Scalable Manufacturing of High-Efficiency Bis-carbazole Photoinitiators for Advanced LCD and Coating Applications
Scalable Manufacturing of High-Efficiency Bis-carbazole Photoinitiators for Advanced LCD and Coating Applications
The rapid evolution of display technologies, particularly in the realm of RGB key components for next-generation large-screen LCDs, demands photoinitiators with exceptional sensitivity and deep-curing capabilities. Patent CN108484480B introduces a sophisticated molecular architecture known as bis-carbazole-containing benzophenone oxime acetate, which represents a significant leap forward in photo-initiation efficiency. This novel compound leverages the rigid conjugated plane of the carbazole moiety to facilitate strong intramolecular electron transfer, a critical feature for high-performance two-photon photoinitiators. By strategically introducing alkyl chains on the nitrogen atoms and connecting conjugated aromatic acyl and oxime ester electron-withdrawing groups, the synthesis creates a robust 'push-pull' electronic system. 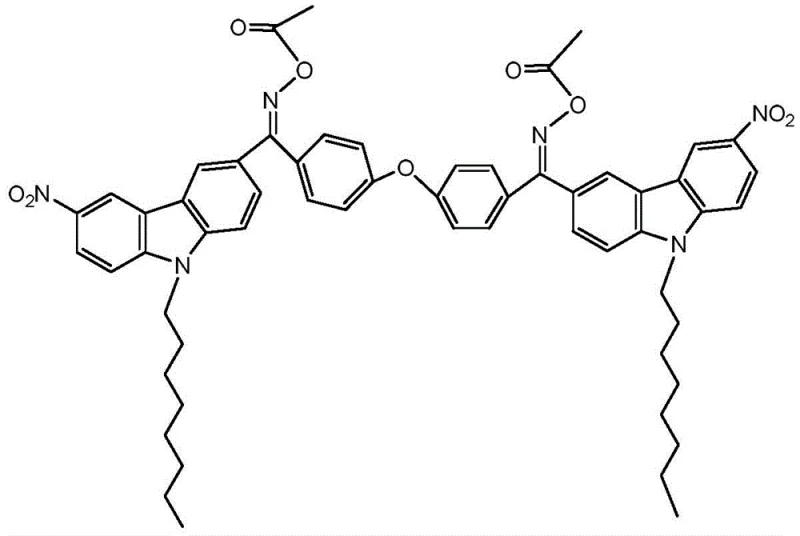 This structural optimization not only enhances the electron transfer degree within the molecule but also drastically improves two-photon fluorescence properties, making it an ideal candidate for demanding applications such as etching resists, hologram recording materials, and pressure-sensitive adhesives. As a reliable photoinitiator supplier, understanding the nuances of this synthesis is key to delivering high-purity polymer additives that meet the rigorous standards of the electronic materials sector.
This structural optimization not only enhances the electron transfer degree within the molecule but also drastically improves two-photon fluorescence properties, making it an ideal candidate for demanding applications such as etching resists, hologram recording materials, and pressure-sensitive adhesives. As a reliable photoinitiator supplier, understanding the nuances of this synthesis is key to delivering high-purity polymer additives that meet the rigorous standards of the electronic materials sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoinitiators often struggle with a trifecta of performance issues: low sensitivity, poor solubility in resin matrices, and significant susceptibility to oxygen inhibition during the photocuring process. Many conventional oxime esters lack the extended conjugation necessary for deep curing, leading to incomplete polymerization in thick films or pigmented systems which is detrimental to the manufacturing of high-quality LCD panels. Furthermore, storage stability is frequently compromised in older generation initiators, leading to premature decomposition and reduced shelf life which complicates inventory management for procurement teams. The reliance on simpler aromatic structures often limits the absorption range, necessitating higher loading levels to achieve desired cure speeds, which can negatively impact the mechanical properties of the final cured coating. These limitations create a bottleneck in cost reduction in electronic chemical manufacturing, as manufacturers are forced to use excessive amounts of initiator or accept suboptimal curing profiles.
The Novel Approach
The innovative synthesis pathway detailed in the patent overcomes these historical barriers by constructing a complex, symmetric molecule that integrates multiple photosensitive functional groups into a single entity. By linking two nitro-substituted carbazole units via a 4,4'-oxydibenzoyl bridge, the new method effectively extends the conjugated chain length, thereby widening the spectrum absorption range and increasing the molar extinction coefficient. This architectural design ensures that the photoinitiator exhibits high photoinitiation speed and excellent deep curing performance, even in challenging acrylic epoxy resin systems. The inclusion of the oxime acetate group provides the necessary lability for efficient radical generation upon UV exposure, while the bulky carbazole groups enhance solubility and reduce crystallization tendencies during storage. This holistic approach to molecular design results in a product that not only outperforms commercial benchmarks like TPO in curing rate tests but also offers superior storage stability, addressing the critical pain points of both R&D and supply chain stakeholders.
Mechanistic Insights into AlCl3-Catalyzed Friedel-Crafts Acylation
The cornerstone of this synthesis is the Friedel-Crafts acylation step, where the nitro-substituted carbazole intermediate reacts with 4,4'-oxydibenzoyl chloride under the catalytic influence of aluminum trichloride (AlCl3). This reaction is pivotal as it forms the central ketone linkage that bridges the two carbazole halves, establishing the extended conjugation system essential for the molecule's photoelectric properties. The mechanism involves the coordination of the Lewis acid AlCl3 with the carbonyl oxygen of the acid chloride, generating a highly electrophilic acylium ion complex. This electrophile then attacks the electron-rich aromatic ring of the carbazole derivative, specifically at the position para to the nitrogen atom, driven by the directing effects of the substituents. 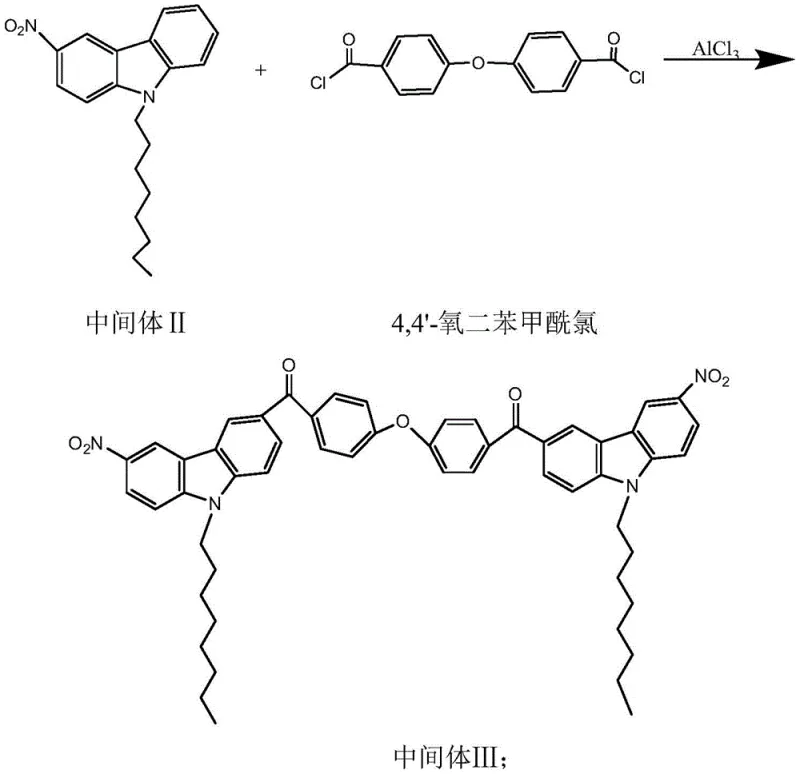 Precise temperature control between 0-30°C is critical during this phase to manage the exothermic nature of the reaction and prevent poly-acylation or degradation of the sensitive nitro groups. The use of dichloroethane as a solvent ensures adequate solubility of the polar intermediates while maintaining a reaction medium that supports the stability of the AlCl3 catalyst complex throughout the 8-10 hour reaction window.
Precise temperature control between 0-30°C is critical during this phase to manage the exothermic nature of the reaction and prevent poly-acylation or degradation of the sensitive nitro groups. The use of dichloroethane as a solvent ensures adequate solubility of the polar intermediates while maintaining a reaction medium that supports the stability of the AlCl3 catalyst complex throughout the 8-10 hour reaction window.
Impurity control in this stage is paramount for ensuring the final product meets high-purity photoinitiator specifications, as residual aluminum salts or unreacted acid chlorides can act as quenchers in the final application. The patent specifies a rigorous workup procedure involving quenching with 15% hydrochloric acid followed by multiple washes with water and sodium bicarbonate solution. This multi-stage washing protocol is designed to effectively hydrolyze and remove the aluminum complexes and neutralize any acidic byproducts, preventing contamination of the organic phase. Subsequent recrystallization further purifies Intermediate III, removing regio-isomers or oligomeric byproducts that could otherwise compromise the clarity and color stability of the final coating. This attention to detail in the purification mechanism underscores the feasibility of the process for commercial scale-up of complex organic intermediates, ensuring consistent quality batch after batch.
How to Synthesize Bis-carbazole Benzophenone Oxime Acetate Efficiently
The complete synthetic route described in the patent offers a robust framework for producing this advanced photoinitiator, moving from simple alkylation to the final acetylation in five distinct, manageable steps. The process begins with the N-alkylation of carbazole using 1-bromooctane, followed by nitration to introduce the electron-withdrawing nitro group, setting the stage for the subsequent coupling reactions. Each step has been optimized for yield and purity, utilizing common industrial solvents like DMSO and dichloroethane which facilitates easy solvent recovery and recycling. For R&D teams looking to replicate or adapt this chemistry, the detailed parameters regarding temperature, stoichiometry, and reaction times provided in the patent serve as a valuable baseline for process development.
- Alkylation of carbazole with 1-bromooctane in DMSO/KOH system to form Intermediate I.
- Nitration of Intermediate I using nitric acid at low temperatures to yield Intermediate II.
- Friedel-Crafts acylation of Intermediate II with 4,4'-oxydibenzoyl chloride using AlCl3 catalyst to form the ketone backbone.
- Conversion of the ketone to oxime using hydroxylamine hydrochloride, followed by acetylation with acetic anhydride to finalize the photoinitiator.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route presents compelling advantages that directly address the core concerns of procurement managers and supply chain heads regarding cost, reliability, and scalability. The process is designed with industrial practicality in mind, utilizing raw materials such as carbazole, 1-bromooctane, and 4,4'-oxydibenzoyl chloride which are readily available in the global chemical market, thereby mitigating supply risk. The operational simplicity of the reactions, which do not require extreme cryogenic conditions or high-pressure equipment, allows for implementation in standard glass-lined or stainless steel reactors found in most fine chemical facilities. This accessibility translates to lower capital expenditure requirements for new production lines and reduces the barrier to entry for scaling up production volumes to meet growing market demand.
- Cost Reduction in Manufacturing: A significant driver for cost optimization in this process is the strategic selection of solvents and the implementation of recovery protocols. Solvents such as dimethyl sulfoxide (DMSO) and dichloroethane are used in substantial quantities but are explicitly noted in the patent as being recoverable via reduced pressure distillation. This capability to recycle solvents drastically reduces the variable cost per kilogram of the final product, as solvent purchase and waste disposal often constitute a major portion of manufacturing expenses in fine chemical synthesis. Furthermore, the elimination of expensive transition metal catalysts in favor of abundant Lewis acids like aluminum trichloride removes the need for costly heavy metal scavenging steps, streamlining the downstream processing and further lowering the overall production cost.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the starting materials ensures a stable and resilient supply chain that is less susceptible to the volatility often seen with specialized reagents. Carbazole and bromoalkanes are produced at massive scales for various industries, guaranteeing a continuous flow of raw materials even during periods of market tightness. The robustness of the synthetic steps, characterized by wide operating temperature windows and tolerance to minor variations in addition rates, enhances the reliability of production scheduling. This predictability allows supply chain planners to maintain leaner inventories with confidence, knowing that production runs can be executed consistently without frequent delays caused by sensitive reaction failures or hard-to-source reagents.
- Scalability and Environmental Compliance: The process is inherently scalable due to its batch-wise nature and the use of standard unit operations such as stirring, heating, cooling, and crystallization. The workup procedures involve liquid-liquid extraction and washing, which are easily adapted from laboratory to pilot and finally to full commercial scale without fundamental changes to the chemistry. From an environmental standpoint, the ability to recover and reuse solvents aligns with modern green chemistry principles, reducing the volume of hazardous waste generated. Additionally, the high yield and purity achieved reduce the burden on waste treatment facilities, as there is less off-spec material to dispose of, facilitating easier compliance with increasingly stringent environmental regulations governing chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this bis-carbazole photoinitiator, derived directly from the patent specifications and performance data. Understanding these details helps stakeholders evaluate the fit of this material for their specific formulation needs and manufacturing capabilities.
Q: What are the key advantages of this bis-carbazole photoinitiator over traditional TPO?
A: The bis-carbazole structure provides a rigid conjugated plane that enhances intramolecular electron transfer, resulting in superior two-photon fluorescence properties and deeper curing capabilities compared to standard initiators like TPO.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the process utilizes common organic solvents like dichloroethane and DMSO which are easily recoverable, and the reaction conditions (0-30°C for acylation) are mild enough for safe scale-up without requiring exotic cryogenic equipment.
Q: How does the nitro group affect the performance of the photoinitiator?
A: The introduction of the nitro group acts as a strong electron-withdrawing group, which works in tandem with the electron-donating alkyl chains to create a 'push-pull' electronic system that significantly broadens the absorption spectrum and increases initiation efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-carbazole-containing Benzophenone Oxime Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance photoinitiators play in the advancement of electronic materials and specialty coatings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our expertise in handling complex organic syntheses, particularly those involving sensitive functional groups like oxime esters and nitro compounds, positions us as an ideal partner for your supply chain.
We invite you to collaborate with us to explore how this advanced photoinitiator can enhance your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your goals for cost reduction in electronic chemical manufacturing and supply chain efficiency.
