Advanced Acyl-Substituted Pyrazoline Sulfonium Salts: Enabling Efficient LED-Driven Hybrid Polymerization for Industrial Coatings
Advanced Acyl-Substituted Pyrazoline Sulfonium Salts: Enabling Efficient LED-Driven Hybrid Polymerization for Industrial Coatings
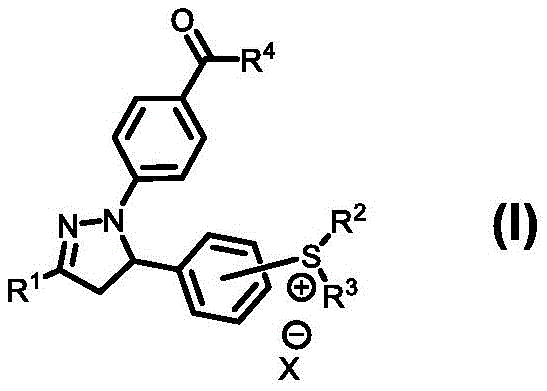
The rapid transition from traditional mercury-vapor lamps to Light Emitting Diode (LED) technology in the photocuring industry has created an urgent demand for photoinitiators capable of absorbing longer wavelengths while maintaining high reactivity. Patent CN112574110B introduces a groundbreaking class of N-1-position acyl-substituted pyrazoline sulfonium salt derivatives, represented by Formula (I), which address these critical technological gaps. These novel compounds are engineered to possess a significantly red-shifted absorption spectrum, ensuring optimal compatibility with commercial LED light sources commonly used in modern electronic packaging, printing inks, and 3D printing applications. Unlike conventional photoinitiators that often require complex blends to achieve hybrid curing, this single-molecule solution integrates both cationic and radical initiating functionalities, streamlining formulation chemistry and enhancing the performance of UV-Vis-LED curable coatings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photocuring technologies have long relied on a dichotomy between free-radical and cationic mechanisms, each presenting distinct operational challenges that complicate industrial manufacturing processes. Free-radical photocuring, while offering a wide selection of monomers and rapid curing speeds, suffers severely from oxygen inhibition, where atmospheric oxygen quenches active radical species, leading to incomplete surface curing and tacky finishes that compromise product quality. Conversely, cationic photocuring is immune to oxygen inhibition and exhibits low volume shrinkage, yet it is notoriously sensitive to moisture and temperature fluctuations, often requiring high-temperature post-curing steps to achieve full conversion. Furthermore, most commercial photoinitiators are optimized for the short-wavelength output of mercury lamps, rendering them inefficient under the longer-wavelength emission of energy-saving LED arrays, which necessitates higher loading levels or the addition of sensitizers that increase formulation costs and residual small molecule content.
The Novel Approach
The innovative strategy outlined in the patent data leverages the unique electronic properties of the pyrazoline heterocyclic ring to overcome these historical limitations through molecular engineering. By introducing a sulfonium salt group capable of generating strong acids for cationic polymerization and simultaneously incorporating a ketone group that functions as a Type II radical photoinitiator, the invention achieves true hybrid polymerization within a single molecular entity. This dual-functionality not only mitigates the oxygen sensitivity of radical curing and the moisture sensitivity of cationic curing but also ensures that the ketone group remains intact after the initial photolysis of the sulfonium salt, allowing it to continue initiating radical polymerization. The result is a robust curing profile that combines the speed of radical systems with the dimensional stability of cationic systems, all while being excitable by cost-effective and environmentally friendly LED light sources without the need for complex multi-component initiator packages.
Mechanistic Insights into Intramolecular Charge Transfer and Dual Initiation
The exceptional photochemical performance of these acyl-substituted pyrazoline sulfonium salts stems from a sophisticated intramolecular charge transfer (ICT) phenomenon occurring between the nitrogen atom at the N-1 position and the carbon atom at the 3-position of the five-membered heterocyclic ring. This electronic delocalization significantly lowers the energy gap required for excitation, effectively shifting the absorption maximum into the visible and near-UV range (365-405 nm) where LED sources emit peak intensity. Upon irradiation, the sulfonium moiety undergoes homolytic cleavage to generate a strong Bronsted acid, which serves as the catalyst for the ring-opening polymerization of epoxy monomers, driving the cationic curing pathway. Simultaneously, the acyl group attached to the pyrazoline nitrogen absorbs photon energy to enter an excited triplet state, facilitating hydrogen abstraction or electron transfer processes that generate free radicals necessary for the polymerization of acrylate and methacrylate monomers.
This mechanistic duality is further reinforced by the stability of the degradation products; unlike some conventional initiators that decompose into inactive fragments, the ketone-containing remnants of this molecule retain their photo-initiating capability, ensuring a sustained cure even after the primary sulfonium activation event. The synthetic versatility allowed by the variable R groups (R1 through R5) enables fine-tuning of the HOMO-LUMO energy levels, permitting chemists to optimize the absorption cross-section for specific LED wavelengths used in different industrial sectors. This level of control over the photophysical properties ensures that the material can be adapted for diverse applications ranging from high-speed printing inks, where surface cure is paramount, to thick-section composite curing, where deep penetration and low shrinkage are critical for structural integrity.
How to Synthesize Acyl-Substituted Pyrazoline Sulfonium Salt Derivatives Efficiently
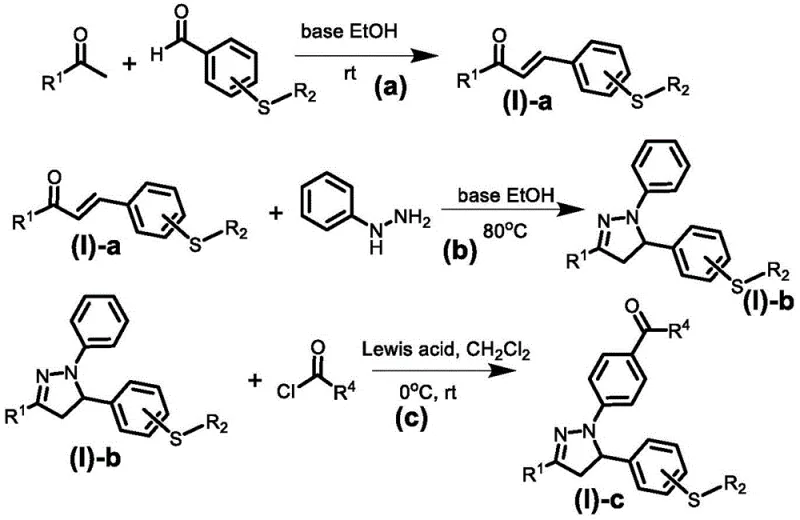
The preparation of these high-performance photoinitiators follows a logical and scalable four-step synthetic sequence that utilizes commodity chemicals and standard unit operations familiar to process chemists. The route begins with a base-catalyzed Claisen-Schmidt condensation to form a chalcone intermediate, followed by cyclization with phenylhydrazine to construct the pyrazoline core. Subsequent N-acylation introduces the radical-initiating functionality, and the final quaternization of the sulfur atom installs the cationic-initiating sulfonium group. This modular approach allows for the easy variation of substituents to tailor the final product's solubility and reactivity. For a detailed breakdown of reaction conditions, stoichiometry, and purification protocols, please refer to the standardized synthesis guide below.
- Perform Claisen-Schmidt condensation between substituted acetophenone and thioether-substituted benzaldehyde in ethanol with base catalysis to form the chalcone intermediate.
- Execute cyclization by reacting the chalcone intermediate with phenylhydrazine in ethanol under reflux conditions to construct the pyrazoline core structure.
- Conduct Friedel-Crafts acylation on the pyrazoline nitrogen using acyl chloride and Lewis acid catalyst in dichloromethane to introduce the radical-initiating ketone group.
- Complete the synthesis via quaternization of the sulfur atom using trifluoromethanesulfonic ester or formate, followed by anion exchange to obtain the final sulfonium salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this acyl-substituted pyrazoline sulfonium salt technology offers profound advantages that extend beyond mere technical performance metrics. The ability to replace dual-initiator systems (one for radical, one for cationic) with a single multifunctional molecule drastically simplifies the raw material inventory management for coating manufacturers, reducing the complexity of quality control testing and minimizing the risk of formulation errors. Furthermore, the synthesis route described in the patent relies on widely available starting materials such as substituted acetophenones and benzaldehydes, which are produced on a massive global scale, thereby insulating the supply chain from the volatility often associated with exotic or specialized fine chemical intermediates. This reliance on commodity feedstocks ensures a stable and continuous supply flow, which is critical for maintaining uninterrupted production schedules in high-volume industrial settings.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the elimination of redundant processing steps and the reduction of total additive loading in the final formulation. By consolidating two initiation mechanisms into one molecule, formulators can reduce the total weight percentage of photoinitiators required, which directly lowers the bill of materials cost for UV-curable coatings and inks. Additionally, the synthetic process operates under mild conditions, typically ranging from room temperature to moderate heating (80°C), which significantly reduces energy consumption during the manufacturing of the photoinitiator itself compared to high-temperature or high-pressure alternatives. The high yields reported in the experimental examples across multiple steps indicate a material-efficient process that minimizes waste generation and maximizes the output per batch, contributing to substantial overall cost savings.
- Enhanced Supply Chain Reliability: The robustness of the synthetic pathway, which avoids the use of sensitive transition metal catalysts or hazardous reagents that require special handling and disposal, enhances the reliability of the supply chain. The absence of heavy metals simplifies the regulatory compliance landscape, particularly for applications in food packaging or electronics where strict limits on metal residues exist. This chemical simplicity translates to fewer potential bottlenecks in production, as the process is less susceptible to the supply fluctuations of niche catalysts. Moreover, the final product's stability and ease of handling facilitate safer logistics and storage, reducing the risks associated with transportation and warehousing of hazardous photoinitiating compounds.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing common solvents like ethanol and dichloromethane that are easily recovered and recycled in industrial distillation columns, aligning with green chemistry principles and reducing the environmental footprint of production. The high atom economy of the condensation and cyclization steps ensures that a large proportion of the input mass is incorporated into the final product, minimizing the generation of hazardous byproducts. This environmental efficiency is increasingly becoming a key procurement criterion for multinational corporations aiming to meet sustainability goals, making this photoinitiator a future-proof choice for long-term supply agreements. The compatibility with LED curing also supports the end-user's sustainability initiatives by enabling lower energy consumption during the application phase.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel photoinitiator technology in industrial workflows. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance expectations and integration strategies. Understanding these nuances is essential for R&D teams evaluating the switch from legacy mercury-lamp systems to modern LED-curable formulations.
Q: How does this photoinitiator improve upon traditional mercury lamp systems?
A: Unlike traditional photoinitiators designed for short-wavelength mercury lamps, this acyl-substituted pyrazoline sulfonium salt exhibits a red-shifted absorption spectrum that aligns perfectly with commercial LED light sources (365-405 nm), enabling energy-efficient curing without ozone generation.
Q: What is the advantage of the dual-cure mechanism in this molecule?
A: The molecule integrates both a sulfonium salt group for cationic polymerization and a ketone group for radical polymerization. This allows a single additive to initiate hybrid curing, eliminating oxygen inhibition issues associated with pure radical systems and reducing volume shrinkage compared to pure cationic systems.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the patented method utilizes readily available raw materials like substituted acetophenones and benzaldehydes, and employs standard reactions such as condensation and acylation under mild conditions (room temperature to 80°C), making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acyl-Substituted Pyrazoline Sulfonium Salt Supplier
As the global demand for high-performance, LED-compatible photoinitiators continues to surge, NINGBO INNO PHARMCHEM stands ready to support your transition to next-generation curing technologies with our advanced manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pilot testing phase or full-scale industrial rollout. Our facility is equipped with stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to guarantee that every batch of acyl-substituted pyrazoline sulfonium salts meets the exacting standards required for electronic and coating applications.
We invite you to engage with our technical procurement team to discuss how this innovative chemistry can drive value in your specific product lines. By requesting a Customized Cost-Saving Analysis, you can quantify the potential reductions in formulation complexity and energy usage tailored to your current operations. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this superior photoinitiator into your supply chain.
