Commercializing High-Efficiency Sulfur-Linked Benzophenone Photoinitiators for UV Curing
Commercializing High-Efficiency Sulfur-Linked Benzophenone Photoinitiators for UV Curing
The rapid evolution of ultraviolet (UV) curing technology has created an urgent demand for photoinitiators that offer superior efficiency, stability, and compatibility within complex polymer matrices. Patent CN1789245A introduces a groundbreaking class of sulfur-containing polymerizable benzophenone photoinitiators that address the critical limitations of traditional small-molecule systems. By strategically integrating a thioether linkage and a polymerizable maleimide moiety into the benzophenone backbone, this innovation delivers a substantial red shift in ultraviolet absorption and drastically improved initiation kinetics. For R&D directors and procurement specialists seeking reliable polymer synthesis additives supplier partnerships, understanding the structural advantages of these compounds is essential for next-generation coating and electronic material formulations.
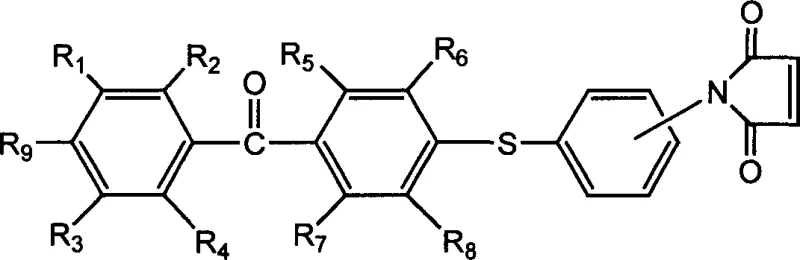
Traditional photoinitiators often suffer from volatility and migration over time, leading to product degradation, unpleasant odors, and potential toxicity in sensitive applications like food packaging or medical devices. The molecular design detailed in this patent overcomes these hurdles by creating a macromer-compatible structure. The presence of the sulfur atom not only modifies the electronic properties of the benzophenone core to enhance light absorption but also serves as a robust linkage point. Furthermore, the terminal maleimide group allows the initiator to become an integral part of the cured polymer network, effectively locking it in place. This dual functionality positions these compounds as ideal candidates for cost reduction in electronic chemical manufacturing, where long-term stability and low extractables are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied on simple benzophenone derivatives or acryloyl chloride-modified initiators to achieve UV curing. However, these conventional approaches present significant technical and safety challenges. Small molecule initiators are prone to sublimation and migration, which reduces the effective concentration of the active species within the coating over its shelf life. Additionally, previous attempts to create polymerizable versions often involved the use of acryloyl chloride, a reagent known for its high toxicity and corrosive nature, complicating handling and waste disposal protocols. The resulting products from these older methods frequently exhibited limited red-shifting capabilities, meaning they required higher energy UV sources or longer exposure times to achieve full cure, thereby limiting throughput in high-speed industrial lines.
The Novel Approach
The synthetic strategy outlined in the patent data offers a sophisticated alternative that bypasses the hazards of acid chlorides while delivering superior performance. By utilizing aminothiophenols and halogenated benzophenones as starting materials, the process constructs a stable thioether bridge that is less susceptible to hydrolytic degradation than ester linkages found in acrylate-based initiators. The subsequent introduction of the maleimide ring via maleic anhydride provides a highly reactive double bond for copolymerization without the need for hazardous acylating agents. This novel approach results in a high-purity OLED material precursor or coating additive that exhibits initiation speeds nearly four times faster than standard benzophenone, enabling faster line speeds and lower energy consumption in UV curing facilities.
Mechanistic Insights into Nucleophilic Substitution and Imidization
The synthesis of these advanced photoinitiators relies on a robust three-step sequence that ensures high regioselectivity and yield. The first stage involves a nucleophilic aromatic substitution (SNAr) reaction where the thiol group of the aminothiophenol attacks the electron-deficient carbon attached to the halogen on the benzophenone ring. This reaction is typically conducted in polar aprotic solvents like N-methyl-2-pyrrolidone (NMP) or dimethylformamide (DMF) at temperatures ranging from 100°C to 185°C. The presence of a base, such as potassium hydroxide or sodium carbonate, facilitates the deprotonation of the thiol, generating a highly nucleophilic thiolate anion that drives the displacement of the halide leaving group.
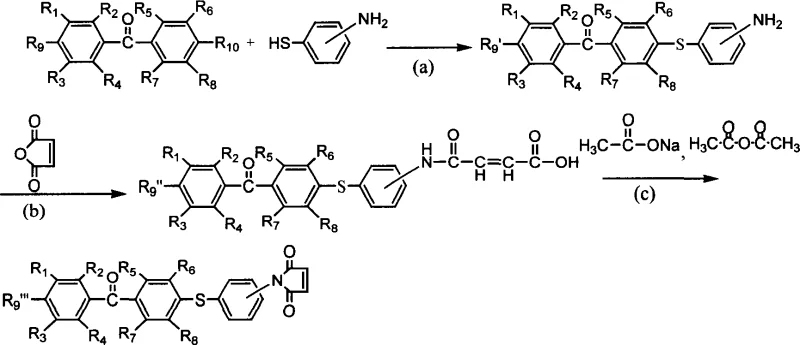
Following the formation of the amino-benzophenone intermediate, the second step involves the ring-opening acylation with maleic anhydride. This exothermic reaction is carefully controlled at lower temperatures, typically between 0°C and 30°C, to prevent premature polymerization or side reactions. The resulting maleamic acid intermediate is then subjected to a dehydration cyclization in the final step. Using acetic anhydride and anhydrous sodium acetate under nitrogen protection, the maleamic acid undergoes intramolecular condensation to form the thermally stable maleimide ring. This precise control over reaction conditions ensures the formation of the desired commercial scale-up of complex polymer additives with minimal impurities, crucial for maintaining the optical clarity and mechanical integrity of the final UV-cured product.
How to Synthesize Sulfur-Containing Benzophenone Photoinitiators Efficiently
The preparation of these high-performance initiators follows a logical progression from readily available commodity chemicals to specialized functional additives. The process begins with the coupling of the aromatic cores, followed by functionalization with the polymerizable group, and concludes with ring closure. Each step has been optimized in the patent examples to maximize yield and purity, utilizing standard workup procedures such as acid precipitation and recrystallization.
- Perform nucleophilic aromatic substitution between halogenated benzophenone and aminothiophenol in polar aprotic solvents at elevated temperatures.
- React the resulting amino-benzophenone intermediate with maleic anhydride at low temperatures to form the maleamic acid derivative.
- Execute dehydration cyclization using acetic anhydride and sodium acetate to close the maleimide ring and finalize the photoinitiator structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this new class of photoinitiators offers compelling economic and logistical benefits. The synthesis route avoids the use of expensive and hazardous reagents like acryloyl chloride, replacing them with cost-effective and widely available alternatives such as maleic anhydride and substituted benzophenones. This shift not only lowers the raw material cost basis but also simplifies the safety infrastructure required for production, as the process operates under atmospheric pressure without the need for specialized corrosion-resistant equipment. The elimination of toxic intermediates translates directly into reduced waste treatment costs and a smaller environmental footprint, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The synthetic pathway utilizes common organic solvents and reagents that are sourced from mature global supply chains, ensuring price stability and availability. By eliminating the need for expensive transition metal catalysts or exotic ligands, the overall production cost is significantly optimized. Furthermore, the high reaction efficiency and straightforward purification steps reduce solvent consumption and energy usage per kilogram of product, contributing to substantial operational savings in large-scale manufacturing environments.
- Enhanced Supply Chain Reliability: The starting materials, including various halogenated benzophenones and aminothiophenols, are produced by multiple suppliers worldwide, mitigating the risk of single-source bottlenecks. The robustness of the chemical steps means that the process is less sensitive to minor fluctuations in reaction parameters, leading to consistent batch-to-batch quality. This reliability is critical for reducing lead time for high-purity photoinitiators, allowing manufacturers to maintain lean inventory levels while ensuring uninterrupted production schedules for their downstream customers in the coatings and electronics sectors.
- Scalability and Environmental Compliance: The reaction conditions described, such as reflux temperatures and ambient pressure filtration, are easily transferable from pilot plant to multi-ton reactors without requiring fundamental process redesign. The absence of heavy metals and the use of recyclable solvents facilitate compliance with stringent international environmental regulations. The solid-state nature of the intermediates and final products simplifies logistics and storage, reducing the risks associated with transporting liquid hazardous chemicals and enhancing overall supply chain safety and resilience.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these sulfur-containing photoinitiators in industrial applications. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clear guidance for formulation chemists and process engineers.
Q: How does the sulfur linkage affect the photoinitiator's performance compared to traditional benzophenone?
A: The introduction of the sulfur atom extends the conjugation system, causing a significant red shift in UV absorption. This allows the initiator to absorb light more efficiently at longer wavelengths, resulting in initiation speeds approximately three to four times faster than standard benzophenone in certain acrylate systems.
Q: What are the primary advantages of the polymerizable maleimide group in this structure?
A: The maleimide group contains an unsaturated double bond that allows the photoinitiator to copolymerize directly into the polymer matrix during curing. This covalent bonding eliminates the issues of volatility, migration, and odor associated with traditional small-molecule initiators, while simultaneously enhancing the heat resistance of the final cured product.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes standard organic synthesis operations such as reflux, precipitation, and recrystallization. The raw materials, including halogenated benzophenones and maleic anhydride, are commercially available commodity chemicals, facilitating straightforward scale-up from laboratory to multi-ton production without requiring exotic catalysts or extreme pressure conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfur-Containing Benzophenone Photoinitiator Supplier
As the demand for high-performance UV curing solutions continues to grow across the electronics, automotive, and packaging industries, securing a dependable source of advanced photoinitiators is more critical than ever. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of sulfur-linked benzophenone derivative performs consistently in your demanding applications.
We invite you to collaborate with our technical team to explore how these innovative initiators can enhance your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to help you accelerate your time-to-market with confidence and precision.
