Advanced Synthesis of Fluorinated Nitrobenzimidazoles for Scalable Pharmaceutical Manufacturing
Introduction to Advanced Fluorinated Heterocycle Synthesis
The pharmaceutical and agrochemical industries are increasingly reliant on fluorinated heterocyclic scaffolds due to their enhanced metabolic stability and bioavailability. Patent CN100415721C introduces a groundbreaking preparation method for fluorine-containing nitrobenzimidazoles, specifically targeting 2-trifluoromethyl-5(6)-nitrobenzimidazole and its difluoro analogues. This technology represents a significant leap forward in organic fluorine chemistry, addressing the critical need for efficient, high-yielding synthetic routes for complex API intermediates. By leveraging a mild, one-pot cyclization strategy, this method overcomes the severe limitations of traditional acid-catalyzed processes, offering a robust solution for manufacturers seeking to optimize their supply chains for high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted benzimidazoles relied heavily on harsh acidic conditions, typically utilizing hydrogen chloride as a catalyst under prolonged heating. As detailed in the background art of the patent, these conventional methods suffer from critically low yields and excessive reaction times, often extending up to 24 hours. Furthermore, the post-processing requirements are cumbersome, necessitating recrystallization in dilute hydrochloric acid to isolate the final solid product. This not only increases operational complexity and safety risks associated with handling strong acids but also generates significant environmental pollution, making such processes unsustainable for modern green chemistry standards and large-scale commercial manufacturing.
The Novel Approach
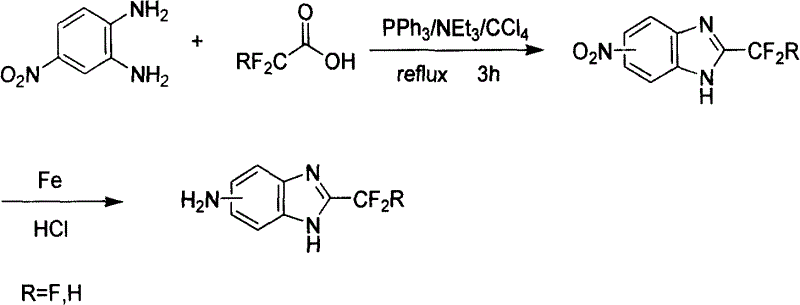
In stark contrast, the inventive method described in CN100415721C employs a sophisticated reagent system comprising triphenylphosphine, triethylamine, and carbon tetrachloride to facilitate a rapid, one-step ring-forming reaction. This approach allows for the simultaneous introduction of the trifluoromethyl or difluoromethyl group while cyclizing the 4-nitro-o-phenylenediamine precursor. The reaction is remarkably efficient, completing in merely 3 to 4 hours under reflux conditions, which drastically improves throughput. The workup is equally streamlined, involving simple solvent removal and filtration, thereby eliminating the need for difficult acidic recrystallization and ensuring a much cleaner crude product profile suitable for direct purification.
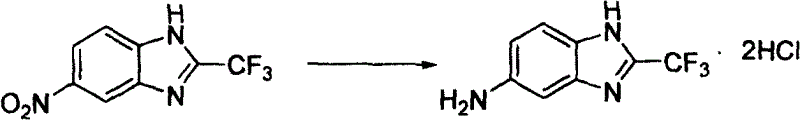
Mechanistic Insights into PPh3-Mediated Cyclization
The core of this technological breakthrough lies in the activation of the fluoroacetic acid by the triphenylphosphine and carbon tetrachloride system, effectively generating a reactive acyl halide intermediate in situ. This activated species then undergoes nucleophilic attack by the diamine, followed by dehydration and cyclization to form the benzimidazole ring. The presence of triethylamine acts as a proton scavenger, driving the equilibrium forward and neutralizing the generated acid byproducts. This mechanistic pathway avoids the high-energy barriers associated with direct thermal cyclization, allowing the reaction to proceed smoothly at moderate temperatures (around 60°C oil bath temperature) while maintaining excellent control over the reaction exotherm.
Beyond the primary cyclization, the utility of the resulting nitrobenzimidazole is underscored by its versatility as a precursor for further derivatization. The nitro group at the 5(6)-position is strategically placed for reduction to the corresponding amine, a crucial transformation for accessing a wide array of biologically active compounds. As illustrated in the downstream processing capabilities, the nitro intermediate can be efficiently reduced using iron and hydrochloric acid to yield the amino-benzimidazole derivative. This flexibility confirms the compound's status as a high-value building block, enabling the synthesis of diverse pharmacophores used in anticancer, antifungal, and antiviral drug development pipelines.
How to Synthesize 2-Trifluoromethyl-5-nitrobenzimidazole Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for producing high-purity fluorinated benzimidazoles. The process begins with the careful preparation of the reaction mixture under an inert nitrogen atmosphere to prevent oxidation of the phosphine reagent. Operators must strictly adhere to the temperature controls, initiating the reaction at 0°C to manage the initial exotherm upon acid addition, before gradually heating to reflux. This precise control ensures safety and maximizes the formation of the desired regioisomer. For a comprehensive, step-by-step guide including exact molar ratios and purification parameters, please refer to the standardized synthesis instructions provided below.
- Under nitrogen protection, mix triphenylphosphine and triethylamine in carbon tetrachloride at 0°C.
- Add trifluoroacetic acid, stir until white smoke disappears, then add 4-nitro-o-phenylenediamine.
- Heat to reflux for 3-4 hours, remove solvent, and purify via column chromatography to obtain the solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere technical feasibility. The elimination of prolonged reaction times and harsh acidic workups translates directly into improved asset utilization and reduced energy consumption per batch. By simplifying the isolation procedure to a straightforward filtration and solvent swap, the process minimizes the risk of product loss during purification, thereby enhancing the overall mass balance. These operational efficiencies contribute to a more resilient supply chain, capable of responding rapidly to fluctuating market demands for critical pharmaceutical intermediates without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The replacement of expensive, time-consuming recrystallization steps with a simple filtration and column chromatography workflow significantly lowers processing costs. Furthermore, the high conversion efficiency reduces the consumption of raw materials per kilogram of finished product, optimizing the cost of goods sold (COGS). The avoidance of heavy metal catalysts or specialized high-pressure equipment further decreases capital expenditure requirements, making this route economically attractive for both pilot and commercial-scale operations.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as triphenylphosphine and triethylamine, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages. The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is critical for maintaining long-term supply contracts with multinational pharmaceutical clients. This reliability reduces the need for excessive safety stock, allowing for leaner inventory management and improved cash flow for the manufacturing entity.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent's explicit suitability for large-scale production. The reduction in reaction time from 24 hours to roughly 3 hours allows for increased production capacity within existing facility footprints. Additionally, the simplified workup generates less aqueous acidic waste compared to traditional methods, aligning with increasingly stringent environmental regulations and sustainability goals, thus reducing the burden and cost associated with waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of fluorinated nitrobenzimidazoles. These insights are derived directly from the experimental data and technical specifications detailed in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these nuances is essential for evaluating the feasibility of integrating this intermediate into your specific drug discovery or process development programs.
Q: What are the key advantages of this new synthesis method over traditional HCl catalysis?
A: The new method utilizing PPh3/NEt3/CCl4 significantly reduces reaction time from 24 hours to approximately 3-4 hours and eliminates the need for harsh acidic recrystallization, resulting in higher purity and easier operation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for large-scale production due to its simple operation, high yield (up to 86% in examples), and straightforward workup procedure involving solvent evaporation and filtration.
Q: Can the nitro group be further functionalized?
A: Absolutely. The resulting nitrobenzimidazole serves as a versatile intermediate that can be readily reduced to the corresponding amino-benzimidazole using standard reducing agents like iron and hydrochloric acid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl-5-nitrobenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 2-trifluoromethyl-5-nitrobenzimidazole meets the highest international standards, providing you with a secure foundation for your synthesis campaigns.
We invite you to collaborate with us to leverage this advanced technology for your projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall project timelines.
