Advanced Phosphorescent Host Materials: Enhancing OLED Efficiency and Commercial Viability
The rapid evolution of the display industry has placed immense pressure on material scientists to develop organic electroluminescent materials that surpass the theoretical limits of traditional fluorescence. Patent CN113620951B represents a significant technological leap in this domain, introducing a novel class of phosphorescent compounds designed to serve as high-performance host materials in Organic Light Emitting Diodes (OLEDs). Unlike conventional fluorescent emitters that waste approximately 75% of energy through non-emissive triplet states, these new phosphorescent structures harness both singlet and triplet excitons, theoretically achieving 100% internal quantum efficiency. The core innovation lies in the unique molecular architecture defined by General Formula I, which integrates rigid polycyclic aromatic systems with specific heteroatom linkages to enhance thermal stability and charge transport properties.
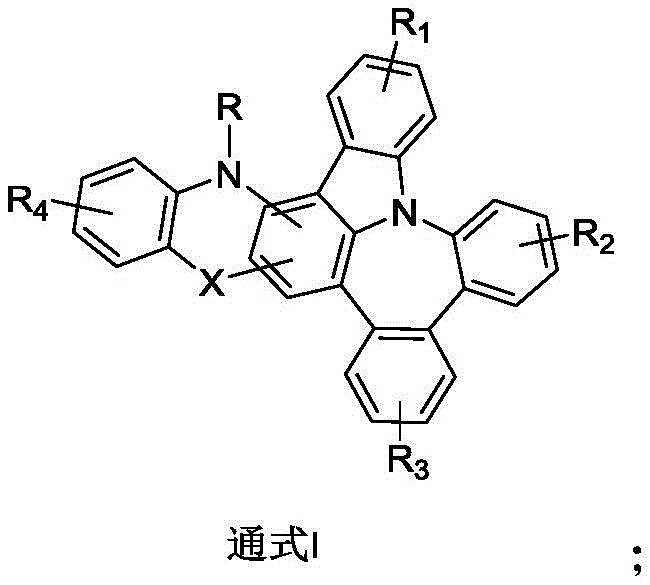
For R&D directors evaluating new material platforms, the structural versatility offered by this patent is paramount. The general formula allows for extensive modulation of the Highest Occupied Molecular Orbital (HOMO) and Lowest Unoccupied Molecular Orbital (LUMO) energy levels through the variation of substituents R1 through R4, which can range from simple alkyl groups to complex fused aryl systems like phenanthryl or pyrenyl groups. This tunability is critical for matching the energy levels of dopant materials, thereby minimizing energy loss during exciton transfer. Furthermore, the inclusion of linkers such as oxygen, sulfur, or nitrogen bridges (represented by X) provides additional degrees of freedom to optimize the glass transition temperature (Tg), a key parameter for device longevity. By preventing crystallization during device operation, these structural features directly contribute to the extended service life observed in the experimental data, addressing one of the most persistent challenges in commercial OLED deployment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-efficiency phosphorescent host materials has been plagued by convoluted multi-step pathways that suffer from poor atom economy and difficult purification protocols. Traditional methods often rely on harsh reaction conditions or expensive transition metal catalysts that leave behind trace metallic impurities, which act as quenching sites and drastically reduce device efficiency. Moreover, many prior art compounds exhibit insufficient thermal stability, leading to morphological changes in the thin film during prolonged operation at high brightness. This instability manifests as a rapid increase in driving voltage and a precipitous drop in luminance over time, rendering such materials unsuitable for the rigorous demands of consumer electronics and automotive displays. The complexity of synthesizing fused ring systems with precise regioselectivity has also been a bottleneck, often resulting in low overall yields that drive up the cost of goods sold (COGS) prohibitively for mass market adoption.
The Novel Approach
The methodology disclosed in CN113620951B circumvents these historical bottlenecks through a streamlined, two-step palladium-catalyzed strategy that prioritizes both yield and purity. Instead of attempting to construct the entire fused core in a single high-risk transformation, the process builds the molecule modularly. The first step establishes the foundational carbon-carbon or carbon-heteroatom bonds under mild conditions, while the second step executes a precise intramolecular cyclization to lock the conformation. This divide-and-conquer approach not only simplifies the reaction monitoring but also allows for the purification of the intermediate, effectively removing side products before the final ring-closing step. The result is a robust synthetic route that consistently delivers high-purity products with yields exceeding 80% in pilot examples, demonstrating a clear path toward cost reduction in electronic chemical manufacturing without sacrificing performance metrics.
Mechanistic Insights into Palladium-Catalyzed Sequential Coupling
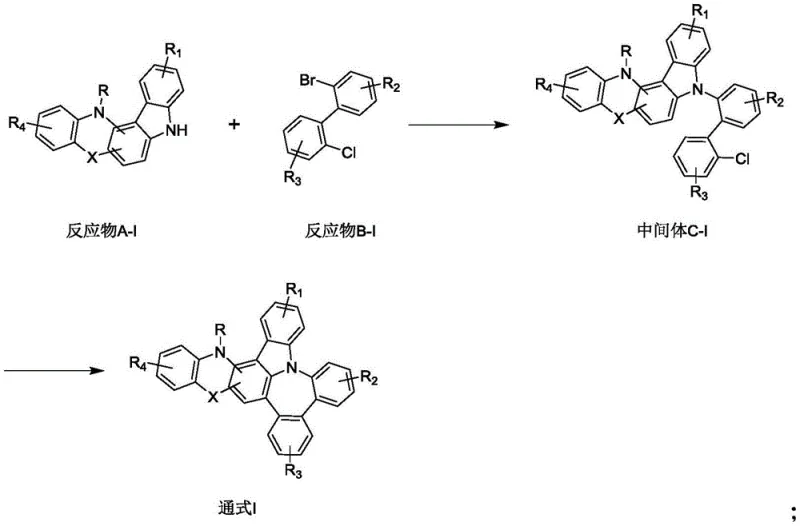
The chemical elegance of this synthesis lies in the sequential application of palladium catalysis to achieve distinct bond formations with high fidelity. The first stage involves a classic cross-coupling reaction, likely a Suzuki-Miyaura type given the use of boronic acid derivatives or halides (Reactant A-I and B-I) in the presence of tetrakis(triphenylphosphine)palladium and a base like potassium carbonate. This step operates at a moderate temperature of 80°C in a biphasic solvent system of toluene, ethanol, and water, facilitating the efficient coupling of the aryl halide with the nucleophilic partner. The choice of ligands and solvent polarity is critical here to ensure complete conversion while minimizing homocoupling byproducts. Following isolation and recrystallization of the intermediate C-I, the second stage employs a different catalytic system utilizing palladium acetate and bulky phosphine ligands such as Pcy3. This system is tailored for C-H activation or intramolecular arylation, driving the cyclization at 150°C in polar aprotic solvents like DMF to form the rigid fused core essential for phosphorescence.
From an impurity control perspective, the mechanistic pathway is designed to minimize the formation of difficult-to-remove isomers. The use of specific directing groups and steric bulk in the reactants guides the palladium catalyst to the desired position on the aromatic ring, ensuring regioselectivity. Furthermore, the purification protocol described—utilizing extraction followed by column chromatography with dichloromethane and petroleum ether gradients—is highly effective at separating the target phosphorescent compound from residual palladium species and unreacted starting materials. For electronic applications, where parts-per-million levels of metal impurities can be detrimental, this rigorous purification capability is a significant advantage. The high mass spectrometry agreement between theoretical and found values in the examples confirms the structural integrity and purity of the final products, validating the effectiveness of this mechanistic approach in producing electronic-grade materials.
How to Synthesize Phosphorescent Compound Efficiently
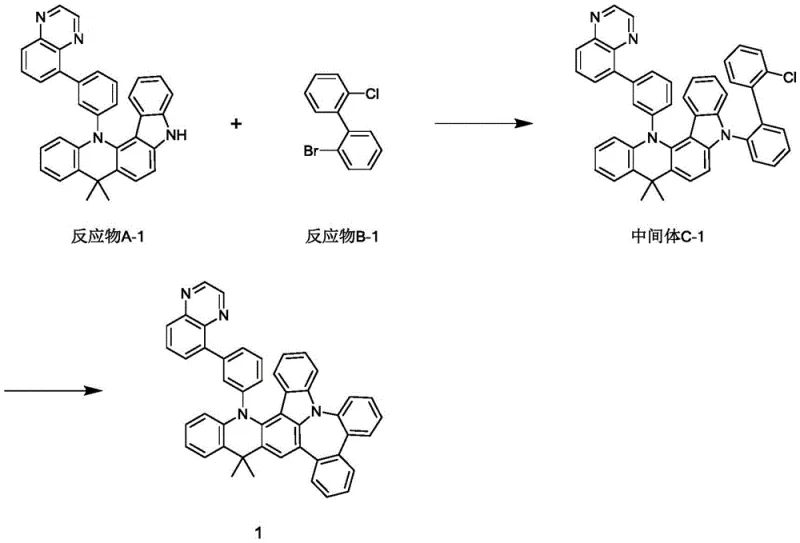
The practical implementation of this synthesis requires careful attention to reaction parameters to maximize yield and reproducibility. The process begins with the preparation of the intermediate through cross-coupling, followed by the critical cyclization step which forms the emissive core. Detailed operational guidelines regarding stoichiometry, temperature ramping, and workup procedures are essential for scaling this chemistry from the benchtop to the pilot plant. The following guide outlines the standardized steps derived from the patent examples to ensure consistent quality.
- Perform a Suzuki-Miyaura cross-coupling reaction between Reactant A-I and Reactant B-I using tetrakis(triphenylphosphine)palladium and potassium carbonate in a toluene/ethanol/water solvent system at 80°C to obtain Intermediate C-I.
- Execute an intramolecular cyclization of Intermediate C-I using palladium acetate and tricyclohexylphosphine ligands with potassium carbonate in DMF at 150°C to close the ring structure.
- Purify the final crude product through aqueous workup, extraction with ethyl acetate, and column chromatography using dichloromethane and petroleum ether to achieve high-purity phosphorescent compounds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this new class of phosphorescent compounds offers tangible strategic benefits beyond mere performance metrics. The synthetic route relies on commodity chemicals and widely available catalysts, reducing the risk of supply chain disruptions associated with exotic or proprietary reagents. The high yields observed in the experimental data translate directly to reduced raw material consumption per kilogram of finished product, which is a primary driver for cost optimization. Additionally, the use of standard industrial solvents like toluene and DMF simplifies waste management and solvent recovery processes, aligning with increasingly stringent environmental regulations. This operational simplicity lowers the barrier for commercial scale-up, allowing manufacturers to respond more agilely to market demand fluctuations without the need for specialized, capital-intensive reactor configurations.
- Cost Reduction in Manufacturing: The elimination of complex, low-yielding steps significantly lowers the overall production cost. By achieving yields consistently above 80% in key examples, the process minimizes waste generation and maximizes the throughput of existing manufacturing assets. The avoidance of cryogenic conditions or ultra-high vacuum requirements further reduces energy consumption and operational overhead. Consequently, the total cost of ownership for these materials is substantially lower compared to legacy phosphorescent hosts that require intricate purification or fragile handling protocols.
- Enhanced Supply Chain Reliability: The starting materials, such as substituted bromobiphenyls and carbazole derivatives, are produced by multiple global suppliers, ensuring a competitive and resilient supply base. This diversification mitigates the risk of single-source dependency, a critical factor for long-term product lifecycle management in the electronics sector. Furthermore, the robustness of the reaction conditions means that production schedules are less susceptible to delays caused by sensitive reaction failures, ensuring a steady flow of high-purity OLED material to downstream device fabricators.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot runs without fundamental changes to the chemistry. The solvent systems employed are amenable to standard distillation and recycling technologies, reducing the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only satisfies corporate sustainability goals but also future-proofs the supply chain against tightening regulatory frameworks regarding volatile organic compound (VOC) emissions and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the adoption of these phosphorescent compounds. The answers are derived directly from the technical disclosures and performance data within the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of these phosphorescent compounds over traditional fluorescent materials?
A: Unlike fluorescent materials which theoretically limit internal quantum efficiency to 25%, these phosphorescent compounds utilize both singlet and triplet excitons, enabling up to 100% internal quantum efficiency. This results in significantly improved luminous efficiency, lower driving voltage, and extended operational lifetime for OLED devices.
Q: How does the synthetic route ensure high purity for electronic grade applications?
A: The process employs a robust two-step palladium-catalyzed sequence with specific recrystallization and column chromatography purification steps. The use of standard solvents like toluene and DMF allows for effective removal of metal catalysts and organic impurities, ensuring the stringent purity specifications required for display manufacturing.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method utilizes readily available starting materials and standard reaction conditions (80°C and 150°C) without requiring exotic reagents. The high yields reported (consistently above 75-80% in examples) and the use of common industrial solvents indicate strong potential for scalable manufacturing from kilogram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorescent Compound Supplier
As the demand for high-efficiency display materials continues to surge, partnering with an experienced chemical manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis critical for OLED applications. We understand that the transition to new host materials requires not just a product, but a reliable partner who can guarantee batch-to-batch reproducibility and technical support throughout the integration process.
We invite you to engage with our technical procurement team to discuss how these advanced phosphorescent compounds can be integrated into your specific device architectures. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this superior material platform. We are prepared to provide specific COA data and route feasibility assessments to accelerate your evaluation timeline. Let us collaborate to bring the next generation of high-performance, long-lifetime OLED displays to market faster and more efficiently.
