Advanced Synthesis of Benzophenone Maleimide Photoinitiators for High-Performance UV Curing Applications
Advanced Synthesis of Benzophenone Maleimide Photoinitiators for High-Performance UV Curing Applications
The rapid evolution of ultraviolet (UV) curing technology since the mid-20th century has driven an insatiable demand for high-efficiency photoinitiators that can meet stringent industrial standards for performance and safety. Patent CN1727364A introduces a significant breakthrough in this domain by detailing the preparation of benzophenone photoinitiators containing maleimide groups. This innovation addresses critical limitations found in traditional small-molecule photoinitiator systems, specifically regarding volatility, migration, and toxicity during long-term storage and application. By chemically integrating a polymerizable maleimide moiety into the robust benzophenone backbone, this technology enables the creation of macromolecular photoinitiators that are inherently compatible with curing matrices. For R&D directors and technical procurement specialists in the coatings, inks, and electronic materials sectors, understanding the mechanistic advantages and scalable synthesis of these compounds is essential for next-generation product development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reliance on traditional small-molecule photoinitiators has presented persistent challenges in high-performance industrial applications, particularly concerning physical stability and environmental safety. In conventional UV curing systems, small molecule initiators often exhibit poor compatibility with the polymer matrix, leading to phenomena such as volatilization and migration over extended storage periods. This physical instability not only diminishes the photopolymerization initiation efficiency but also results in undesirable side effects like persistent odor and increased toxicity in the final cured product. Furthermore, earlier attempts to modify benzophenone structures to improve compatibility often relied on the introduction of acryloyl chloride or methacryloyl chloride groups. These synthetic routes are fraught with hazards due to the high toxicity and corrosivity of acyl chlorides, posing significant safety risks in manufacturing environments. Additionally, the resulting products from these older methods frequently failed to demonstrate substantial improvements in ultraviolet red-shift or initiation efficiency compared to unmodified benzophenone, limiting their commercial viability.
The Novel Approach
The methodology outlined in patent CN1727364A represents a paradigm shift by utilizing a safer and more effective molecular design strategy that incorporates maleimide groups directly into the benzophenone structure. This approach leverages the unique chemical properties of the maleimide ring, which contains a highly reactive unsaturated double bond capable of participating directly in the photopolymerization process. Unlike the volatile small molecules of the past, these new derivatives can act as monomers that copolymerize with hydrogen donors, such as tertiary amines containing unsaturated double bonds, to form polymeric photoinitiators with co-initiator systems. This structural integration effectively anchors the photoinitiator within the cured network, drastically reducing migration and toxicity issues. Moreover, the versatility of this chemical architecture allows it to function not only in standard free-radical systems but also as a copolymerization photoinitiator in cationic UV curing applications, thereby significantly broadening its utility across diverse industrial sectors including electronic packaging and advanced adhesives.
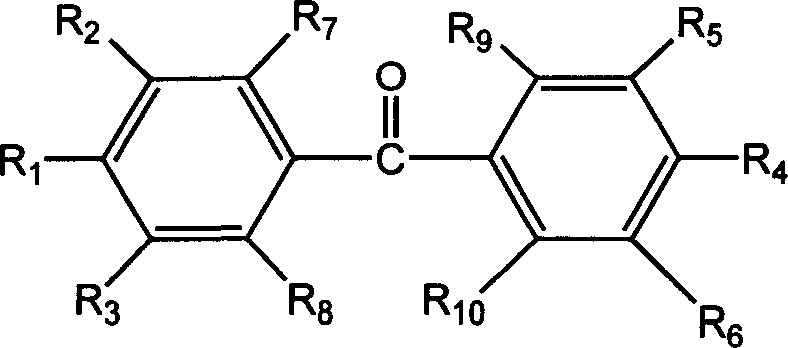
Mechanistic Insights into Maleimide Ring Formation and Photoinitiation
The core of this technological advancement lies in the precise two-step synthetic pathway that transforms amino-substituted benzophenones into the target maleimide derivatives through a controlled ring-closing mechanism. The process begins with a nucleophilic attack where the amino group of the benzophenone derivative reacts with maleic anhydride in an organic solvent medium at mild temperatures ranging from 0°C to 30°C. This initial step yields a maleamic acid intermediate, which serves as the crucial precursor for the subsequent cyclization. The reaction conditions are carefully optimized to ensure high conversion rates while minimizing side reactions, utilizing solvents such as acetone, ether, or N,N-dimethylformamide to maintain homogeneity and reaction kinetics. The formation of the maleamic acid bond is a fundamental transformation that sets the stage for the introduction of the polymerizable functionality without compromising the integrity of the benzophenone chromophore responsible for light absorption.
Following the formation of the intermediate, the synthesis proceeds to a dehydration and ring-closure step that is critical for generating the active maleimide moiety. This transformation is achieved by heating the maleamic acid intermediate in the presence of acetic anhydride and anhydrous sodium acetate, typically at temperatures between 60°C and 120°C, with specific embodiments favoring a range of 98°C to 100°C. The acetic anhydride acts as a dehydrating agent, facilitating the elimination of water to close the five-membered imide ring, while the sodium acetate serves as a mild base catalyst to drive the equilibrium forward. This cyclization creates the rigid, planar maleimide structure that is essential for the compound's thermal stability and polymerization capability. The resulting product exhibits enhanced photoinitiation efficiency, with photoelectric differential calorimetry data indicating that the maximum initiation speed in 1,6-hexanediol diacrylate systems is approximately double that of standard benzophenone, validating the superior performance of this molecular design.
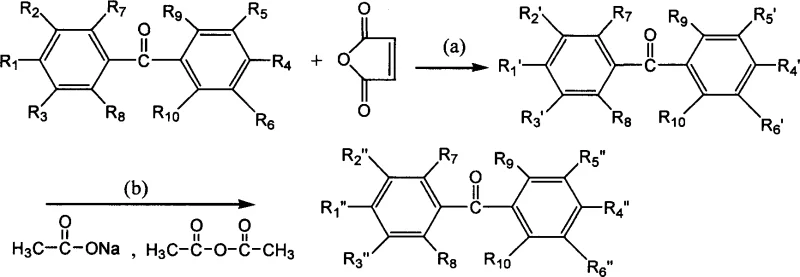
How to Synthesize Benzophenone Maleimide Photoinitiator Efficiently
The practical implementation of this synthesis route offers a robust framework for laboratory and pilot-scale production, relying on widely available reagents and straightforward unit operations. The process is designed to be adaptable to various substituted benzophenone starting materials, allowing for the customization of the final photoinitiator's properties to suit specific formulation requirements. The reaction sequence involves precise control of temperature and stoichiometry during the addition of maleic anhydride, followed by a thermal cyclization step that requires nitrogen protection to prevent oxidation. After the reaction is complete, the product is isolated through precipitation into ice-water mixtures, a technique that ensures high purity by removing soluble impurities and residual reagents.
- React amino-containing benzophenone with maleic anhydride in an organic solvent at 0-30°C to form the maleamic acid intermediate.
- Perform dehydration and ring-closure using acetic anhydride and anhydrous sodium acetate at elevated temperatures (60-120°C).
- Isolate the final product via precipitation in ice-water, filtration, and recrystallization from suitable solvents.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this maleimide-based synthesis route offers compelling advantages related to cost structure, safety compliance, and operational reliability. The elimination of hazardous acyl chloride reagents from the manufacturing process removes the need for specialized corrosion-resistant equipment and complex scrubbing systems required to handle toxic off-gases, leading to significant capital expenditure savings and reduced operational overhead. Furthermore, the use of common organic solvents and reagents like maleic anhydride and acetic anhydride ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or regulated chemicals. This stability translates into more predictable lead times and consistent production schedules, which are critical for maintaining continuity in high-volume industrial applications such as coating manufacturing and electronic material production.
- Cost Reduction in Manufacturing: The streamlined two-step synthesis avoids the expensive and dangerous handling protocols associated with acyl chlorides, directly lowering the cost of goods sold through reduced safety infrastructure and waste treatment expenses. By utilizing a dehydration cyclization method with acetic anhydride, the process achieves high atom economy and minimizes the generation of hazardous byproducts, further contributing to overall cost efficiency. The ability to produce high-purity intermediates through simple precipitation and recrystallization techniques reduces the need for complex chromatographic purification, making the process economically viable for large-scale commercial production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as amino-benzophenones and maleic anhydride ensures that raw material availability is not subject to the volatility often seen with niche specialty reagents. This foundational stability allows manufacturers to secure long-term supply contracts and maintain buffer stocks without incurring prohibitive costs, thereby safeguarding against market fluctuations. Additionally, the robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, provides operational flexibility that enhances the resilience of the manufacturing supply chain against unexpected disruptions.
- Scalability and Environmental Compliance: The synthesis protocol is inherently scalable, utilizing standard batch reactor operations that can be easily transitioned from kilogram to multi-ton scales without requiring fundamental changes to the process chemistry. The absence of heavy metal catalysts and toxic halogenated reagents simplifies the environmental compliance landscape, reducing the regulatory burden and facilitating easier permitting for new production facilities. This alignment with green chemistry principles not only future-proofs the manufacturing operation against tightening environmental regulations but also enhances the marketability of the final product to eco-conscious downstream customers in the coatings and electronics industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented photoinitiator technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and process safety. Understanding these details is crucial for technical teams evaluating the feasibility of integrating these materials into existing formulations or developing new high-performance UV curing systems.
Q: What are the advantages of maleimide-based photoinitiators over traditional benzophenone?
A: Maleimide-based photoinitiators contain polymerizable unsaturated double bonds that allow them to copolymerize into the cured matrix. This significantly reduces migration and volatilization compared to traditional small molecule benzophenone, thereby minimizing odor and toxicity while improving long-term stability.
Q: Does this synthesis method avoid toxic reagents?
A: Yes, unlike previous methods that utilized toxic acryloyl chloride or methacryloyl chloride, this patented process uses maleic anhydride and acetic anhydride. This eliminates the handling risks associated with corrosive acyl chlorides and simplifies waste treatment protocols.
Q: Can this photoinitiator be used in cationic UV curing systems?
A: Yes, the introduction of the maleimide group broadens the application scope. These derivatives can function as copolymerization photoinitiators in cationic ultraviolet curing systems, offering versatility beyond standard free-radical curing applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzophenone Maleimide Photoinitiator Supplier
As the global demand for high-performance UV curing materials continues to surge, NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses deep expertise in optimizing complex organic syntheses, ensuring that we can deliver benzophenone maleimide photoinitiators with stringent purity specifications tailored to your specific application needs. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify every batch, guaranteeing consistency and reliability that meets the exacting standards of the international fine chemical and electronic materials markets.
We invite you to engage with our technical procurement team to discuss how this innovative technology can enhance your product portfolio and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the economic benefits of switching to our optimized synthesis route. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on comprehensive technical and commercial evidence.
