Advanced Biocatalytic Synthesis of Chiral Biaryl Alcohols for Pharmaceutical Scale-Up
Advanced Biocatalytic Synthesis of Chiral Biaryl Alcohols for Pharmaceutical Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methods for producing chiral intermediates, particularly for antihistamine medications like Betahistine. Patent CN111433357B introduces a groundbreaking approach utilizing engineered alcohol dehydrogenase mutants to synthesize biaryl chiral alcohols with exceptional stereoselectivity. This technology addresses critical bottlenecks in traditional chemical synthesis by offering a biocatalytic route that operates under mild conditions while achieving optical purity levels that meet stringent regulatory standards. For R&D Directors and Supply Chain Heads, this patent represents a significant shift towards sustainable and efficient manufacturing of key pharmaceutical intermediates, specifically targeting the production of (4-chlorophenyl)-(pyridin-2-yl)-methanol (CPMA). The ability to control stereochemistry at the enzymatic level ensures a consistent supply of high-quality materials essential for downstream drug formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis of chiral biaryl alcohols often relies on transition metal catalysts such as Ruthenium or Rhodium complexes, which present significant commercial and technical challenges. These methods typically require high-pressure hydrogenation conditions, posing safety risks and necessitating expensive specialized equipment that increases capital expenditure. Furthermore, chemical catalysts often struggle with substrate specificity, leading to lower enantiomeric excess (ee) values that require costly and yield-reducing recrystallization steps to purify. The reliance on noble metals also introduces the risk of heavy metal contamination in the final API, requiring additional purification stages that extend lead times and complicate regulatory compliance. Additionally, conventional methods often operate at low substrate concentrations to maintain selectivity, which drastically reduces volumetric productivity and increases solvent waste, making them less attractive for large-scale commercial operations.
The Novel Approach
In contrast, the biocatalytic method described in the patent utilizes specific alcohol dehydrogenase mutants that function efficiently at ambient pressure and moderate temperatures, significantly simplifying the operational requirements. This enzymatic approach eliminates the need for toxic heavy metals, thereby reducing environmental impact and simplifying waste treatment protocols for the supply chain team. The mutants exhibit remarkable tolerance to high substrate concentrations, allowing for more concentrated reaction mixtures that improve space-time yield and reduce solvent consumption. By coupling the reduction with a coenzyme regeneration system, the process ensures economic viability by minimizing the cost of expensive cofactors. This novel route not only enhances the safety profile of the manufacturing process but also aligns with green chemistry principles, offering a sustainable alternative for the production of reliable pharmaceutical intermediates supplier networks seek.
Mechanistic Insights into ADH-Catalyzed Asymmetric Reduction
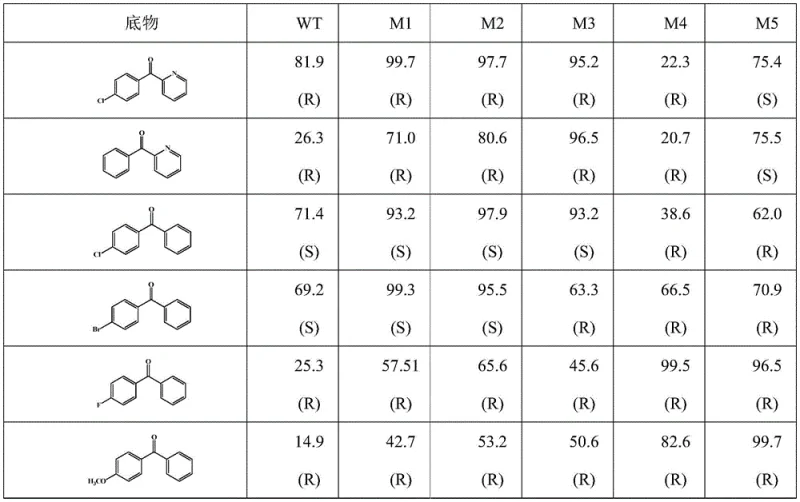
The core innovation lies in the precise molecular engineering of the alcohol dehydrogenase enzyme, where specific amino acid residues at positions 161 and 196 are mutated to alter the steric environment of the active site. For instance, mutant M1 replaces Serine at position 196 with Valine, which creates a tighter binding pocket that favors the formation of the (R)-enantiomer with an ee value of 98.7%, a substantial improvement over the wild-type enzyme. This structural modification enhances the enzyme's ability to discriminate between pro-chiral faces of the ketone substrate, ensuring that the hydride transfer from the cofactor occurs with high fidelity. The mechanism involves a concerted transfer of a hydride ion from NADPH to the carbonyl carbon, facilitated by the precise orientation of the substrate within the mutated active site. This level of control is critical for R&D Directors who need to guarantee the optical purity of the intermediate to prevent the formation of inactive or toxic isomers in the final drug product.
Furthermore, the patent demonstrates the versatility of these mutants across a range of biaryl ketone substrates, indicating a broad applicability for cost reduction in pharmaceutical intermediates manufacturing. Mutant M5, for example, exhibits a reversed stereoselectivity compared to the wild type, producing the (S)-enantiomer for certain substrates with high efficiency. This flexibility allows manufacturers to access different chiral configurations without developing entirely new catalytic systems, streamlining the development pipeline for diverse drug candidates. The enzyme's stability under reaction conditions ensures consistent performance over extended periods, reducing the frequency of catalyst replenishment and minimizing process variability. Understanding these mechanistic details is essential for scaling up the process, as it informs the optimization of parameters such as pH, temperature, and cofactor concentration to maximize yield and purity.
How to Synthesize Chiral CPMA Efficiently
The synthesis of high-purity chiral CPMA using this biocatalytic method involves a streamlined workflow that integrates enzyme expression, reaction setup, and product isolation into a cohesive process. The initial step requires the cultivation of recombinant E. coli strains expressing the specific ADH mutant, followed by induction to produce the active enzyme in sufficient quantities for the reduction reaction. Once the biocatalyst is prepared, it is introduced into a buffered aqueous system containing the pro-chiral ketone substrate and a coenzyme regeneration system to sustain the catalytic cycle. The detailed standardized synthesis steps see the guide below.
- Construct recombinant E. coli expressing the specific alcohol dehydrogenase mutant (e.g., M1, M4, or M5) via plasmid transformation and induction.
- Prepare the biocatalytic reaction system containing the pro-chiral ketone substrate, cofactor NADP+, and a coenzyme regeneration system such as glucose dehydrogenase.
- Incubate the mixture under mild physiological conditions (30-35°C, pH 6-8) to achieve high conversion and optical purity, followed by product extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of expensive noble metal catalysts removes a significant variable cost component, leading to substantial cost savings over the lifecycle of the product. Moreover, the mild reaction conditions reduce energy consumption associated with heating, cooling, and pressurization, further enhancing the economic efficiency of the manufacturing process. The high substrate tolerance allows for smaller reactor volumes to produce the same amount of product, optimizing facility utilization and reducing capital investment requirements for new production lines. These factors combine to create a more competitive cost structure that can be leveraged in negotiations with downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts eliminates the need for complex and costly metal scavenging steps, which are often required to meet strict residual metal limits in pharmaceuticals. This simplification of the downstream processing workflow reduces the consumption of specialized resins and solvents, directly lowering the cost of goods sold. Additionally, the high conversion rates achieved by the mutants minimize the amount of unreacted starting material that needs to be recovered or disposed of, improving overall material efficiency. The economic benefits are compounded by the ability to operate at higher concentrations, which reduces the volume of wastewater generated and lowers waste treatment costs significantly.
- Enhanced Supply Chain Reliability: Biocatalytic processes are less susceptible to the supply chain volatility often associated with precious metals, which can be subject to geopolitical tensions and market fluctuations. The recombinant enzymes can be produced consistently through fermentation, ensuring a stable and predictable supply of the catalyst for continuous manufacturing operations. The robustness of the mutants under various conditions also reduces the risk of batch failures due to minor process deviations, enhancing the reliability of delivery schedules. This stability is crucial for maintaining the continuity of supply for critical drug intermediates, preventing costly production stoppages for downstream partners.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic reaction aligns well with environmental regulations, reducing the release of volatile organic compounds (VOCs) and hazardous waste into the environment. Scaling up this process is straightforward as it does not require specialized high-pressure equipment, allowing for easier technology transfer between manufacturing sites. The reduced environmental footprint supports corporate sustainability goals and simplifies the permitting process for new facilities. This compliance advantage mitigates regulatory risks and enhances the company's reputation as a responsible manufacturer in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ADH mutant technology in industrial settings. These answers are derived directly from the patent data to provide accurate and actionable insights for decision-makers evaluating this synthesis route. Understanding these details helps in assessing the feasibility of integrating this biocatalytic method into existing production frameworks.
Q: How does the mutant enzyme improve stereoselectivity compared to wild-type ADH?
A: The engineered mutants, such as M1 and M3, feature specific amino acid substitutions at positions 161 and 196 that reshape the active site pocket. This structural modification significantly enhances steric constraints, forcing the substrate into a specific orientation that favors the formation of a single enantiomer with ee values exceeding 98%, compared to the wild-type's lower selectivity.
Q: What are the substrate concentration limits for this enzymatic process?
A: Unlike traditional chemical methods that often struggle at low concentrations, this biocatalytic system demonstrates robust activity at substrate concentrations ranging from 10mM up to 500mM. The patent data indicates that even at 200mM, the mutant M1 achieves near-complete conversion, highlighting its suitability for high-density manufacturing processes.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process eliminates the need for expensive noble metal catalysts and high-pressure hydrogenation equipment. The use of recombinant E. coli allows for scalable fermentation, and the mild reaction conditions reduce energy consumption and safety risks, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable CPMA Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced biocatalytic technologies to deliver high-value pharmaceutical intermediates to the global market. As a CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale processes like the one described in CN111433357B are successfully translated into robust manufacturing operations. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of CPMA meets the highest quality standards required by international regulatory bodies. We understand the critical nature of chiral purity in drug synthesis and are committed to providing materials that support the safety and efficacy of your final pharmaceutical products.
We invite you to collaborate with us to optimize your supply chain and leverage the cost benefits of this enzymatic synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments that demonstrate how we can support your project timelines and budget goals. By partnering with us, you gain access to a reliable supply of high-purity chiral alcohols that can accelerate your drug development and commercialization efforts.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
