Advanced Bis-Carbazole Oxime Ester Photoinitiators for High-Efficiency LED Curing Systems
The rapid transition towards energy-efficient manufacturing has placed immense pressure on the chemical industry to develop materials compatible with LED curing technologies. Patent CN112961099B introduces a groundbreaking class of bis-carbazole oxime ester photoinitiators designed specifically to address the spectral mismatch issues prevalent in current UV-Vis-LED systems. Unlike traditional photoinitiators that rely on mercury lamp spectra, this novel molecular architecture leverages the extensive conjugation of the carbazole unit to achieve absorption maxima that align perfectly with commercial LED emission bands, particularly in the 395-405nm range. This technological leap is not merely an incremental improvement but a fundamental restructuring of how radiation-curing formulations are engineered for high-performance applications such as woodware coatings, ink-jet printing, and flexible electronics. For R&D directors and procurement specialists, understanding the synthesis and application potential of this specific chemical class is critical for maintaining competitiveness in the evolving landscape of electronic materials and specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoinitiator systems have long struggled with the inefficiency of energy transfer when paired with modern LED light sources. Conventional initiators, often designed for broad-spectrum mercury lamps, exhibit poor absorption in the specific narrow bands emitted by high-power LEDs, leading to incomplete curing, tacky surfaces, and the need for excessive initiator loading which compromises the physical properties of the final polymer matrix. Furthermore, many existing solutions suffer from significant migration issues, where unreacted initiator molecules leach out of the cured film, causing odor problems and potential toxicity concerns in food packaging or sensitive electronic applications. The reliance on older chemical scaffolds also limits the ability to tune the solubility and compatibility of the initiator within complex resin systems, often requiring the use of hazardous solvents to achieve adequate dispersion. These limitations create a bottleneck for manufacturers attempting to scale up green coating processes, as the inefficiency translates directly into higher energy consumption and increased waste generation during the production cycle.
The Novel Approach
The innovation disclosed in CN112961099B overcomes these barriers by engineering a dicarbazole core connected via a conjugated carbon-carbon double bond system, effectively extending the electron delocalization across the molecule. This structural modification induces a significant red-shift in the absorption spectrum, ensuring that the photoinitiator captures the photon energy from LED sources with maximum efficiency. By incorporating the oxime ester functional group, the molecule gains the ability to undergo efficient alpha-cleavage upon irradiation, generating free radicals that initiate polymerization rapidly even at low concentrations. This approach not only enhances the curing speed, allowing for high-throughput manufacturing lines, but also drastically reduces the residual odor, as evidenced by patent testing showing odor levels near zero. The versatility of this design allows for further functionalization of the carbazole nitrogen or the phenyl rings, enabling chemists to tailor the solubility profile for specific aqueous or solvent-based formulations without sacrificing photo-reactivity.
Mechanistic Insights into Bis-Carbazole Oxime Ester Photoinitiation
The core of this technology lies in the sophisticated interplay between the carbazole chromophore and the oxime ester cleavage mechanism. The carbazole unit acts as a powerful electron donor, while the conjugated ketone and oxime ester moieties serve as electron acceptors, creating an intramolecular charge transfer (ICT) state upon light absorption. This ICT state lowers the energy gap required for excitation, making the molecule sensitive to visible and near-UV light rather than just short-wave UV. Once excited, the oxime ester bond undergoes homolytic cleavage to produce an iminyl radical and an acyloxy radical, both of which are highly reactive towards ethylenically unsaturated monomers like acrylates. The rigidity of the bis-carbazole structure prevents non-radiative decay pathways, ensuring that the absorbed energy is channeled primarily into bond cleavage rather than heat dissipation. This high quantum yield is essential for thick-film curing applications where light penetration is limited, as it ensures that radicals are generated deep within the coating layer.
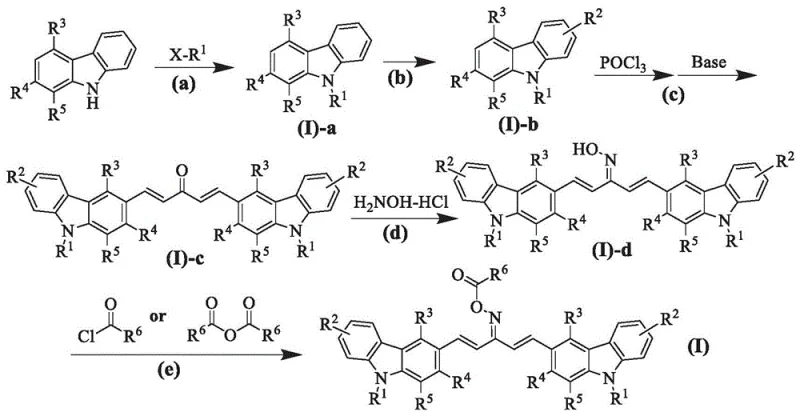
Controlling the impurity profile during the synthesis of these complex molecules is paramount for ensuring consistent curing performance. The multi-step synthesis involves precise control over reaction conditions to prevent side reactions such as over-alkylation or polymerization of the conjugated double bonds. The patent outlines a robust purification strategy involving column chromatography and recrystallization to isolate the target isomers, specifically the (1E, 4E) configuration which offers the optimal planar geometry for conjugation. Impurities that disrupt this planarity can act as quenchers, absorbing light without generating radicals, which would degrade the overall efficiency of the photoinitiator system. By adhering to the strict stoichiometric ratios and temperature controls described in the patent, manufacturers can achieve high purity levels that translate to predictable and reliable curing kinetics in the final application, minimizing the risk of batch-to-batch variability in industrial production.
How to Synthesize Bis-Carbazole Oxime Ester Efficiently
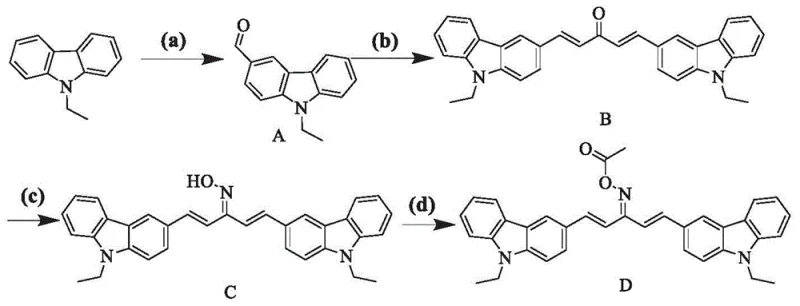
The synthesis of these high-performance photoinitiators follows a logical progression of organic transformations that are amenable to scale-up. The process begins with the N-alkylation of carbazole to protect the nitrogen and tune solubility, followed by a Vilsmeier-Haack formylation to introduce the aldehyde functionality necessary for backbone extension. The subsequent aldol condensation with acetone links two carbazole units, establishing the critical conjugated system. Finally, conversion to the oxime and subsequent esterification yields the active photoinitiator. Each step has been optimized in the patent examples to maximize yield and minimize waste, using common reagents like phosphorus oxychloride, hydroxylamine hydrochloride, and acetic anhydride. The detailed standardized synthesis steps for the specific embodiment of (1E, 4E)-1,5-bis(9-ethyl-9H-carbazol-3-yl)penta-1,4-dien-3-one O-acetyl oxime are outlined below to guide process engineers in replicating this high-value chemistry.
- Perform N-alkylation of carbazole with alkyl halides under basic conditions to form N-alkyl carbazole intermediates.
- Execute Vilsmeier-Haack formylation using POCl3 followed by aldol condensation with acetone to establish the conjugated ketone backbone.
- Convert the ketone to an oxime using hydroxylamine hydrochloride, followed by esterification with acyl chloride or anhydride to finalize the photoinitiator structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this bis-carbazole technology represents a strategic opportunity to optimize manufacturing costs and enhance product quality without compromising on performance. The ability to cure effectively with LED sources translates directly into substantial energy savings, as LEDs consume significantly less power and have a longer operational lifespan compared to traditional mercury arc lamps. Furthermore, the high reactivity of these initiators allows for faster line speeds in coating and printing operations, increasing overall throughput and reducing the cost per unit produced. The low odor and low migration characteristics also reduce the need for extensive post-curing ventilation or secondary processing steps, simplifying the production workflow and lowering facility overheads. These qualitative advantages position the material as a premium yet cost-effective solution for high-end applications where performance and regulatory compliance are non-negotiable.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available starting materials like carbazole and acetone significantly lowers the raw material cost base. Additionally, the high efficiency of the photoinitiator means that lower loading levels are required to achieve the same degree of cure, further reducing the formulation cost. The compatibility with LED curing systems removes the dependency on costly and hazardous mercury lamps, leading to long-term operational expenditure savings. By streamlining the synthesis route to fewer steps with high yields, the overall production cost is minimized, making this a highly attractive option for cost-sensitive mass market applications.
- Enhanced Supply Chain Reliability: The synthetic route relies on commodity chemicals that are widely available from multiple global suppliers, reducing the risk of supply chain disruptions associated with specialized or scarce reagents. The robustness of the reaction conditions, which do not require extreme temperatures or pressures, ensures that production can be maintained consistently across different manufacturing sites. This geographical flexibility allows for a more resilient supply chain, capable of adapting to regional demand fluctuations without significant lead time penalties. The stability of the final photoinitiator product also ensures a long shelf life, reducing waste due to expiration and allowing for strategic stockpiling during periods of low demand.
- Scalability and Environmental Compliance: The synthesis process is designed with green chemistry principles in mind, generating minimal hazardous waste and utilizing solvents that can be easily recovered and recycled. The absence of heavy metals in the final product simplifies regulatory compliance for applications in food packaging and toys, where strict migration limits are enforced. The scalability of the process has been demonstrated through the successful preparation of multi-gram quantities in the patent examples, indicating a clear path to ton-scale production. This environmental profile aligns with the increasing corporate sustainability goals of major end-users, making it a preferred choice for companies looking to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of bis-carbazole oxime ester photoinitiators in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for integrating this new chemistry into existing product lines and maximizing its potential benefits. The insights provided here aim to clarify the operational parameters and performance expectations associated with this advanced material class.
Q: What is the primary advantage of this bis-carbazole structure over traditional photoinitiators?
A: The bis-carbazole structure provides a rigid conjugated plane that significantly red-shifts the absorption spectrum, allowing for efficient matching with long-wave LED emission wavelengths (395-405nm) which traditional initiators often miss.
Q: Does this photoinitiator require thermal post-curing?
A: No, the patent data indicates that the system cures efficiently under UV-Vis-LED irradiation at room temperature, eliminating the need for energy-intensive thermal post-curing processes.
Q: How does the odor profile compare to standard benzophenone initiators?
A: Experimental data in the patent shows odor ratings of 0 to 1 (no smell to very mild), which is a substantial improvement over many traditional small-molecule initiators that often leave residual odors in cured films.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-Carbazole Oxime Ester Supplier
As the demand for high-performance LED curing materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge technology and reliable supply. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your transition to this new photoinitiator technology is seamless and risk-free. Our stringent purity specifications and rigorous QC labs ensure that every batch meets the exacting standards required for electronic and coating applications, providing you with the consistency needed for global distribution. By leveraging our expertise in process optimization, we can help you further reduce costs and improve the environmental profile of your final products.
We invite you to engage with our technical procurement team to discuss how this patented technology can be integrated into your specific formulation needs. Request a Customized Cost-Saving Analysis to quantify the potential economic benefits for your operation. Our team is ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing strategies. Let us help you lead the market with superior curing solutions that combine performance, sustainability, and cost-efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
