Advanced Chiral Phosphinite-Imidazoline Ligands for Scalable Asymmetric Hydrogenation
The chemical landscape of asymmetric synthesis is continuously evolving, driven by the demand for higher purity chiral intermediates in the pharmaceutical and agrochemical sectors. Patent CN1842533A introduces a significant advancement in this field through the disclosure of novel phosphinite-imidazoline compounds and their metal complexes. These compounds, defined by Formula I and Ia, serve as highly efficient chiral ligands for transition metal catalysts, specifically targeting the asymmetric addition of hydrogen, borane, or silane to prochiral organic compounds. The innovation lies in the unique combination of an imidazoline backbone with a phosphinite group attached at the alpha-position, creating a robust P,N-chelating system that offers exceptional stereocontrol.
For R&D directors and process chemists, the significance of this technology cannot be overstated. Traditional methods often rely on oxazoline-based ligands which, while effective, can sometimes lack the necessary versatility for challenging substrates such as cis-alkenes with two prochiral centers. The ligands described in this patent demonstrate surprisingly high catalytic activity and enantioselectivity, often matching or exceeding known standards. By leveraging the structural flexibility of the imidazoline ring and the tunable nature of the phosphinite group, manufacturers can achieve precise control over reaction outcomes, thereby streamlining the development of complex active pharmaceutical ingredients (APIs).
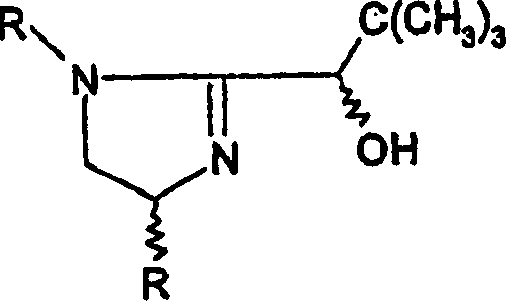
The transition from conventional catalytic systems to these novel phosphinite-imidazoline ligands represents a strategic shift in process efficiency. Conventional methods for asymmetric hydrogenation frequently encounter limitations when dealing with sterically hindered substrates or specific geometric isomers. For instance, standard phosphine-oxazoline ligands might yield acceptable results for trans-alkenes but often falter with cis-configurations, leading to lower enantiomeric excess (ee) and necessitating costly downstream purification steps like recrystallization or chromatography. Furthermore, the synthesis of some traditional ligands can be rigid, limiting the ability to fine-tune the catalyst for specific substrate-catalyst matching without redesigning the entire synthetic route.
In contrast, the novel approach detailed in the patent utilizes a modular synthetic strategy that decouples the formation of the heterocyclic core from the introduction of the phosphorus functionality. This allows for the independent optimization of steric bulk and electronic properties. The imidazoline ring, formed via the cyclization of haloimino esters with primary amines, provides a stable chiral environment. Subsequent functionalization with various secondary phosphines enables the creation of a diverse library of ligands tailored to specific reaction requirements. This modularity not only enhances catalytic performance, as evidenced by high conversion rates and ee values in the examples provided, but also simplifies the supply chain by allowing the use of common intermediates for multiple final products.
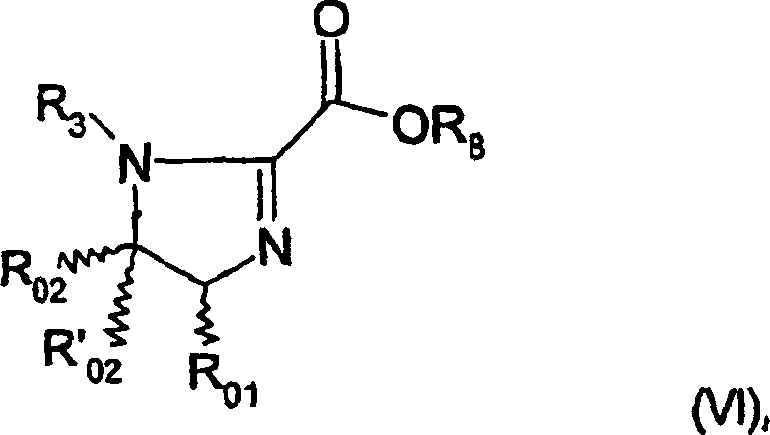
The mechanistic superiority of these phosphinite-imidazoline ligands stems from their ability to form stable, well-defined coordination geometries with Group VIII metals like Iridium and Rhodium. In the catalytic cycle, the P,N-bidentate nature of the ligand creates a rigid chiral pocket around the metal center. The nitrogen atom of the imidazoline ring and the phosphorus atom of the phosphinite group coordinate simultaneously, locking the substrate into a specific orientation that favors the formation of one enantiomer over the other. The steric environment is critically influenced by the substituents on the imidazoline ring (R01, R4) and the phosphorus atom (R1, R2). For example, bulky groups like tert-butyl or phenyl on the phosphorus can effectively block one face of the substrate, directing the hydride transfer to the opposite face with high fidelity.
Furthermore, the electronic properties of the phosphinite group play a pivotal role in the activation of the metal-hydride species. Unlike simple phosphines, phosphinites possess distinct electron-donating characteristics that can accelerate the oxidative addition or migratory insertion steps within the catalytic cycle. The patent data indicates that these ligands are particularly effective for the hydrogenation of carbon-carbon double bonds, including challenging cis-alkenes. The ability to maintain high enantioselectivity even with substrates possessing two prochiral centers suggests a highly discriminating transition state. This level of control minimizes the formation of diastereomeric impurities, which is a critical quality attribute for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Phosphinite-Imidazoline Ligands Efficiently
The synthesis of these high-value chiral ligands follows a logical, step-wise progression that ensures high overall yields and purity. The process begins with the preparation of key imidazoline intermediates, which serve as the chiral scaffold for the final ligand. By adhering to the standardized protocols outlined in the patent, manufacturers can reliably produce these compounds with consistent optical purity. The following guide summarizes the critical stages involved in transforming simple starting materials into the sophisticated phosphinite-imidazoline structures required for advanced catalytic applications.
- Cyclization of haloimino esters with primary amines to form the imidazoline core structure.
- Nucleophilic addition of organometallic reagents (Grignard or Alkyllithium) to the ester group to generate the tertiary alcohol intermediate.
- Metallation of the hydroxyl group followed by reaction with secondary halophosphines to install the phosphinite moiety.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this technology offers substantial strategic benefits beyond mere technical performance. The modular nature of the synthesis means that raw material sourcing is more flexible; common precursors like amino alcohols and oxalic acid derivatives are widely available in the global chemical market. This reduces dependency on single-source suppliers for exotic starting materials, thereby enhancing supply chain resilience. Additionally, the high catalytic efficiency reported in the patent implies that lower catalyst loadings may be sufficient to achieve complete conversion, which directly translates to reduced consumption of precious metals like Iridium or Rhodium, representing a significant cost avoidance measure in large-scale operations.
Moreover, the exceptional enantioselectivity achieved with these ligands drastically simplifies downstream processing. In traditional processes where ee is moderate, extensive purification steps such as multiple recrystallizations or preparative chiral HPLC are often required to meet specification limits. By achieving high ee directly from the reaction, the number of unit operations is reduced, leading to lower solvent consumption, reduced waste generation, and shorter production cycles. This efficiency gain contributes to a more sustainable manufacturing footprint and lowers the overall cost of goods sold (COGS) for the final chiral intermediate. The ability to scale these reactions from gram to kilogram levels without loss of performance further de-risks the commercialization timeline for new drug candidates.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal removal steps and the reduction in purification cycles due to high selectivity lead to substantial operational savings. The modular synthesis also allows for economies of scale in producing intermediate batches that can be diverted to different final ligand variants based on demand.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials mitigates the risk of supply disruptions. The robustness of the synthetic route ensures consistent quality output, which is vital for maintaining long-term contracts with pharmaceutical clients who require rigorous batch-to-batch consistency.
- Scalability and Environmental Compliance: The process conditions described, such as the use of standard solvents and moderate temperatures, are amenable to large-scale reactor operations. Furthermore, the high atom economy and reduced waste profile align with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of phosphinite-imidazoline ligands in industrial settings. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for their process development pipelines.
Q: What are the primary advantages of phosphinite-imidazoline ligands over traditional oxazolines?
A: According to patent CN1842533A, these ligands offer superior enantioselectivity, particularly for the hydrogenation of prochiral cis-isomers of alkenes, where traditional ligands may struggle. They also provide greater modularity for tuning steric and electronic properties.
Q: Which metals are compatible with these chiral ligands for catalytic applications?
A: The patent specifies that these ligands form effective complexes with Transition Metals of Group I and VIII, specifically highlighting Ruthenium (Ru), Rhodium (Rh), and Iridium (Ir) for asymmetric hydrogenation and other addition reactions.
Q: How does the modular synthesis impact supply chain reliability?
A: The synthetic route allows for the independent variation of substituents on the nitrogen and the phosphorus atoms. This modularity means that diverse ligand libraries can be generated from common intermediates, reducing the risk of supply bottlenecks for specific custom catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphinite-Imidazoline Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral ligands play in the synthesis of next-generation therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the optical purity and metal content of these sensitive catalytic systems, guaranteeing that every batch meets the exacting standards required for GMP production.
We invite you to collaborate with our technical team to explore how these novel ligands can optimize your specific synthetic routes. Whether you require custom synthesis of specific ligand variants or comprehensive process development services, we are prepared to deliver a Customized Cost-Saving Analysis tailored to your project needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence and efficiency.
