Advanced Synthesis of Deuterium-Labeled Danofloxacin for High-Precision Veterinary Drug Analysis
Advanced Synthesis of Deuterium-Labeled Danofloxacin for High-Precision Veterinary Drug Analysis
The pharmaceutical and agrochemical industries are increasingly demanding higher precision in residue analysis and pharmacokinetic studies, driving the need for robust stable isotope-labeled standards. Patent CN112881542A introduces a groundbreaking synthetic method for stable isotope deuterium-labeled danofloxacin, specifically Danofloxacin-D3, which serves as a critical internal standard for Isotope Dilution Mass Spectrometry (IDMS). This technology addresses the long-standing reliance on imported isotope standards by providing a domestically viable, high-efficiency synthesis route. The method ensures that the deuterium label remains stable throughout the reaction process, preventing isotopic dilution that could compromise analytical accuracy. For R&D directors and procurement specialists, this patent represents a significant opportunity to secure a reliable veterinary drugs supplier capable of delivering high-purity reference materials essential for regulatory compliance and quality control in livestock medicine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of stable isotope-labeled veterinary drug standards has been fraught with challenges, primarily due to the complexity of introducing isotopic labels without affecting the molecular integrity of the active pharmaceutical ingredient. Conventional methods often suffer from low isotopic abundance, where the deuterium atoms may exchange with protons in the solvent or during workup, leading to impure standards that fail to meet the rigorous requirements of LC-MS quantification. Furthermore, existing synthetic routes frequently involve harsh reaction conditions or expensive transition metal catalysts that complicate the purification process and introduce heavy metal impurities. These factors contribute to high production costs and extended lead times, creating supply chain bottlenecks for laboratories and manufacturers requiring consistent batches of internal standards. The lack of domestic production capabilities has historically forced reliance on foreign suppliers, exacerbating cost reduction in veterinary pharmaceuticals manufacturing and limiting accessibility for local quality control labs.
The Novel Approach
The innovative synthesis method disclosed in the patent overcomes these historical barriers by utilizing a streamlined three-step process that prioritizes mild conditions and high selectivity. By employing deuterated iodomethane as the labeling reagent in the initial step, the process ensures that the deuterium is introduced at a specific, stable position on the diazabicyclo heptane ring, minimizing the risk of isotopic shedding. The subsequent deprotection and coupling steps are optimized to maintain the integrity of the label while achieving high chemical purity. This approach eliminates the need for complex chromatographic separations often required to remove isotopic impurities, thereby simplifying the workflow. For supply chain heads, this translates to a more robust production capability where commercial scale-up of complex veterinary drugs becomes feasible without the need for specialized, expensive equipment, ensuring a continuous supply of high-quality analytical standards.
Mechanistic Insights into Deuterium Labeling and Coupling
The core of this synthesis lies in the precise nucleophilic substitution and subsequent amide coupling reactions that construct the Danofloxacin-D3 molecule. In the first step, (1S, 4S)-2-BOC-2, 5-diazabicyclo[2.2.1]heptane acts as the nucleophile, reacting with deuterated iodomethane in the presence of sodium hydride within a tetrahydrofuran solvent system. The reaction is meticulously controlled at 0-5°C to prevent side reactions and ensure the exclusive formation of the N-deuteromethyl bond. This low-temperature control is critical for preserving the stereochemistry of the bicyclic system and preventing the loss of the expensive deuterated reagent. Following the introduction of the label, the BOC protecting group is removed under acidic conditions using hydrochloric acid in 1, 4-dioxane. This step regenerates the secondary amine necessary for the final coupling, and the mild temperature profile ensures that the newly formed C-D bonds remain intact without undergoing hydrogen-deuterium exchange.
The final stage involves the coupling of the deuterated amine intermediate with 1-cyclopropyl-6, 7-difluoro-1, 4-dihydro-4-oxoquinoline-3-carboxylic acid. This reaction utilizes 1, 8-diazabicyclo[5.4.0]undec-7-ene (DBU) as a base in pyridine, facilitating the nucleophilic attack of the amine on the carboxylic acid derivative at elevated temperatures of 95-105°C. The choice of DBU and pyridine is strategic, providing a basic environment that promotes amide bond formation while solubilizing the fluoroquinolone substrate. The result is a stable isotope deuterium-labeled danofloxacin molecule where the deuterium atoms are securely locked within the methyl group attached to the piperazine ring. 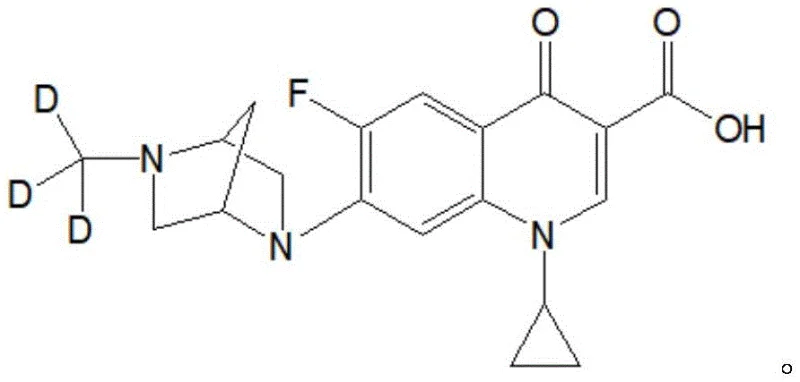 As illustrated in the structural analysis, the molecule retains the critical pharmacophore of danofloxacin while incorporating the mass shift required for mass spectrometric differentiation. The process achieves a chemical purity and stable isotope abundance of more than 99%, validating the efficacy of this mechanistic approach for producing high-fidelity analytical standards.
As illustrated in the structural analysis, the molecule retains the critical pharmacophore of danofloxacin while incorporating the mass shift required for mass spectrometric differentiation. The process achieves a chemical purity and stable isotope abundance of more than 99%, validating the efficacy of this mechanistic approach for producing high-fidelity analytical standards.
How to Synthesize Danofloxacin-D3 Efficiently
Implementing this synthesis route requires strict adherence to the patented parameters to ensure the high isotopic abundance required for IDMS applications. The process begins with the preparation of the deuterated intermediate, followed by deprotection and final coupling, with each step requiring precise temperature control and stoichiometric balance. The detailed standardized synthesis steps, including specific reagent grades, mixing rates, and purification protocols, are outlined in the technical guide below to assist R&D teams in replicating this high-yield process. This structured approach ensures that laboratories can transition from bench-scale experimentation to pilot production with confidence, maintaining the rigorous quality standards necessary for regulatory submission.
- React (1S, 4S)-2-BOC-2, 5-diazabicyclo[2.2.1]heptane with deuterated iodomethane and sodium hydride in THF at 0-5°C to introduce the deuterium-labeled methyl group.
- Remove the BOC protecting group by treating the intermediate with hydrochloric acid in 1, 4-dioxane, maintaining temperature control to prevent isotope loss.
- Couple the deprotected amine with 1-cyclopropyl-6, 7-difluoro-1, 4-dihydro-4-oxoquinoline-3-carboxylic acid using DBU in pyridine at 95-105°C to finalize the Danofloxacin-D3 structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers substantial benefits for procurement managers and supply chain directors looking to optimize their sourcing strategies for veterinary drug standards. The use of cheap and easily obtained raw materials, such as deuterated iodomethane and common quinoline derivatives, significantly reduces the direct material costs associated with production. Unlike methods requiring rare earth catalysts or exotic reagents, this route relies on commodity chemicals that are readily available in the global market, mitigating the risk of supply disruptions. Furthermore, the mild reaction conditions reduce energy consumption and equipment wear, contributing to overall operational efficiency. For organizations seeking cost reduction in veterinary pharmaceuticals manufacturing, adopting this method allows for a more competitive pricing structure without compromising on the purity or reliability of the final product.
- Cost Reduction in Manufacturing: The elimination of complex transition metal catalysts and the use of straightforward purification techniques drastically simplify the production workflow. By avoiding the need for expensive heavy metal removal steps and reducing solvent consumption through efficient reaction design, the overall manufacturing cost is significantly lowered. This economic efficiency makes the domestic production of isotope-labeled standards financially viable, reducing reliance on high-cost imports and allowing for better budget allocation in quality control departments.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining the integrity of analytical data over time. The availability of raw materials and the simplicity of the process mean that production can be scaled rapidly to meet fluctuating demand without long lead times. Reducing lead time for high-purity veterinary drugs is essential for laboratories that cannot afford delays in their testing schedules, and this method provides a dependable source of supply that enhances overall operational resilience.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory flasks to industrial reactors. The absence of toxic heavy metals and the use of standard organic solvents simplify waste treatment and environmental compliance, aligning with modern green chemistry principles. This facilitates the commercial scale-up of complex veterinary drugs, ensuring that manufacturers can meet increasing regulatory demands for residue monitoring while maintaining a sustainable production footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of deuterium-labeled danofloxacin. These insights are derived directly from the patent specifications and are intended to clarify the capabilities and limitations of the synthesis method for potential partners and technical stakeholders. Understanding these details is crucial for making informed decisions about integrating this technology into your quality control and supply chain frameworks.
Q: What is the isotopic abundance of the synthesized Danofloxacin-D3?
A: According to patent CN112881542A, the synthesis method achieves a stable isotope abundance of more than 99%, ensuring high precision for Isotope Dilution Mass Spectrometry (IDMS) applications.
Q: Why is deuterium labeling critical for Danofloxacin quantification?
A: Deuterium-labeled internal standards possess identical chemical properties to the target analyte but distinct mass numbers. This allows for the correction of recovery rates and matrix effects during LC-MS analysis, significantly improving quantification accuracy.
Q: Does this synthesis route involve harsh reaction conditions?
A: No, the patented process utilizes mild reaction conditions, with key steps performed at 0-5°C and final coupling at 95-105°C. This minimizes the risk of deuterium shedding and simplifies the purification process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Danofloxacin-D3 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity isotope standards play in ensuring the safety and efficacy of veterinary medicines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global pharmaceutical market. We are committed to delivering products with stringent purity specifications and maintaining rigorous QC labs to verify every batch against the highest international standards. Our capability to synthesize complex molecules like Danofloxacin-D3 demonstrates our commitment to innovation and quality in the fine chemical sector.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for isotope-labeled intermediates. By requesting a Customized Cost-Saving Analysis, you can discover how our manufacturing efficiencies can translate into tangible value for your organization. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments tailored to your specific analytical needs, ensuring a partnership built on transparency and technical excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
