Advanced Synthesis of Benzoborazole 7-Chalcone Derivatives for Commercial Oncology Applications
Advanced Synthesis of Benzoborazole 7-Chalcone Derivatives for Commercial Oncology Applications
The pharmaceutical landscape is constantly evolving with the discovery of novel scaffolds that offer improved therapeutic indices, and patent CN108191898B introduces a significant breakthrough in this domain with its disclosure of benzoborazole 7-chalcone derivatives. These compounds represent a sophisticated fusion of the established benzoborazole pharmacophore, known for its bioavailability and enzyme inhibition capabilities, with the versatile chalcone backbone, which is renowned for its broad-spectrum biological activities including anticancer properties. The invention specifically targets the unmet need for low-toxicity antitumor agents that can effectively inhibit the proliferation of aggressive cancer cell lines such as ovarian SKOV3, breast MDA-MB231, and colon HCT116 without causing severe damage to normal human tissues. This dual functionality positions these derivatives as highly valuable candidates for next-generation oncology drug development programs. 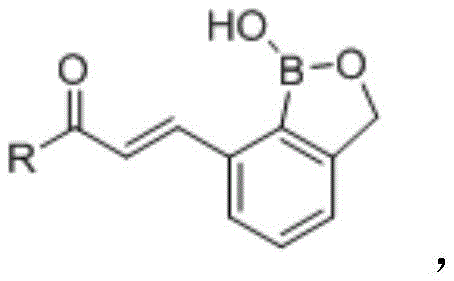
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing complex heterocyclic chalcone derivatives often rely on harsh reaction conditions that can compromise the integrity of sensitive functional groups, particularly the boron-containing moieties which are prone to protodeboronation under extreme pH or thermal stress. Conventional methodologies frequently necessitate the use of anhydrous organic solvents and strong, non-nucleophilic bases that generate substantial hazardous waste streams, thereby increasing the environmental footprint and disposal costs for large-scale manufacturing facilities. Furthermore, many existing protocols depend on transition metal catalysts to facilitate carbon-carbon bond formation, which introduces the risk of heavy metal contamination in the final active pharmaceutical ingredient (API), requiring expensive and time-consuming purification steps to meet stringent regulatory limits. These factors collectively contribute to prolonged lead times and inflated production costs, creating significant bottlenecks for procurement teams aiming to secure reliable supplies of high-purity intermediates for preclinical and clinical trials.
The Novel Approach
In stark contrast, the methodology outlined in the patent data employs a remarkably mild and environmentally benign Claisen-Schmidt condensation strategy that operates efficiently in aqueous alcohol mixtures, drastically reducing the reliance on volatile organic compounds. By utilizing simple inorganic bases such as sodium hydroxide or potassium hydroxide, the process avoids the complexities associated with handling pyrophoric reagents or moisture-sensitive catalysts, making it inherently safer for operators and more robust for scale-up operations. The reaction proceeds smoothly at room temperature or with minimal heating over a flexible time window ranging from 0.5 to 72 hours, allowing for precise control over conversion rates and minimizing the formation of unwanted byproducts. This streamlined approach not only simplifies the operational workflow but also enhances the overall atom economy of the synthesis, aligning perfectly with modern green chemistry principles and offering a sustainable pathway for the commercial production of these high-value pharmaceutical intermediates.
Mechanistic Insights into Base-Catalyzed Claisen-Schmidt Condensation
The core chemical transformation driving the formation of these benzoborazole chalcone derivatives is a classic base-catalyzed aldol condensation followed by dehydration, commonly referred to as the Claisen-Schmidt reaction, which facilitates the construction of the critical alpha,beta-unsaturated ketone linkage. The mechanism initiates with the deprotonation of the alpha-carbon of the acetophenone derivative by the hydroxide ion present in the mixed solvent system, generating a resonance-stabilized enolate nucleophile that is highly reactive towards electrophilic carbonyl centers. This enolate subsequently attacks the aldehyde group located at the 7-position of the benzoborazole ring, forming a beta-hydroxy ketone intermediate which spontaneously undergoes elimination of a water molecule to yield the thermodynamically stable conjugated enone system. The presence of the electron-withdrawing boron atom within the heterocyclic ring may further activate the aldehyde towards nucleophilic attack, potentially accelerating the reaction kinetics and allowing for milder conditions compared to standard benzaldehyde substrates. 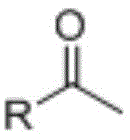
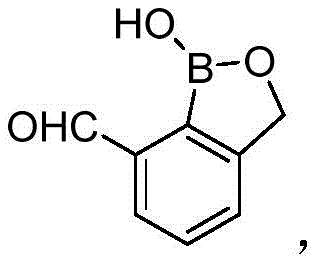
From an impurity control perspective, the simplicity of this mechanistic pathway is advantageous because it minimizes the generation of complex side products that are difficult to separate. The use of aqueous media helps to solubilize inorganic salts and polar byproducts, while the final product, being less polar, precipitates out upon acidification, providing a natural purification step before chromatography. The patent specifies adjusting the pH to acidity using hydrochloric acid post-reaction, which serves to protonate any remaining enolate species and neutralize the base, effectively quenching the reaction and preventing retro-aldol decomposition. This controlled workup ensures that the final filter cake contains the desired chalcone derivative with high chemical purity, reducing the burden on downstream silica gel column chromatography and ensuring that the impurity profile remains within acceptable limits for biological testing and potential therapeutic use.
How to Synthesize Benzoborazole Chalcone Derivatives Efficiently
The synthesis protocol described in the patent offers a robust and reproducible method for generating these antitumor candidates, suitable for both laboratory research and pilot-scale production environments. The process begins by preparing a homogeneous mixture of alcohol and water, into which the base is dissolved to create the reactive medium, followed by the sequential addition of the ketone and the specialized benzoborazole aldehyde. Detailed standardized operating procedures regarding specific molar ratios, solvent volumes, and stirring speeds are critical for maintaining batch-to-batch consistency and maximizing yield. For comprehensive step-by-step instructions including exact quantities and safety precautions, please refer to the technical guide below.
- Prepare a mixed solvent system of methanol or ethanol and water with a volume ratio ranging from 0.5: 1 to 10:1, then add sodium hydroxide or potassium hydroxide.
- Introduce the ketone substrate and the benzoborazole aldehyde precursor into the mixture, stirring continuously for a reaction period between 0.5 to 72 hours.
- Adjust the reaction pH to acidity (pH 1-7) using hydrochloric acid, filter the precipitate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents a compelling value proposition driven by significant operational efficiencies and cost optimization opportunities inherent in the process design. The elimination of expensive transition metal catalysts and the use of commodity-grade reagents like sodium hydroxide and ethanol directly translate to a reduction in raw material expenditure, while the simplified purification workflow decreases the consumption of silica gel and organic eluents. Moreover, the flexibility in reaction time and solvent ratios allows manufacturing partners to adapt the process to existing infrastructure without requiring major capital investment in new reactor types or specialized containment systems, thereby enhancing supply chain resilience and continuity. 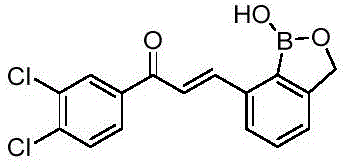
- Cost Reduction in Manufacturing: The economic benefits of this technology are primarily derived from the substitution of precious metal catalysts with inexpensive inorganic bases, which removes the necessity for costly metal scavenging steps and reduces the overall cost of goods sold (COGS). Additionally, the ability to use water as a co-solvent significantly lowers the volume of organic solvents required, leading to substantial savings in solvent purchase, recovery, and waste disposal fees. The high selectivity of the reaction minimizes the formation of hard-to-remove impurities, which further reduces the loss of material during purification and improves the overall process yield, contributing to a more favorable cost structure for large-scale API manufacturing.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the key starting materials, such as substituted acetophenones and the benzoborazole aldehyde precursor, are either commercially available or can be synthesized via well-established routes using abundant feedstocks. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by minor variations in temperature or reagent quality, ensuring consistent output even in diverse manufacturing settings. This stability allows suppliers to maintain steady inventory levels and meet tight delivery schedules, mitigating the risks associated with supply shortages that often plague more complex synthetic pathways reliant on specialized reagents.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being manageable in standard stirred-tank reactors and the absence of hazardous gases or unstable intermediates that would complicate scale-up. From an environmental compliance standpoint, the use of ethanol and water aligns with green chemistry initiatives, reducing the facility's volatile organic compound (VOC) emissions and simplifying the permitting process for expansion. The aqueous waste streams generated are easier to treat compared to those containing heavy metals or chlorinated solvents, facilitating adherence to increasingly stringent environmental regulations and supporting the company's sustainability goals without compromising production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzoborazole derivatives, based on the detailed specifications provided in the patent documentation. Understanding these aspects is crucial for R&D teams evaluating the feasibility of incorporating these intermediates into their drug discovery pipelines and for procurement specialists assessing supplier capabilities. The answers reflect the specific advantages of the disclosed methodology in terms of purity, safety, and biological efficacy.
Q: What represents the primary therapeutic advantage of these benzoborazole derivatives?
A: According to patent CN108191898B, these derivatives exhibit potent antitumor activity against ovarian, breast, and colon cancer cell lines while demonstrating significantly lower toxicity towards normal human cell lines compared to traditional agents.
Q: Does the synthesis process require expensive transition metal catalysts?
A: No, the described methodology utilizes simple inorganic bases like sodium hydroxide or potassium hydroxide in aqueous alcohol solvents, eliminating the need for costly palladium or other transition metal catalysts and simplifying downstream purification.
Q: How does the structural variability of the R group impact biological activity?
A: The patent indicates that substituents such as trifluoromethyl or dichloro groups on the phenyl ring significantly enhance proliferation inhibition, with specific derivatives showing IC50 values in the low micromolar range against SKOV3 cells.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoborazole Chalcone Derivatives Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to translate the innovative synthesis described in patent CN108191898B from laboratory concept to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the growing demand for these promising antitumor intermediates with unwavering consistency. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of benzoborazole chalcone derivative meets the highest standards required for pharmaceutical applications, providing our clients with the confidence needed to advance their projects through clinical stages.
We invite global pharmaceutical companies and research institutions to collaborate with us to leverage this advanced technology for their oncology programs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, as well as obtain specific COA data and route feasibility assessments for your target molecules. Let us help you accelerate your drug development timeline with a reliable supply of high-quality intermediates that combine cutting-edge science with commercial viability.
