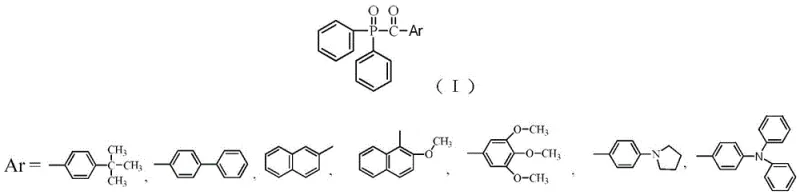
Advanced Acylphosphine Oxide Photoinitiators: Enhancing LED Curing Efficiency and Stability
The rapid evolution of photocuring technology has necessitated the development of advanced photoinitiators capable of matching the spectral output of modern LED light sources. Patent CN110950977B introduces a groundbreaking class of acylphosphine oxide photoinitiators designed to overcome the limitations of traditional systems like TPO. By strategically modifying the molecular architecture through the introduction of alkyl, alkoxy, or amino substituents, this invention achieves a significant red-shift in light absorption wavelengths. This structural innovation not only enhances initiation efficiency in the visible region but also drastically reduces initiator mobility, addressing critical safety concerns in food packaging and dental applications. As a reliable photoinitiator supplier, understanding these structural nuances is key to selecting the right additive for high-performance polymer formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional acylphosphine oxide photoinitiators, such as TPO and IRGACURE 819, have long served as industry standards for UV curing; however, they exhibit distinct deficiencies when applied to modern LED curing systems. The primary limitation lies in their absorption spectra, which are predominantly concentrated in the near-ultraviolet region between 350 nm and 400 nm. This spectral mismatch results in suboptimal energy utilization when paired with visible light LED sources, leading to incomplete curing and reduced mechanical properties in the final polymer matrix. Furthermore, conventional initiators often suffer from high mobility within the cured network. This high migration rate poses significant regulatory and safety challenges, particularly in applications involving direct human contact or food storage, where leaching of unreacted initiator molecules is strictly prohibited. Additionally, the storage stability of some traditional formulations can be compromised due to their specific structural characteristics, limiting their shelf life and operational reliability in large-scale manufacturing environments.
The Novel Approach
The innovative approach detailed in the patent addresses these shortcomings by engineering photoinitiators with enhanced electron cloud density and expanded conjugated systems. By incorporating specific functional groups such as alkylamino, alkoxy, or arylamino moieties onto the aromatic ring, the resulting molecules exhibit a bathochromic shift, moving their maximum absorption peaks into the visible light region. This red-shift ensures superior compatibility with LED light sources, thereby maximizing photon absorption and radical generation efficiency. Moreover, the increased molecular weight and steric bulk associated with these substituents effectively anchor the initiator within the polymer network, significantly reducing mobility. This dual improvement in optical performance and physical stability represents a paradigm shift in cost reduction in polymer additive manufacturing, as it allows for lower loading levels while achieving superior curing depth and material safety profiles.
Mechanistic Insights into Substituent-Driven Spectral Tuning
The core mechanism behind the enhanced performance of these novel photoinitiators lies in the electronic modulation of the acylphosphine oxide core. When electron-donating groups such as methoxy or pyrrolidinyl are introduced at the para- or ortho-positions of the benzoyl ring, they increase the electron density across the conjugated pi-system. This elevation in electron density lowers the energy gap between the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO). Consequently, the energy required for electronic excitation decreases, resulting in the absorption of longer wavelengths of light. This phenomenon is critical for aligning the initiator's activation profile with the emission spectrum of high-power LEDs, which typically operate in the 405 nm to 450 nm range. The result is a more efficient cleavage of the phosphorus-carbon bond upon irradiation, generating the necessary free radicals to initiate polymerization with greater speed and depth.
Beyond spectral tuning, the structural modifications play a pivotal role in controlling impurity profiles and migration behavior. The synthesis pathway utilizes a nucleophilic addition followed by oxidation, which, when controlled precisely, yields high-purity intermediates. The presence of bulky substituents, such as the tert-butyl group or the naphthyl ring, creates steric hindrance that physically restricts the diffusion of the initiator molecule within the cross-linked polymer matrix. This restriction is quantified by a reduction in mobility by up to two orders of magnitude compared to unsubstituted analogues. For R&D teams focusing on high-purity OLED material or biomedical grade polymers, this mechanism ensures that the final product meets stringent regulatory standards for extractables and leachables, thereby enhancing the overall safety and marketability of the end-use application.
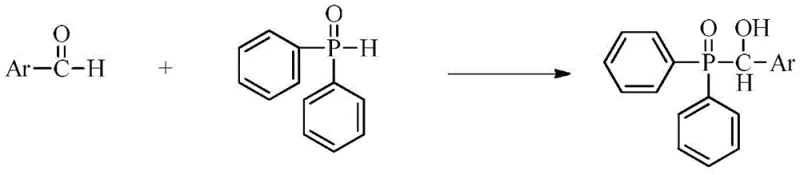

How to Synthesize 2-Methoxy-1-Naphthoyl-Diphenylphosphine Oxide Efficiently
The synthesis of these advanced photoinitiators follows a robust two-step protocol that is amenable to scale-up. The process begins with the nucleophilic addition of diphenylphosphine oxide to a substituted aromatic aldehyde, such as 2-methoxy-1-naphthaldehyde, in a suitable solvent system. This reaction proceeds under mild conditions to form a hydroxy-phosphine oxide intermediate, which is then isolated and purified. The second step involves the oxidation of this intermediate to the corresponding ketone using an oxidizing agent like activated manganese dioxide. This methodology avoids the use of hazardous heavy metal catalysts often found in older protocols, aligning with green chemistry principles. For detailed operational parameters and stoichiometry, please refer to the standardized synthesis guide below.
- React substituted aromatic aldehyde with diphenylphosphine oxide in a solvent like ethyl acetate at 15-50°C to form the hydroxy intermediate.
- Oxidize the intermediate using activated manganese dioxide or a vanadium/peroxide system in dichloromethane to yield the final acylphosphine oxide.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers substantial strategic benefits. The reliance on readily available aromatic aldehydes and diphenylphosphine oxide ensures a stable raw material supply, mitigating the risks associated with sourcing exotic or proprietary precursors. The reaction conditions are relatively mild, with the first step occurring at ambient temperatures and the second step requiring only moderate heating or room temperature stirring. This translates to significantly reduced energy consumption during manufacturing, contributing to lower operational expenditures and a smaller carbon footprint. Furthermore, the simplified work-up procedures, which involve standard filtration and crystallization techniques, streamline the production process and reduce the need for complex purification equipment.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of common oxidants like manganese dioxide drastically simplify the cost structure of the final product. By avoiding complex downstream processing steps required to remove trace metal contaminants, manufacturers can achieve substantial cost savings while maintaining high product purity. The higher initiation efficiency of these new initiators also implies that lower dosages may be required to achieve the same curing performance as traditional initiators, further driving down the cost-in-use for formulators.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted benzaldehydes and naphthaldehydes, are commodity chemicals with established global supply chains. This availability ensures that production schedules are not disrupted by raw material shortages. Additionally, the robustness of the synthetic method, which tolerates a range of reaction conditions without significant yield loss, enhances the predictability of manufacturing output. This reliability is crucial for maintaining continuous supply to downstream customers in the coatings and 3D printing industries.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to kilogram scales in the patent examples. The use of standard organic solvents like ethyl acetate and dichloromethane allows for easy solvent recovery and recycling, minimizing waste generation. Moreover, the absence of heavy metal residues in the final product simplifies regulatory compliance for applications in food contact materials and medical devices, reducing the time and cost associated with safety testing and certification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these novel photoinitiators. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating these materials into existing production lines.
Q: How does the new structure improve LED curing compared to TPO?
A: By introducing electron-donating groups like alkoxy or amino substituents, the new initiators achieve a red-shift in absorption wavelength, better matching visible LED sources and improving initiation efficiency.
Q: What are the mobility advantages of these new photoinitiators?
A: The modified structures exhibit significantly reduced mobility (up to two orders of magnitude lower than TPO), minimizing migration issues in sensitive applications like food packaging and dental materials.
Q: Are the raw materials for this synthesis readily available?
A: Yes, the synthesis utilizes commercially available substituted aromatic aldehydes and diphenylphosphine oxide, ensuring a robust and scalable supply chain for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acylphosphine Oxide Photoinitiator Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance photoinitiators play in the advancement of polymer science. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial application is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require custom derivatives for specialized LED curing applications or bulk quantities of standard initiators, our infrastructure is designed to meet your most demanding requirements.
We invite you to collaborate with us to optimize your formulation costs and performance. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced acylphosphine oxide photoinitiators can enhance your product portfolio while ensuring supply chain security.
