Advanced Synthetic Route for 3-Methyl Sodium Salicylate Enabling Commercial Scale-Up and Purity Control
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for the production of critical organic intermediates. A recent technological breakthrough documented in patent CN114835574A introduces a novel preparation process for 3-methyl sodium salicylate (CAS 32768-20-0), a vital building block in the synthesis of various active pharmaceutical ingredients and specialty chemicals. This innovation addresses long-standing inefficiencies in traditional manufacturing routes by optimizing starting materials and refining purification protocols to achieve superior product quality. The disclosed methodology eliminates the need for extreme reaction conditions, thereby enhancing operational safety and reducing energy consumption significantly. For R&D directors and procurement managers alike, this represents a pivotal shift towards more sustainable and economically viable supply chains for high-purity pharmaceutical intermediates. By leveraging this advanced synthetic pathway, manufacturers can overcome previous bottlenecks related to yield and purity, ensuring a consistent supply of material that meets stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of methylsalicylic acid derivatives has relied heavily on carboxylation techniques that impose severe engineering constraints on production facilities. Prior art, such as the methods described by Kh.A. Suerbaev, utilizes carbon dioxide as a carbon source reacting with alkyl phenols under extremely harsh conditions. Specifically, these legacy processes necessitate high-pressure environments reaching up to 10atm and elevated temperatures exceeding 180°C to facilitate the formation of ethyl sodium carbonate intermediates. Such aggressive parameters not only demand specialized, expensive high-pressure reactor equipment but also pose significant safety risks during large-scale operations. Furthermore, the chemical efficiency of these conventional routes is notoriously poor, with reported yields for 3-methylsalicylic acid hovering around a mere 38%. This low conversion rate results in substantial waste generation and increased raw material costs, making the final product economically uncompetitive for mass-market applications. The difficulty in separating by-products under these conditions further complicates the purification process, often requiring multiple energy-intensive steps to achieve acceptable purity levels.
The Novel Approach
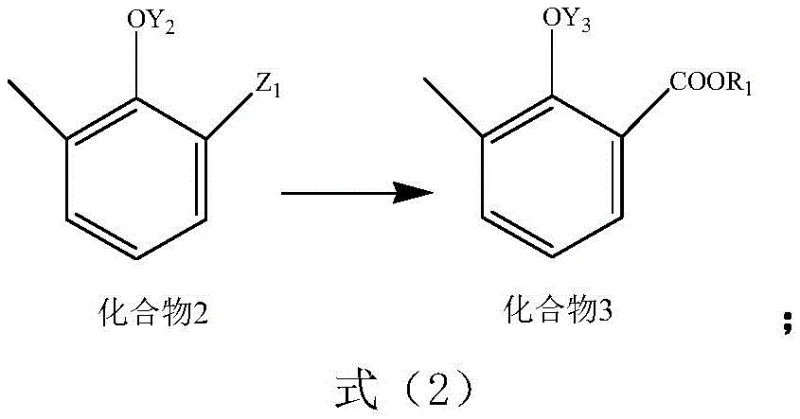
In stark contrast to the cumbersome legacy techniques, the process outlined in patent CN114835574A offers a streamlined, three-step synthetic route that operates under remarkably mild conditions. This innovative approach begins with the functionalization of a substituted phenol derivative, referred to as Compound 1, using a specific reagent system designated as Reagent A. The reaction proceeds through a controlled halomethylation or hydroxymethylation mechanism to generate Compound 2, avoiding the need for high-pressure carbon dioxide insertion entirely. Subsequent oxidation of the side chain converts the intermediate into the corresponding carboxylic acid derivative, Compound 3, which is finally transformed into the target sodium salt through a straightforward acid-base treatment. The entire sequence is characterized by its operational simplicity, utilizing readily available commodity chemicals and standard laboratory glassware or industrial reactors without the need for autoclaves. This fundamental shift in reaction design not only drastically improves the overall yield but also simplifies the downstream processing, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
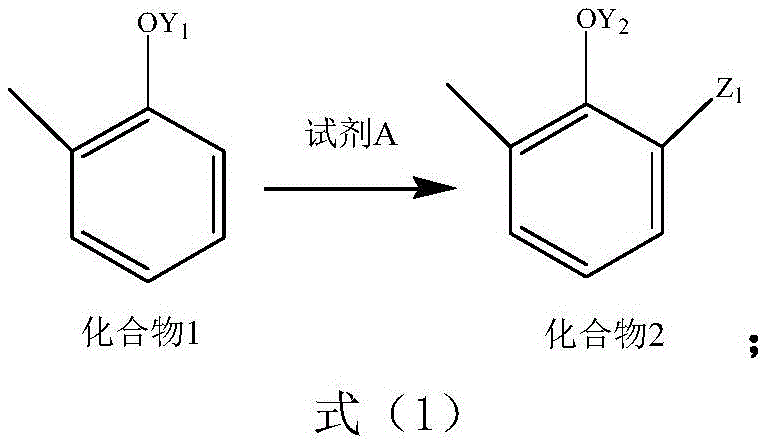
Mechanistic Insights into Electrophilic Substitution and Side-Chain Oxidation
The core of this synthetic strategy lies in the precise control of electrophilic aromatic substitution and subsequent oxidative transformations. In the first stage, Compound 1 undergoes reaction with Reagent A, which may comprise a mixture of formaldehyde and a halogenating agent such as hydrogen chloride, bromine, or thionyl chloride, or alternatively, a dihalomethane like dibromomethane. This step effectively installs a reactive halomethyl or hydroxymethyl group at the ortho-position relative to the phenolic hydroxyl group, directed by the activating nature of the oxygen substituent. The reaction conditions are highly tunable, with temperatures ranging from -50°C to 200°C and pressures from -0.05 to 1MPa, allowing chemists to optimize kinetics for specific substrate batches. The use of additives such as metal chlorides or bromides can further catalyze this transformation, ensuring high conversion rates while minimizing the formation of poly-substituted impurities. This level of control is crucial for maintaining a clean impurity profile, which is a primary concern for R&D teams focused on regulatory compliance.
Following the initial functionalization, the second critical phase involves the oxidation of the benzylic position to generate the carboxylic acid functionality. This transformation is achieved using a versatile array of oxidizing agents, including hydrogen peroxide, potassium permanganate, hypochlorous acid, or ammonium persulfate, dissolved in common organic solvents like tetrahydrofuran, acetone, or acetonitrile. The flexibility in oxidant selection allows manufacturers to balance cost, safety, and environmental impact according to their specific facility capabilities. For instance, hydrogen peroxide offers a green chemistry advantage by producing water as the only by-product, whereas permanganate provides robust oxidation power for difficult substrates. The final step involves a simple neutralization or hydrolysis reaction to convert the acid or ester intermediate into the stable sodium salt form. This modular approach to synthesis ensures that the process can be adapted to various scales, from kilogram-level pilot runs to multi-ton commercial production, without compromising on the structural integrity or purity of the final 3-methyl sodium salicylate product.
How to Synthesize 3-Methyl Sodium Salicylate Efficiently
Implementing this synthesis requires careful attention to stoichiometry and solvent selection to maximize yield and minimize waste. The process is designed to be robust, tolerating slight variations in reaction parameters while still delivering high-quality output. Detailed standard operating procedures for each step, including specific molar ratios and workup protocols, are essential for reproducibility. For a comprehensive guide on the exact experimental conditions and purification techniques referenced in the patent, please refer to the standardized synthesis steps provided below.
- React Compound 1 (o-cresol derivative) with Reagent A (formaldehyde and halogen source) to form Compound 2 via hydrolysis or esterification.
- Subject Compound 2 to an oxidation reaction using oxidizing agents like hydrogen peroxide or potassium permanganate to yield Compound 3.
- Perform hydrolysis and acid-base treatment on Compound 3, followed by recrystallization to obtain purified 3-methyl sodium salicylate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound advantages for procurement managers and supply chain directors tasked with optimizing operational expenditures. The elimination of high-pressure and high-temperature requirements translates directly into reduced capital expenditure for reactor infrastructure, as standard glass-lined or stainless steel vessels can be utilized instead of specialized autoclaves. Furthermore, the use of abundant and inexpensive raw materials, such as formaldehyde and common halogenated solvents, ensures a stable and cost-effective supply chain that is less susceptible to market volatility compared to specialized reagents. The simplified purification process, which relies on straightforward recrystallization techniques using solvents like ethyl acetate or water, significantly reduces the time and energy required for downstream processing. These factors collectively contribute to a substantial reduction in the overall cost of goods sold, enabling more competitive pricing strategies for the final pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The process avoids the use of expensive transition metal catalysts and high-energy inputs associated with high-pressure carboxylation, leading to significant operational savings. By utilizing commodity chemicals and ambient pressure conditions, the manufacturing overhead is drastically lowered, allowing for better margin management in high-volume production scenarios. The high crude yield reported in the examples minimizes raw material waste, further enhancing the economic viability of the process for large-scale commercial application.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures that production schedules are not disrupted by the scarcity of niche reagents. The robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites or scaled up without extensive re-validation, securing a continuous supply of critical intermediates. This reliability is paramount for maintaining uninterrupted production lines for downstream API manufacturers who depend on timely deliveries of high-quality precursors.
- Scalability and Environmental Compliance: The mild reaction conditions and the ability to use greener oxidants like hydrogen peroxide align well with modern environmental, health, and safety (EHS) standards. The simplified waste stream, devoid of heavy metal contaminants often found in catalytic processes, reduces the burden on wastewater treatment facilities and lowers disposal costs. This environmental compatibility facilitates easier regulatory approval and supports the sustainability goals of modern chemical enterprises aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide accurate guidance for potential adopters. Understanding these details is crucial for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the advantages of this new synthesis method over traditional CO2 carboxylation?
A: Unlike traditional methods requiring 10atm pressure and 180°C temperatures with only 38% yield, this patent describes a mild process operating at atmospheric or low pressure with significantly higher yields and easier purification.
Q: What oxidizing agents are suitable for the second step of the reaction?
A: The process allows for a wide range of oxidizing reagents including hydrogen peroxide, potassium permanganate, hypochlorous acid, and ammonium persulfate, providing flexibility for scale-up.
Q: How is the final purity of 3-methyl sodium salicylate ensured?
A: High purity exceeding 99% is achieved through a dedicated recrystallization step using solvents such as ethyl acetate, dimethyl carbonate, or water, effectively removing impurities and by-products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methyl Sodium Salicylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical landscape. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific reaction conditions outlined in this patent, including precise temperature control and specialized oxidation setups, guaranteeing stringent purity specifications for every batch. With our rigorous QC labs and commitment to quality assurance, we deliver 3-methyl sodium salicylate that meets the highest industry standards, supporting your drug development timelines with reliability and precision.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can enhance your bottom line. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions about your supply chain strategy. Let us help you engineer success with our expertise in complex organic synthesis and commercial scale-up.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
