance on DIC condensation exacerbates these issues by generating urea byproducts that are notoriously difficult to remove, often requiring complex purification steps that drive up costs and result in substantial chemical waste discharge, rendering the process economically and ecologically unsustainable for modern large-scale manufacturing.
The Novel Approach
In stark contrast, the innovative methodology presented in the patent data revolutionizes the synthesis by adopting a direct esterification strategy that bypasses the problematic acetic anhydride route entirely. By utilizing maleic anhydride and glycine as the primary feedstocks, the new process initiates with the formation of a stable intermediate, AMAS-1, under mild conditions that minimize side reactions. This strategic pivot not only shortens the synthetic route significantly but also replaces toxic, odorous reagents with benign, cost-effective alternatives that are abundant in the global chemical market. The subsequent conversion to the final N-hydroxysuccinimide ester employs EDCI as a coupling agent, which offers superior solubility characteristics and easier byproduct removal compared to DIC. This holistic redesign of the synthetic pathway results in a dramatic improvement in yield, often exceeding 80%, while simultaneously simplifying the downstream processing requirements, thereby offering a compelling value proposition for cost reduction in biochemical reagent manufacturing.
Mechanistic Insights into EDCI-Mediated Condensation and Esterification
The core of this technological advancement lies in the precise control of reaction kinetics and thermodynamics during the two-stage synthesis. In the first stage, the esterification reaction between maleic anhydride and glycine occurs in a polar aprotic solvent, specifically N,N-dimethylformamide (DMF), which ensures excellent solubility for all reactants and the resulting intermediate. The reaction proceeds efficiently at ambient temperatures ranging from 20 to 25°C, where the nucleophilic attack of the glycine amine group on the maleic anhydride ring leads to ring opening and subsequent cyclization to form the maleimide structure. This step is critical as it establishes the foundational maleimide scaffold without the need for harsh dehydrating agents, preserving the integrity of the functional groups required for subsequent bioconjugation applications.
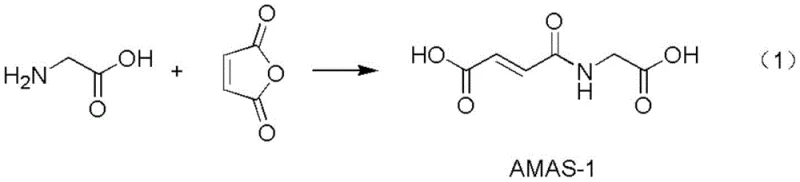
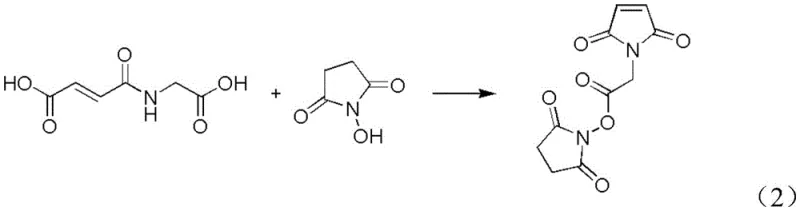
Following the formation of the intermediate AMAS-1, the process transitions to the activation and coupling phase, which is the determinant step for final product quality. Here, N-hydroxysuccinimide (HOSU) is introduced to the reaction mixture, and EDCI acts as the carbonyl activation reagent to facilitate the formation of the active ester bond. The mechanism involves the initial formation of an O-acylisourea intermediate by the reaction of the carboxylic acid group of AMAS-1 with EDCI, which is then rapidly attacked by the hydroxyl group of HOSU to form the stable N-hydroxysuccinimide ester. Conducting this reaction at controlled low temperatures between 10 and 15°C is vital to suppress hydrolysis of the activated species and prevent polymerization of the maleimide double bond. The addition of EDCI in portions further stabilizes the reaction pH and thermal profile, ensuring a clean conversion with minimal formation of N-acylurea byproducts, which are common impurities in carbodiimide chemistry.
How to Synthesize 2-Maleimidoacetic Acid N-Hydroxysuccinimide Ester Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize the benefits of the patented process. The procedure begins with the dissolution of equimolar amounts of maleic anhydride and glycine in DMF, allowing the mixture to stir at room temperature for several hours to ensure complete conversion to the AMAS-1 intermediate. Once the first step is verified, the reaction mixture is cooled, and the coupling reagents are added sequentially to drive the formation of the final ester. The detailed standardized synthesis steps, including specific molar ratios, addition rates, and workup parameters, are outlined in the guide below to assist process engineers in replicating these high-yield results.
- Perform esterification reaction between maleic anhydride and glycine in DMF at 20-25°C to generate the intermediate AMAS-1.
- React the crude AMAS-1 solution directly with N-hydroxysuccinimide (HOSU) and EDCI at 10-15°C to form the target ester.
- Purify the final product by concentrating the solvent, extracting with dichloromethane, and slurring the crude solid with methyl tert-butyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route translates into tangible strategic advantages that extend beyond mere technical feasibility. The shift away from specialized, high-cost reagents like acetic anhydride and DIC towards commodity chemicals like glycine and maleic anhydride fundamentally alters the cost structure of the supply chain. This transition mitigates the risk of price volatility associated with niche reagents and ensures a more stable sourcing environment, as the new raw materials are produced in massive volumes globally for various industries. Furthermore, the simplification of the purification process reduces the consumption of auxiliary solvents and energy, directly impacting the operational expenditure (OPEX) of the manufacturing facility.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-handle reagents significantly lowers the direct material costs associated with production. By avoiding the complex post-treatment procedures required by the traditional DIC method, such as extensive chromatography or multiple recrystallizations, the process reduces labor hours and solvent usage. The high yield achieved through this optimized pathway means that less raw material is wasted per kilogram of finished product, leading to substantial cost savings that can be passed down the supply chain or reinvested into R&D initiatives.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials like glycine and maleic anhydride drastically reduces the lead time for raw material procurement. Unlike specialized coupling agents that may have limited suppliers and long delivery windows, these commodity chemicals can be sourced from multiple vendors worldwide, ensuring business continuity even during market disruptions. The robustness of the reaction conditions, which do not require extreme temperatures or pressures, also means that production can be easily transferred between different manufacturing sites without significant requalification efforts, enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with mild reaction conditions that pose minimal safety risks even in large reactors. The reduction in hazardous waste generation, particularly the avoidance of acetic acid byproducts and difficult-to-remove urea derivatives, simplifies compliance with increasingly stringent environmental regulations. This 'green' aspect of the synthesis not only lowers waste disposal costs but also aligns with the sustainability goals of major pharmaceutical and biotech clients, making the manufacturer a more attractive partner for long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-maleimidoacetic acid N-hydroxysuccinimide ester. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the practical implementation of this advanced synthesis method.
Q: What are the primary advantages of the glycine-based route over the traditional acetic anhydride method?
A: The glycine-based route described in patent CN113087650A eliminates the use of acetic anhydride and sodium acetate, which are associated with strong odors and high waste generation. This novel approach utilizes cheaper, low-toxicity raw materials and achieves a significantly shorter reaction route with higher yields (over 80%) and simplified post-treatment procedures compared to the difficult DIC condensation methods of the past.
Q: How does the new purification method improve industrial scalability?
A: The patented purification process avoids complex chromatography or recrystallization steps that often bottleneck production. By simply concentrating the DMF solvent, performing a standard aqueous workup with dichloromethane, and finally slurring the crude product in methyl tert-butyl ether, manufacturers can achieve high purity with minimal solvent consumption and reduced three-waste discharge, making it highly suitable for large-scale commercial manufacturing.
Q: Why is EDCI preferred over DIC for this specific condensation reaction?
A: EDCI (1-(3-dimethylaminopropyl)-3-ethylcarbodiimide) is a water-soluble carbodiimide that facilitates easier removal of urea byproducts during the aqueous workup phase. Unlike DIC, which can lead to difficult post-treatment scenarios and lower yields due to side reactions, EDCI allows for mild reaction conditions (10-15°C) and promotes a cleaner conversion of the intermediate AMAS-1 to the final N-hydroxysuccinimide ester.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Maleimidoacetic Acid N-Hydroxysuccinimide Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN113087650A are fully realized in a commercial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-maleimidoacetic acid N-hydroxysuccinimide ester meets the exacting standards required for protein conjugation and polymer synthesis applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and efficiency of your supply chain.
