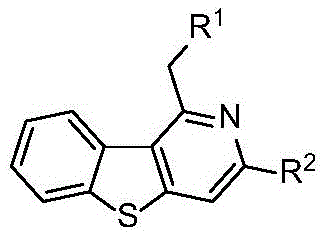
Advanced Transition-Metal-Free Synthesis of Polysubstituted Benzothienopyridine Compounds for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to construct complex heterocyclic scaffolds, which serve as the backbone for numerous active pharmaceutical ingredients (APIs). Patent CN112592352A introduces a groundbreaking methodology for the preparation of polysubstituted benzothienopyridine compounds, a privileged structural motif known for its rich biological activity and potential as a nitrogen ligand in catalysis. This novel approach leverages a transition-metal-free cyclization strategy, utilizing readily available 2-methyl-3-alkynyl benzothiophenes and nitriles as starting materials. By operating under relatively mild thermal conditions without the reliance on precious metal catalysts, this technology addresses critical pain points in modern process chemistry, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates. The versatility of this synthetic route allows for the introduction of diverse functional groups, enabling medicinal chemists to rapidly explore structure-activity relationships (SAR) while maintaining high atom economy.
For procurement managers and supply chain directors, the shift away from transition metal catalysis represents a significant strategic advantage. Traditional methods often rely on palladium or other rare earth metals, which are subject to volatile market pricing and stringent regulatory limits regarding residual metals in final drug products. The methodology disclosed in CN112592352A circumvents these issues entirely by employing inexpensive amino metal salts and alkali bases. This fundamental change in the reaction design not only simplifies the supply chain by reducing dependency on critical raw materials but also streamlines the purification process. As a reliable pharmaceutical intermediate supplier, understanding these upstream process innovations is crucial for ensuring long-term supply continuity and achieving substantial cost reduction in API manufacturing without compromising on the quality or purity of the final output.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzothienopyridine skeleton has been fraught with synthetic challenges that hinder large-scale production. Conventional strategies often involve regioselective cyclization of internal alkynes with aryl imines or vinyl azides, which require specialized and often unstable precursors that are difficult to source in bulk quantities. Other established routes rely heavily on palladium-catalyzed sequential cyclization reactions or tert-butyl nitrite-promoted intramolecular cyclizations. These traditional methods suffer from several inherent drawbacks: they typically demand harsh reaction conditions, exhibit limited substrate scope due to sensitivity to functional groups, and necessitate the use of expensive transition metal catalysts. Furthermore, the presence of heavy metals in the reaction mixture imposes a significant burden on downstream processing, requiring additional purification steps such as scavenging or recrystallization to meet strict pharmacopeial standards for residual metals. These factors collectively increase the cost of goods sold (COGS) and extend the lead time for high-purity benzothienopyridine production, making them less attractive for industrial applications where efficiency and sustainability are paramount.
The Novel Approach
In stark contrast, the invention detailed in CN112592352A offers a streamlined, one-pot synthetic route that dramatically simplifies the manufacturing landscape. By utilizing simple and easily accessible 2-methyl-3-alkynyl benzothiophenes and nitriles, the method achieves high step economy and atom efficiency. The reaction proceeds smoothly in common organic solvents such as cyclopentyl methyl ether, tetrahydrofuran, or 1,4-dioxane, at temperatures ranging from 90°C to 130°C. Crucially, the complete absence of transition metals eliminates the risk of metal contamination, thereby reducing the complexity of the workup procedure. The process demonstrates exceptional tolerance for a wide array of functional groups, including halogens, ethers, and amines, allowing for the direct synthesis of diverse derivatives without the need for protecting group strategies. This operational simplicity, combined with the use of commodity chemicals, positions this technology as a superior alternative for the commercial scale-up of complex heterocycles, directly addressing the needs of both R&D teams seeking rapid analog synthesis and production teams focused on robust manufacturing.
Mechanistic Insights into Base-Mediated Cyclization
The mechanistic pathway of this transformation is a testament to the power of organometallic chemistry driven by strong bases rather than transition metals. The reaction initiates with the deprotonation of the methyl group on the 2-methyl-3-alkynyl benzothiophene by the amino metal salt, such as lithium bis(trimethylsilyl)amide (LiHMDS), generating a reactive benzylic metal species. This nucleophilic intermediate then attacks the electrophilic carbon of the nitrile triple bond, forming a new carbon-carbon bond. Subsequent steps involve a cascade of intramolecular rearrangements, including migration insertion of the alkynyl group and protonation events. A key feature of this mechanism is the 1,5-hydrogen migration, which facilitates the aromatization of the pyridine ring, ultimately yielding the stable polysubstituted benzothienopyridine core. This elegant sequence avoids the oxidative addition and reductive elimination steps typical of palladium cycles, relying instead on the intrinsic reactivity of the carbon nucleophile and the nitrile electrophile.

From an impurity control perspective, this mechanism offers distinct advantages. The absence of transition metals removes a major source of inorganic impurities that are notoriously difficult to remove. Furthermore, the high selectivity of the base-mediated cyclization minimizes the formation of side products such as homocoupling derivatives or polymerization byproducts often seen in radical or metal-catalyzed processes. The reaction conditions are sufficiently mild to preserve sensitive functional groups on the R1 and R2 substituents, ensuring that the impurity profile remains clean and manageable. For R&D directors, this means that the resulting high-purity pharmaceutical intermediates require less intensive purification, leading to higher overall yields and reduced solvent consumption. The ability to predictably control the reaction outcome through stoichiometry and temperature modulation further enhances the reproducibility of the process, a critical factor when transferring technology from the laboratory to pilot and commercial scales.
How to Synthesize Polysubstituted Benzothienopyridine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The protocol involves dissolving the amino metal salt and base in an anhydrous organic solvent before introducing the alkyne and nitrile substrates. Maintaining an inert atmosphere is recommended to prevent moisture interference with the strong base. The reaction mixture is then heated to the optimal temperature range and stirred for a prolonged period to ensure complete conversion. Following the reaction, a standard aqueous workup followed by column chromatography allows for the isolation of the target compound. For detailed operational parameters and specific stoichiometric ratios tailored to different substrates, please refer to the standardized synthesis guide below.
- Charge a reactor with 2-methyl-3-alkynyl benzothiophene, nitrile, lithium bis(trimethylsilyl)amide (LiHMDS), potassium tert-butoxide, and an organic solvent like cyclopentyl methyl ether.
- Stir the reaction mixture at a temperature between 90°C and 130°C for 18 to 24 hours to facilitate the cyclization.
- Cool the reaction, extract with ethyl acetate, dry the organic phase, and purify the crude product via column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this transition-metal-free methodology offers profound commercial benefits that extend beyond the laboratory bench, directly impacting the bottom line and supply chain resilience. By fundamentally altering the input materials and process conditions, manufacturers can achieve significant operational efficiencies. The elimination of precious metal catalysts not only reduces the direct cost of raw materials but also mitigates the supply risk associated with geopolitically sensitive elements like palladium. Moreover, the simplified purification train reduces solvent usage and waste generation, aligning with green chemistry principles and lowering environmental compliance costs. These factors combine to create a more agile and cost-effective manufacturing process capable of responding quickly to market demands for high-purity benzothienopyridine derivatives.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the removal of expensive transition metal catalysts and ligands, which can constitute a significant portion of the material cost in traditional syntheses. Additionally, the avoidance of heavy metal scavengers and the associated filtration steps reduces both material and labor costs. The use of commodity bases like potassium tert-butoxide and widely available solvents further drives down the variable cost per kilogram. This streamlined approach allows for a more competitive pricing structure for the final intermediate, providing a clear advantage in cost reduction in API manufacturing while maintaining high margins.
- Enhanced Supply Chain Reliability: Dependence on a narrow range of specialized catalysts can create bottlenecks in the supply chain. By switching to a chemistry based on abundant nitriles and alkynes, the supply base is significantly broadened, reducing the risk of shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output. This reliability is crucial for maintaining continuous production schedules and meeting the strict delivery timelines required by global pharmaceutical clients, effectively reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and pressures, making it inherently safer and easier to scale from gram to ton quantities without requiring specialized high-pressure equipment. The absence of toxic heavy metals simplifies waste treatment and disposal, lowering the environmental footprint of the manufacturing site. This ease of scale-up ensures that the process can seamlessly transition from clinical trial material production to commercial manufacturing, supporting the long-term growth of the product portfolio without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these benzothienopyridine compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing accurate insights for decision-makers evaluating this technology for their own pipelines.
Q: What are the primary advantages of this synthesis method over traditional palladium-catalyzed routes?
A: This method eliminates the need for expensive and toxic transition metal catalysts like palladium. It utilizes simple, commercially available bases and amino metal salts, significantly reducing raw material costs and removing the complex downstream processing required for heavy metal removal, thereby enhancing overall process safety and environmental compliance.
Q: What is the substrate scope for the R1 and R2 substituents in this reaction?
A: The reaction demonstrates excellent functional group tolerance. R1 can include various substituted benzene rings with groups such as methoxy, dimethylamino, fluorine, or chlorine. R2 offers similar diversity, accommodating benzene rings, pyridines, and substituents like methyl or methoxymethyl ether, allowing for the synthesis of a wide library of derivatives for SAR studies.
Q: Can this methodology be applied to the late-stage functionalization of existing drug molecules?
A: Yes, the patent explicitly demonstrates the utility of this method for post-modification of pharmaceutical molecules. A specific example includes the successful derivatization of Citalopram, proving that the mild reaction conditions are compatible with complex bioactive scaffolds, making it highly valuable for medicinal chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothienopyridine Supplier
The technological advancements described in CN112592352A represent a significant leap forward in heterocyclic chemistry, offering a pathway to high-value intermediates with superior efficiency and purity. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative academic and patent technologies into robust, commercial-grade manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves smoothly from concept to market. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next drug development program. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in driving innovation and efficiency in your supply chain.
