Scalable Ruthenium-Catalyzed Synthesis of 5-Maleimide Chromone Intermediates for Drug Discovery
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for new drug candidates. Patent CN110590751B discloses a groundbreaking preparation method for 5-maleimide chromone compounds, utilizing a highly efficient ruthenium-catalyzed carbon-hydrogen bond activation strategy. This innovation addresses long-standing synthetic challenges by enabling the direct coupling of chromone and maleimide derivatives without the need for pre-functionalized substrates. The process operates under mild oxidative conditions using air as the terminal oxidant, significantly simplifying the operational complexity typically associated with transition metal catalysis. For R&D directors and process chemists, this technology represents a pivotal shift towards more atom-economical and cost-effective routes for generating biologically active intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of maleimide-fused heterocycles has relied heavily on the use of pre-functionalized starting materials, such as aryl diazonium salts, aryl boronic acids, or halogenated aryl compounds. These traditional approaches necessitate multi-step sequences to install the requisite leaving groups or organometallic handles prior to the coupling event, thereby increasing the overall step count and reducing the cumulative yield. Furthermore, earlier transition metal-catalyzed strategies, particularly those employing rhodium catalysts for Heck-type couplings, often suffered from moderate efficiency, with reported yields frequently ranging between 42% and 68%. The reliance on expensive noble metals like rhodium, combined with the need for strict inert atmosphere conditions to prevent catalyst deactivation, created substantial barriers to cost-effective commercial manufacturing. Additionally, the generation of stoichiometric metallic waste from pre-functionalization steps posed significant environmental and disposal challenges for large-scale operations.
The Novel Approach
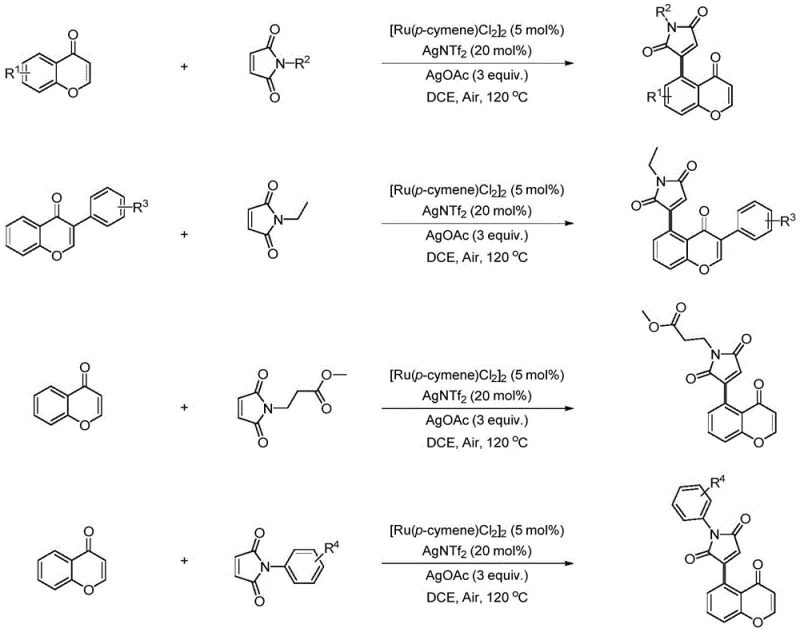
The methodology outlined in the patent introduces a paradigm shift by leveraging the weak coordination ability of the chromone ketone carbonyl group to direct regioselective C-H activation at the C-5 position. By employing a dual-catalyst system comprising [Ru(p-cymene)Cl2]2 and AgNTf2, supplemented with silver acetate as a crucial additive, the reaction achieves direct olefination with maleimides. This approach eliminates the need for pre-halogenation or borylation, streamlining the synthesis into a single convergent step. The use of ruthenium, a more earth-abundant and cost-effective metal compared to rhodium, coupled with the ability to run the reaction under ambient air atmosphere, drastically reduces both raw material costs and engineering overhead. The process demonstrates exceptional versatility, accommodating a wide range of substituents on both the chromone and maleimide rings, making it a powerful tool for library synthesis and lead optimization campaigns.
Mechanistic Insights into Ru-Catalyzed C-H Activation
The catalytic cycle initiates with the generation of an active cationic ruthenium species through the interaction of the dimeric precursor [Ru(p-cymene)Cl2]2 with the silver salt AgNTf2. This active cation subsequently coordinates with the carbonyl oxygen of the chromone substrate, forming a transient complex that positions the metal center in proximity to the C-5 hydrogen atom. This coordination is critical as it lowers the activation energy for the subsequent C-H bond cleavage, leading to the formation of a stable five-membered ruthenacycle intermediate. The presence of silver acetate plays a dual role, acting both as a halide scavenger to maintain the cationic nature of the catalyst and as a base to facilitate the deprotonation step during the metallacycle formation. This chelation-assisted mechanism ensures high regioselectivity, preventing functionalization at other potential sites on the chromone ring.
Following the C-H activation, the maleimide substrate inserts into the carbon-ruthenium bond of the metallacycle, generating a bicyclic intermediate. The final product release occurs through a deprotonation or radical-mediated process assisted by the acetate ligand, regenerating the active catalyst species for the next turnover. This mechanistic pathway effectively bypasses the issue of beta-hydride elimination that typically plagues similar olefination reactions, which would otherwise lead to saturated succinimide byproducts. The robustness of this catalytic system is evidenced by its tolerance to various functional groups, including halogens, esters, and nitro groups, without significant loss in catalytic activity. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as temperature and additive loading, to maximize throughput and minimize impurity profiles in commercial production settings.
How to Synthesize 5-Maleimide Chromone Efficiently
The standardized protocol for this transformation involves charging a pressure-resistant vessel with the chromone substrate and the N-substituted maleimide in a molar ratio optimized to drive the reaction to completion, typically favoring an excess of the maleimide coupling partner. The catalyst system is introduced as [Ru(p-cymene)Cl2]2 at a loading of 5 mol%, alongside 20 mol% of AgNTf2 and 3 equivalents of silver acetate to ensure rapid turnover and high conversion. The reaction is conducted in 1,2-dichloroethane (DCE) or chlorobenzene at an elevated temperature of 120°C, allowing the reaction to proceed to full conversion within a timeframe of 0.5 to 4 hours depending on the electronic nature of the substrates. Detailed standard operating procedures for scaling this reaction from gram to kilogram scale are provided in the technical guide below.
- Charge a pressure-resistant tube with chromone substrate, N-substituted maleimide, [Ru(p-cymene)Cl2]2 catalyst, AgNTf2 co-catalyst, and silver acetate additive.
- Add DCE solvent and heat the reaction mixture to 120°C under air atmosphere with stirring for 0.5 to 4 hours.
- Quench with water, extract with ethyl acetate, dry over sodium sulfate, and purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this ruthenium-catalyzed methodology offers distinct strategic advantages that directly impact the bottom line and operational reliability. The elimination of pre-functionalized starting materials removes the need to source or synthesize expensive halogenated or boronated intermediates, which are often subject to volatile pricing and supply constraints. By utilizing commodity chemicals like unsubstituted chromone and readily available maleimides, manufacturers can secure a more stable and predictable supply chain, reducing the risk of production delays caused by raw material shortages. Furthermore, the simplified workup procedure, which involves standard aqueous extraction and column chromatography, minimizes the consumption of specialized reagents and solvents, contributing to a leaner and more cost-efficient manufacturing process.
- Cost Reduction in Manufacturing: The substitution of expensive rhodium catalysts with more economical ruthenium complexes results in substantial savings on catalyst costs, which is particularly impactful when scaling to multi-kilogram or ton-level production. The ability to operate under air atmosphere eliminates the capital expenditure and ongoing operational costs associated with maintaining inert gas lines and glovebox facilities. Additionally, the high atom economy of the direct C-H activation strategy reduces the generation of chemical waste, lowering the costs associated with waste treatment and environmental compliance. These factors collectively contribute to a significantly reduced cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available and commercially stable starting materials ensures a robust supply chain that is less susceptible to geopolitical or logistical disruptions. The short reaction times, often completing within an hour for optimized substrates, increase the throughput capacity of existing reactor infrastructure, allowing suppliers to respond more rapidly to fluctuating market demands. The simplicity of the reaction setup also reduces the likelihood of batch failures due to operator error or equipment malfunction, ensuring consistent delivery schedules for downstream customers. This reliability is crucial for pharmaceutical companies managing tight development timelines and regulatory submission deadlines.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, having been validated across a diverse range of substrates with consistent high yields, indicating its readiness for commercial scale-up of complex pharmaceutical intermediates. The use of air as the oxidant avoids the safety hazards and handling complexities associated with strong chemical oxidants or high-pressure hydrogenation equipment. Moreover, the reduced waste profile aligns with green chemistry principles, facilitating easier regulatory approval and supporting corporate sustainability goals. The combination of safety, efficiency, and environmental friendliness makes this technology an ideal candidate for sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing clarity on catalyst selection, substrate compatibility, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects.
Q: What are the advantages of using Ruthenium over Rhodium for this synthesis?
A: Ruthenium catalysts offer a significant cost advantage over Rhodium while maintaining high catalytic activity. The patent demonstrates that Ru-catalyzed systems achieve superior yields (up to 95%) compared to previous Rh-catalyzed methods which often ranged between 42% and 68%.
Q: Does this reaction require inert atmosphere conditions?
A: No, one of the key operational benefits of this method is that it proceeds efficiently under air atmosphere. This eliminates the need for expensive inert gas setups like nitrogen or argon gloves boxes, simplifying the engineering requirements for scale-up.
Q: What is the substrate scope for the maleimide component?
A: The method is highly versatile, accommodating N-alkyl (methyl, ethyl, cyclohexyl, benzyl) and N-aryl (phenyl, substituted phenyl) maleimides. Electron-withdrawing and electron-donating groups on the N-aryl ring are generally tolerated.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Maleimide Chromone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates to accelerate your drug discovery programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of 5-maleimide chromone meets the highest international standards, providing you with the confidence needed to advance your clinical candidates. We leverage advanced catalytic technologies like the ruthenium-mediated C-H activation described herein to deliver superior value and performance.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this direct coupling strategy. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in delivering complex organic solutions.
