Scaling High-Performance Phosphorus-Nitrogen-Silicon Intumescent Flame Retardants for Industrial Polymers
Scaling High-Performance Phosphorus-Nitrogen-Silicon Intumescent Flame Retardants for Industrial Polymers
The global demand for environmentally friendly, halogen-free flame retardants has driven significant innovation in polymer additive chemistry, specifically targeting the limitations of traditional intumescent systems. Patent CN107417912B introduces a groundbreaking synthesis method for a phosphorus-nitrogen-silicon intumescent flame retardant that integrates a triazine ring and a rigid cage-like structure into a single macromolecular framework. This technological advancement addresses critical failure points in existing fire safety materials, such as poor thermal stability, water solubility, and the migration of small molecules to the polymer surface. By covalently bonding the acid source, carbon source, and gas source within one complex architecture, this new class of additives offers a reliable polymer additive supplier solution that meets stringent international safety and environmental standards without compromising mechanical properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the flame retardant industry has relied heavily on halogenated compounds or simple physical blends of intumescent ingredients, both of which present severe drawbacks for modern high-performance applications. Halogenated flame retardants, while effective, release corrosive gases and dense smoke during combustion, posing significant environmental and health hazards that violate increasingly strict global regulations. Furthermore, traditional intumescent flame retardants (IFR) often utilize small molecule carbonizing agents that exhibit high water solubility and a tendency to migrate out of the polymer matrix over time, leading to a phenomenon known as blooming which degrades surface quality and reduces long-term fire resistance. These conventional systems frequently require complex compounding with ammonium polyphosphate to achieve efficacy, resulting in formulations that are hygroscopic and thermally unstable at processing temperatures above 250°C, limiting their utility in engineering plastics.
The Novel Approach
The synthetic strategy outlined in the patent data overcomes these deficiencies by constructing a high-molecular-weight species where the flame-retarding elements are chemically locked into the backbone. This novel approach utilizes a stepwise condensation reaction starting with phosphorus oxychloride and a cage-like hydroxyl compound (PEPA) to create a stable phosphorus intermediate, which is then linked to a triazine core modified with silane coupling agents. 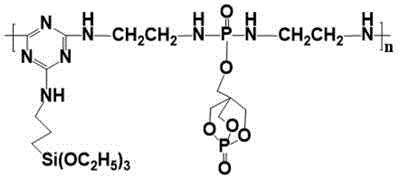 This integration ensures that the acid source, carbon source, and blowing agent are present in optimal stoichiometric ratios within every molecule, promoting a rapid and cohesive char formation upon exposure to heat. The inclusion of silicon further reinforces the char layer, creating a ceramic-like barrier that provides superior insulation against oxygen and heat transfer, effectively preventing melt dripping and sustaining the structural integrity of the polymer during a fire event.
This integration ensures that the acid source, carbon source, and blowing agent are present in optimal stoichiometric ratios within every molecule, promoting a rapid and cohesive char formation upon exposure to heat. The inclusion of silicon further reinforces the char layer, creating a ceramic-like barrier that provides superior insulation against oxygen and heat transfer, effectively preventing melt dripping and sustaining the structural integrity of the polymer during a fire event.
Mechanistic Insights into P-N-Si Synergistic Char Formation
The efficacy of this flame retardant relies on a sophisticated intramolecular synergistic mechanism where phosphorus, nitrogen, and silicon function in concert to suppress combustion through both condensed and gas phase actions. Upon thermal decomposition, the phosphorus components generate polyphosphoric acid derivatives that catalyze the dehydration and carbonization of the polymer matrix and the triazine/cage carbon sources, leading to the rapid formation of a swollen, multicellular char layer. Simultaneously, the nitrogen-rich triazine rings release inert gases such as ammonia and nitrogen, which dilute the concentration of combustible volatiles and oxygen near the burning surface, effectively stifling the flame propagation. 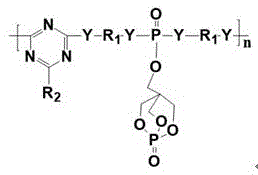 Crucially, the silicon moieties undergo oxidation to form silica or silicate networks that interpenetrate the carbonaceous char, significantly enhancing its mechanical strength and thermal resistance, thereby preventing the char from cracking or collapsing under the stress of evolving gases.
Crucially, the silicon moieties undergo oxidation to form silica or silicate networks that interpenetrate the carbonaceous char, significantly enhancing its mechanical strength and thermal resistance, thereby preventing the char from cracking or collapsing under the stress of evolving gases.
From a purity and impurity profile perspective, the stepwise synthesis allows for precise control over the substitution patterns on the triazine ring, minimizing the presence of unreacted chlorines or low-molecular-weight oligomers that could act as plasticizers or degradation initiators. The use of acid scavengers like triethylamine during the nucleophilic substitution steps ensures the complete neutralization of HCl byproducts, which is critical for preventing corrosion in processing equipment and avoiding acid-catalyzed degradation of the final polymer product. This high level of chemical definition results in a product with a decomposition onset temperature exceeding 300°C under nitrogen atmosphere, making it compatible with high-temperature engineering thermoplastics like polyamides and polyesters where traditional IFRs would decompose prematurely during extrusion or molding.
How to Synthesize Triazine-Based P-N-Si Flame Retardant Efficiently
The synthesis protocol described in the patent involves a multi-stage batch process that prioritizes temperature control and stoichiometric precision to maximize yield and molecular weight consistency. The procedure begins with the low-temperature activation of phosphorus oxychloride followed by the careful addition of the cage-like alcohol to prevent exothermic runaway, ensuring the formation of the mono-substituted phosphorus intermediate with high selectivity. Subsequent steps involve the coupling of this intermediate with diamines and the parallel preparation of a silane-modified triazine core, which are finally condensed under elevated temperatures to drive the polymerization to completion. For detailed operational parameters, solvent choices, and purification workflows, please refer to the standardized synthesis guide below which outlines the critical process controls necessary for reproducible manufacturing.
- Synthesize the phosphorus-containing intermediate by reacting phosphorus oxychloride with PEPA and an acid scavenger at low temperatures.
- React the phosphorus intermediate with a diamine or diol to form a phosphorus-containing diamine precursor.
- Perform stepwise substitution on cyanuric chloride with an aminosilane coupling agent to introduce the silicon component.
- Condense the silicon-containing triazine intermediate with the phosphorus-diamine precursor under controlled heating to finalize the polymer structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this unified P-N-Si molecular architecture offers substantial strategic benefits by simplifying the raw material portfolio and reducing the complexity of the compounding process. By integrating multiple functional roles into a single additive, manufacturers can eliminate the need to source, store, and handle separate acid sources and carbonizing agents, thereby reducing inventory costs and minimizing the risk of formulation errors due to incorrect blending ratios. The robust thermal stability of the final product also translates to cost reduction in flame retardant manufacturing by allowing for higher throughput rates during polymer compounding, as the material can withstand the shear and heat of twin-screw extrusion without significant degradation or discoloration.
- Cost Reduction in Manufacturing: The synthetic route utilizes commodity chemicals such as cyanuric chloride, phosphorus oxychloride, and common aminosilanes, which are readily available in the global chemical market at competitive price points, ensuring a stable cost base for production. The elimination of expensive transition metal catalysts and the reliance on simple precipitation and filtration for purification significantly lowers the operational expenditure associated with waste treatment and solvent recovery. Furthermore, the high yield reported in the patent examples, consistently exceeding 90%, indicates a highly efficient atom economy that minimizes raw material waste and maximizes the output per batch, directly contributing to improved gross margins for large-scale producers.
- Enhanced Supply Chain Reliability: Sourcing a single, high-performance multifunctional additive reduces the dependency on multiple suppliers for different flame retardant components, thereby mitigating the risk of supply disruptions caused by logistics bottlenecks or regional shortages. The chemical stability of the intermediate species allows for flexible manufacturing scheduling, as certain precursors can be stockpiled without significant degradation, providing a buffer against demand fluctuations. This consolidation of functionality into one SKU simplifies the logistics of delivering high-purity flame retardants to customers, reducing lead time for high-purity flame retardants by streamlining the quality control and shipping processes associated with multi-component kits.
- Scalability and Environmental Compliance: The process is inherently scalable as it relies on standard unit operations such as stirred tank reactions, dropwise addition, and solid-liquid separation, which are easily replicated from pilot plant to commercial scale without requiring specialized high-pressure or cryogenic equipment. The halogen-free nature of the final product ensures compliance with RoHS, REACH, and other stringent environmental directives, removing regulatory barriers to entry in sensitive markets like automotive interiors and consumer electronics. Additionally, the reduced generation of hazardous byproducts and the ability to recycle solvents like acetonitrile contribute to a lower environmental footprint, aligning with corporate sustainability goals and reducing the costs associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced flame retardant technology in industrial polymer applications. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering a clear understanding of the material's capabilities and processing requirements. Understanding these factors is essential for R&D teams evaluating the feasibility of incorporating this additive into new polymer formulations or upgrading existing fire-resistant product lines.
Q: How does the cage structure improve thermal stability compared to traditional IFRs?
A: The incorporation of the PEPA cage structure significantly enhances thermal stability by providing a rigid, bulky framework that resists early decomposition. Unlike linear carbon sources that may volatilize prematurely, this bicyclic structure ensures that the acid source and carbon source remain intact until the critical ignition temperature is reached, facilitating a more robust char layer formation.
Q: What are the solubility advantages of this high molecular weight阻燃剂?
A: Traditional intumescent flame retardants often suffer from high water solubility and migration issues, leading to blooming on the polymer surface over time. This novel P-N-Si compound possesses a high molecular weight and a polymeric nature, which drastically reduces its solubility in water and organic solvents, ensuring long-term durability and consistent flame retardant performance within the polymer matrix.
Q: Can this flame retardant be used without additional synergists?
A: Yes, the molecular design integrates the acid source (phosphorus), carbon source (triazine and cage structure), and gas source (nitrogen) into a single molecule. This intramolecular synergy eliminates the need for external compounding with ammonium polyphosphate, simplifying the formulation process while maintaining high efficiency in char formation and smoke suppression.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorus-Nitrogen-Silicon Flame Retardant Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating laboratory-scale innovations into robust, commercial-grade solutions that meet the rigorous demands of the global polymer industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of reactivity and stability required for this P-N-Si synthesis is maintained at every batch size. We operate stringent purity specifications and utilize rigorous QC labs to verify that every shipment meets the exact thermal and compositional profiles defined by the patent, guaranteeing consistent performance in your final polymer products.
We invite you to collaborate with our technical procurement team to explore how this next-generation flame retardant can optimize your current formulations and reduce overall system costs. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific production volumes, and let us provide you with specific COA data and route feasibility assessments to accelerate your project timeline. By partnering with us, you gain access to a supply chain that prioritizes reliability, technical excellence, and the continuous improvement of sustainable fire safety solutions.
