Advanced Manufacturing of Diamine-Bridged Lanthanum Complexes for Next-Generation Oncology
Introduction to Patent CN1560060A Technology
The pharmaceutical landscape is continuously evolving towards targeted therapies that minimize systemic toxicity while maximizing efficacy against resistant tumor strains. Patent CN1560060A introduces a groundbreaking class of diamine-bridged ortho-phenanthroline hydrous lanthanum (III) complexes, representing a significant leap forward in rare earth metal-based oncology treatments. This technology leverages the unique coordination chemistry of lanthanum to create stable, rigid structures capable of interacting with nucleic acid secondary structures, offering a novel mechanism of action distinct from traditional platinum-based drugs. For R&D directors and procurement specialists alike, this patent outlines a robust, chemically feasible pathway to produce high-value anti-cancer intermediates that address the critical limitations of current market standards. The synthesis route described is not only scientifically elegant but also commercially viable, utilizing accessible raw materials to construct complex heterocyclic architectures.
As a leading manufacturer in the fine chemical sector, understanding the nuances of this synthesis is crucial for securing a reliable pharmaceutical intermediate supplier partnership. The patent details a multi-step sequence starting from basic aromatic amines and culminating in sophisticated macrocyclic-like lanthanum chelates. This report delves deep into the technical specifics, analyzing the reaction mechanisms, scalability potential, and strategic supply chain benefits inherent in this proprietary methodology. By mastering this synthesis, we can offer cost reduction in anti-cancer drug manufacturing that translates directly to improved margins for downstream pharmaceutical partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional anti-cancer therapies, particularly those based on cisplatin and its derivatives, have long been the cornerstone of chemotherapy regimens. However, their clinical utility is increasingly compromised by severe dose-limiting toxicities, including nephrotoxicity, ototoxicity, and neurotoxicity, which severely impact patient quality of life. Furthermore, the development of cellular resistance mechanisms against platinum drugs has rendered many tumors refractory to treatment, necessitating the urgent discovery of new chemical entities with different modes of action. From a synthesis perspective, creating complex metal-organic frameworks often involves expensive noble metals like palladium or platinum as catalysts, which drives up the cost of goods sold and introduces challenges in removing trace heavy metal impurities to meet stringent regulatory limits. Conventional methods for synthesizing phenanthroline derivatives can also be cumbersome, requiring harsh conditions or multiple protection-deprotection steps that lower overall yields and complicate waste management.
The Novel Approach
The approach detailed in CN1560060A circumvents these issues by utilizing lanthanum, a rare earth metal that offers a favorable toxicity profile and unique electronic properties suitable for DNA binding. The synthetic strategy employs a convergent route where the organic ligand is constructed efficiently before the final metal coordination step. This modularity allows for the easy variation of the diamine bridge, enabling the rapid generation of analog libraries to optimize biological activity without redesigning the entire synthesis. By replacing precious metal catalysts with more abundant reagents like iron powder for reductions and selenium dioxide for oxidations, the process inherently supports cost reduction in electronic chemical manufacturing and pharma sectors alike. The final coordination with lanthanum perchlorate is straightforward, occurring under mild conditions that preserve the integrity of the sensitive organic framework while ensuring high stability of the resulting complex.
Mechanistic Insights into Fe-Mediated Reduction and La-Coordination
The core of this synthesis lies in the construction of the 1,10-phenanthroline backbone and its subsequent functionalization. The process initiates with a classic Skraup reaction, where o-nitroaniline reacts with glycerol in the presence of arsenic pentoxide and sulfuric acid to form 8-nitroquinoline. This intermediate is then subjected to a reduction using iron powder in acetic acid, a method chosen for its selectivity and cost-effectiveness compared to catalytic hydrogenation which might require high-pressure equipment. The resulting 8-aminoquinoline undergoes a Friedländer-type condensation with o-nitrobenzaldehyde and crotonaldehyde, facilitated by aluminum chloride, to close the second ring and form the 2-methyl-1,10-phenanthroline core. This step is critical as it establishes the rigid aromatic system necessary for intercalation into DNA base pairs.
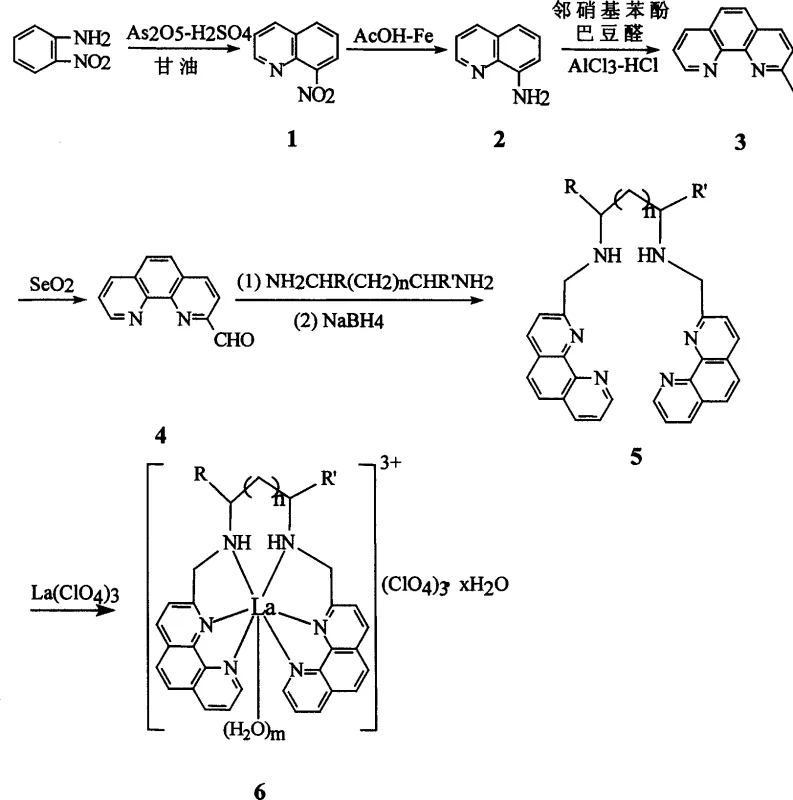
Following the formation of the phenanthroline skeleton, the methyl group at the 2-position is oxidized to an aldehyde using selenium dioxide. This transformation is pivotal as it provides the reactive handle for attaching the diamine bridge. The aldehyde then undergoes a condensation reaction with various aliphatic diamines, such as ethylenediamine or propylenediamine, to form a bis-imine intermediate. This intermediate is immediately reduced in situ using sodium borohydride to yield the stable secondary amine-linked bis-phenanthroline ligand. Finally, this flexible yet pre-organized ligand coordinates with lanthanum perchlorate in methanol. The lanthanum ion, with its high coordination number, binds to the nitrogen atoms of the phenanthroline rings and potentially water molecules, forming a cationic complex stabilized by perchlorate counterions. This precise control over stoichiometry and reaction conditions ensures the formation of the specific hydrate forms described in the patent, which are essential for the observed biological activity.
Impurity control is managed through the selection of reagents and purification techniques. For instance, the use of recrystallization from mixed solvent systems like methanol and diethyl ether effectively removes unreacted ligands and inorganic salts. The patent emphasizes the importance of elemental analysis and spectroscopic characterization (IR, UV-Vis, FABMS) to verify the absence of free metal ions and ensure the correct hydration state. This rigorous attention to detail in the mechanistic execution guarantees a high-purity OLED material or pharmaceutical intermediate that meets the exacting standards required for clinical development. The avoidance of transition metal catalysts in the final steps further simplifies the purification burden, reducing the risk of genotoxic impurities in the final API.
How to Synthesize Diamine-Bridged Lanthanum Complexes Efficiently
Executing this synthesis requires careful attention to reaction parameters, particularly during the Skraup reaction and the final coordination steps. The initial cyclization demands precise temperature control to prevent polymerization of glycerol, while the oxidation step requires stoichiometric balance to avoid over-oxidation of the phenanthroline ring. The reductive amination must be conducted under anhydrous conditions initially to favor imine formation, followed by the addition of the reducing agent. Detailed standardized operating procedures are essential to reproduce the high yields reported in the patent, which range significantly depending on the specific diamine used. For a comprehensive breakdown of the exact molar ratios, temperatures, and workup procedures required to achieve GMP-grade material, please refer to the technical guide below.
- Perform Skraup reaction using o-nitroaniline, glycerol, and arsenic pentoxide in sulfuric acid to generate 8-nitroquinoline.
- Reduce 8-nitroquinoline with iron powder in acetic acid, followed by Friedländer condensation to form 2-methyl-1,10-phenanthroline.
- Oxidize the methyl group to an aldehyde using selenium dioxide, condense with aliphatic diamines, reduce with sodium borohydride, and finally coordinate with lanthanum perchlorate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthesis route outlined in CN1560060A offers substantial strategic advantages over traditional metallo-drug manufacturing. The reliance on commodity chemicals such as glycerol, o-nitroaniline, and simple aliphatic diamines ensures a stable and resilient supply chain, insulating production from the volatility often associated with precious metal markets. This accessibility of raw materials translates directly into enhanced supply chain reliability, allowing for consistent production scheduling and reduced risk of stockouts due to raw material scarcity. Furthermore, the synthetic steps are chemically robust and do not require exotic reagents or specialized high-pressure equipment, making the technology transfer to large-scale manufacturing facilities straightforward and cost-effective.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts like palladium or platinum from the synthetic sequence significantly lowers the direct material costs. Additionally, the use of iron powder for reduction and selenium dioxide for oxidation represents a cost-efficient alternative to catalytic hydrogenation or enzymatic methods, which often require costly infrastructure. The high yields reported in the patent examples indicate minimal waste generation per unit of product, further driving down the cost of goods sold. By streamlining the purification process through simple recrystallization rather than complex chromatography, operational expenses related to solvents and stationary phases are drastically reduced, resulting in substantial cost savings for the final buyer.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are bulk chemicals produced globally in massive quantities, ensuring that supply disruptions are highly unlikely. Unlike specialized chiral ligands or bio-derived precursors that may have limited suppliers, o-nitroaniline and glycerol are available from multiple vendors, fostering a competitive pricing environment. The robustness of the reaction conditions means that production can be scaled up rapidly to meet surging demand without the need for lengthy process re-validation. This flexibility allows supply chain managers to maintain lean inventory levels while still guaranteeing continuity of supply for critical oncology pipelines, thereby reducing lead time for high-purity rare earth intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton commercial production. The waste streams generated, primarily consisting of iron salts and aqueous acidic washes, are well-understood and can be treated using standard effluent treatment protocols, ensuring compliance with environmental regulations. The avoidance of chlorinated solvents in the final steps, favoring alcohols and ethers, aligns with green chemistry principles and reduces the environmental footprint of the manufacturing site. This sustainability profile is increasingly important for pharmaceutical companies aiming to meet their corporate social responsibility goals, making this synthetic route a preferred choice for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these lanthanum complexes. Understanding these details is vital for integrating this technology into your existing drug development portfolio. The answers provided are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of Lanthanum complexes over Cisplatin?
A: According to patent CN1560060A, these lanthanum complexes exhibit broad-spectrum anti-tumor activity against cell lines like PC-3MIE8 and Bel-7402, often surpassing Cisplatin in efficacy while potentially offering a reduced toxicity profile for normal cell lines.
Q: Is the synthesis route scalable for industrial production?
A: Yes, the synthesis utilizes commodity chemicals such as glycerol, o-nitroaniline, and aliphatic diamines. The reaction conditions, including the Skraup reaction and reductive amination, are well-established and amenable to large-scale batch processing.
Q: How is purity controlled in the final coordination step?
A: The process involves rigorous purification steps including recrystallization from mixed solvents like methanol and diethyl ether. Elemental analysis and spectroscopic data (IR, UV-Vis, FABMS) confirm the stoichiometry and removal of unreacted ligands.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lanthanum Phenanthroline Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of rare earth coordination complexes in modern medicine. Our team of expert chemists has thoroughly analyzed the synthetic pathway described in CN1560060A and is fully prepared to support your development needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including residual solvent analysis and heavy metal testing, to guarantee that every batch meets the highest international standards.
We invite you to collaborate with us to unlock the full commercial potential of this innovative anti-cancer technology. By leveraging our manufacturing expertise, you can accelerate your timeline to market while optimizing your budget. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-quality pharmaceutical intermediates.
