Advanced Alkaline Hydrolysis Technology for Commercial Scale Production of Cysteamine Hydrochloride
Introduction to Next-Generation Cysteamine Hydrochloride Synthesis
The global demand for high-purity pharmaceutical intermediates, particularly Cysteamine Hydrochloride, has necessitated a rigorous re-evaluation of traditional synthetic pathways to meet modern efficiency and safety standards. Patent CN101225063A introduces a transformative methodology centered on alkaline hydrolysis, marking a significant departure from the historically cumbersome acid-catalyzed processes that have plagued the industry for decades. This innovative approach leverages the superior nucleophilicity of hydroxide ions to facilitate ring-opening reactions, resulting in a streamlined production cycle that drastically curtails reaction times while simultaneously enhancing overall yield profiles. By shifting the reaction environment from acidic to alkaline conditions, the process not only mitigates the risks associated with toxic gas evolution but also establishes a single-phase system that promotes superior mass transfer and reaction kinetics. For R&D directors and procurement strategists alike, this patent represents a critical opportunity to optimize supply chains for key anti-ulcer medications like Ranitidine and Cimetidine, ensuring a more robust and cost-effective source of this vital chemical building block.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cysteamine Hydrochloride has been hindered by severe inefficiencies and safety hazards inherent in older technological frameworks, such as those relying on prolonged acid hydrolysis or the direct use of hydrogen sulfide. Traditional acid hydrolysis methods often require reaction times exceeding 45 to 160 hours to achieve completion, creating a significant bottleneck in production throughput and tying up reactor capacity for extended periods. Furthermore, these legacy processes frequently suffer from suboptimal yields, with some historical data indicating final product recoveries as low as 42.5% to 65%, leading to substantial raw material wastage and increased unit costs. The reliance on hazardous reagents like hydrogen sulfide, a potent asphyxiant, introduces complex safety engineering requirements and stringent regulatory burdens that complicate facility operations and increase insurance liabilities. Additionally, the isolation of intermediates in these old methods often necessitates energy-intensive recrystallization steps using organic solvents like acetic acid, which complicates downstream purification and solvent recovery efforts.
The Novel Approach
In stark contrast, the novel alkaline hydrolysis method detailed in the patent data offers a paradigm shift by utilizing a base-catalyzed mechanism that fundamentally alters the thermodynamics and kinetics of the ring-opening step. This approach achieves a remarkable reduction in hydrolysis time, completing the critical conversion in approximately 11 hours, which is roughly one-quarter of the time required by conventional acidic methods. The process boasts an impressive recovery rate for the key intermediate, α-mercaptothiazoline, achieving yields upwards of 90%, which directly translates to higher overall process efficiency and reduced waste generation. By operating under atmospheric pressure and utilizing readily available alkaline solutions such as sodium hydroxide or potassium hydroxide, the method eliminates the need for expensive high-pressure reactors and specialized corrosion-resistant alloys. The resulting product exhibits superior physical characteristics, appearing as a white waxy solid with high purity, thereby reducing the need for extensive post-synthesis polishing and ensuring consistent quality for downstream API manufacturing.
Mechanistic Insights into Alkaline Hydrolysis and Cyclization
The core chemical innovation of this process lies in the strategic utilization of alkaline conditions to drive the hydrolysis of the thiazoline ring, a step that is kinetically sluggish under acidic conditions due to the weaker nucleophilicity of water molecules compared to hydroxide ions. In the alkaline environment, the abundance of hydroxide ions acts as a powerful nucleophile, aggressively attacking the electrophilic centers of the five-membered heterocyclic ring of α-mercaptothiazoline, thereby facilitating rapid ring opening and cleavage of the carbon-sulfur bonds. Simultaneously, the acidic nature of the thiol group generated during hydrolysis allows it to immediately react with the excess base to form a stable thiolate salt, which remains soluble in the aqueous phase and prevents the formation of insoluble precipitates that could otherwise inhibit reaction progress. This formation of a homogeneous single-phase system ensures uniform heat distribution and mass transfer throughout the reaction vessel, preventing localized hot spots and ensuring consistent product quality across large batches. Furthermore, any acidic gaseous byproducts such as carbon dioxide or trace hydrogen sulfide generated during the decomposition are instantly neutralized and absorbed by the alkaline medium, driving the chemical equilibrium forward according to Le Chatelier's principle and effectively scrubbing the exhaust stream of harmful emissions.
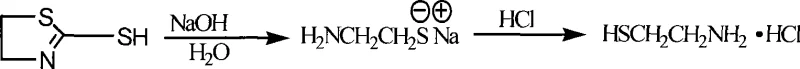
From an impurity control perspective, the alkaline pathway inherently suppresses the formation of oxidative byproducts that are commonly observed in acid-catalyzed environments where free thiols are prone to dimerization into disulfides. The rapid conversion of the free thiol into its salt form protects the sulfhydryl group from oxidation by atmospheric oxygen, preserving the integrity of the molecule until the final acidification step. The subsequent acidification with hydrochloric acid is performed under controlled conditions to precipitate the final Cysteamine Hydrochloride, allowing for precise tuning of the pH to maximize crystal formation while minimizing the co-precipitation of inorganic salts. This mechanistic advantage ensures that the final impurity profile is significantly cleaner compared to traditional methods, reducing the burden on analytical quality control teams and facilitating faster batch release times. The ability to recycle the mother liquor and solvent water further enhances the green chemistry profile of the synthesis, aligning with modern sustainability goals without compromising on the chemical purity required for pharmaceutical applications.
How to Synthesize Cysteamine Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a robust, four-stage framework that is highly amenable to standard chemical manufacturing equipment, requiring no exotic catalysts or extreme operating conditions. The process begins with the esterification of ethanolamine and sulfuric acid to form 2-aminoethyl sulfate, followed by a cyclization step with carbon disulfide to generate the thiazoline intermediate, which is then purified via recrystallization before undergoing the critical alkaline hydrolysis. Each stage is designed to maximize yield and minimize waste, with specific attention paid to temperature control and molar ratios to ensure optimal reaction kinetics throughout the sequence. The detailed standardized synthesis steps, including precise temperature ranges, molar ratios, and workup procedures, are provided in the technical guide below for immediate implementation by process engineering teams.
- Synthesize 2-aminoethyl sulfate by reacting ethanolamine with sulfuric acid under controlled temperature (0-40°C) followed by vacuum distillation.
- Perform cyclization by reacting 2-aminoethyl sulfate with carbon disulfide in an alkaline solution (NaOH/KOH) at 40-65°C to form α-mercaptothiazoline.
- Purify the intermediate α-mercaptothiazoline via recrystallization using boiling distilled water and vacuum drying.
- Execute alkaline hydrolysis of α-mercaptothiazoline using NaOH/KOH solution, followed by acidification with HCl to precipitate the final Cysteamine Hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this alkaline hydrolysis technology presents a compelling value proposition driven by substantial operational efficiencies and risk mitigation strategies. The elimination of high-pressure reaction requirements significantly lowers capital expenditure (CapEx) for new production lines, as standard glass-lined or stainless steel reactors can be utilized without the need for specialized pressure-rated vessels. Moreover, the dramatic improvement in yield for the intermediate stage means that less raw material is required to produce the same amount of final product, directly impacting the cost of goods sold (COGS) and improving margin potential in a competitive market. The ability to recycle solvents such as water and ethanol further contributes to cost reduction in pharmaceutical intermediate manufacturing by minimizing waste disposal fees and reducing the volume of fresh solvent purchases required per batch.
- Cost Reduction in Manufacturing: The process achieves a significant reduction in production costs primarily through the elimination of expensive high-pressure equipment and the optimization of raw material utilization via higher yields. By avoiding the use of toxic hydrogen sulfide gas, the facility saves on the substantial costs associated with specialized gas handling infrastructure, leak detection systems, and hazardous waste disposal protocols. The recycling of solvent water and ethanol creates a closed-loop system that drastically reduces utility consumption and waste treatment expenses, contributing to a leaner and more economically viable production model. Additionally, the shorter reaction cycle time increases reactor turnover rates, allowing the same asset base to produce a higher volume of product annually without additional capital investment.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as ethanolamine, sulfuric acid, and sodium hydroxide ensures a stable and diversified supply base that is less susceptible to the volatility often seen with specialized or hazardous reagents. Removing the dependency on hydrogen sulfide eliminates a critical single point of failure in the supply chain, as the logistics of transporting and storing this toxic gas are complex and prone to regulatory disruptions. The robustness of the atmospheric pressure process means that production is less likely to be interrupted by equipment maintenance issues related to pressure seals or safety valve failures, ensuring consistent delivery schedules for downstream API manufacturers. This stability is crucial for maintaining the continuity of supply for essential medications like anti-ulcer drugs, where interruptions can have significant clinical and commercial repercussions.
- Scalability and Environmental Compliance: The simplicity of the reaction conditions, specifically the operation at atmospheric pressure and moderate temperatures, makes this process exceptionally easy to scale from pilot plant to commercial tonnage without encountering the non-linear heat transfer issues common in pressurized systems. The inherent safety of the alkaline medium, which traps acidic gases, simplifies environmental compliance by reducing the load on scrubber systems and ensuring that emissions remain well within regulatory limits. The generation of a white, high-purity solid product reduces the need for complex downstream purification steps, streamlining the packaging and shipping logistics for the final intermediate. This scalability ensures that the technology can meet growing global demand for Cysteamine Hydrochloride while adhering to increasingly stringent environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkaline hydrolysis technology, derived directly from the comparative data and mechanistic advantages presented in the patent literature. These insights are intended to clarify the operational benefits and safety improvements over legacy methods for stakeholders evaluating this synthesis route. Detailed answers are provided below to assist in your technical assessment and feasibility studies.
Q: What are the primary advantages of the alkaline hydrolysis method over traditional acid hydrolysis?
A: The alkaline hydrolysis method significantly reduces reaction time from over 45 hours to approximately 11 hours and eliminates the need for hazardous hydrogen sulfide gas, while improving the yield of the α-mercaptothiazoline intermediate to over 90%.
Q: How does this process impact environmental compliance and waste management?
A: The process operates at atmospheric pressure without requiring specialized pressurized equipment, and the alkaline environment effectively absorbs acidic byproduct gases like CO2 and H2S, reducing harmful emissions and simplifying waste treatment protocols.
Q: Is the solvent system in this synthesis method sustainable for large-scale production?
A: Yes, the method utilizes water and ethanol as primary solvents, both of which can be recovered and recycled through distillation processes, substantially lowering raw material consumption and operational costs in commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cysteamine Hydrochloride Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced alkaline hydrolysis technology to deliver superior quality Cysteamine Hydrochloride to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical and agrochemical applications, providing our partners with absolute confidence in material consistency. Our commitment to process excellence means we can offer a reliable pharmaceutical intermediate supplier partnership that balances technical sophistication with commercial pragmatism.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements and cost structures. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages specific to your volume needs and logistical constraints. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the quality and viability of our production capabilities against your internal standards. Let us collaborate to secure a sustainable and efficient supply of this critical intermediate for your future projects.
