Advanced Resolution Technology for High-Purity 1-Indanone-3-Acetic Acid Intermediates
Advanced Resolution Technology for High-Purity 1-Indanone-3-Acetic Acid Intermediates
The pharmaceutical industry continuously seeks robust and cost-effective pathways for synthesizing chiral intermediates, particularly for high-value drugs like Ramelteon, a selective melatonin receptor agonist used in treating insomnia. Patent CN102417497A introduces a groundbreaking preparation method for optically active 1-indanone-3-acetic acid compounds, which serve as critical building blocks in this therapeutic class. This technology addresses long-standing challenges in chiral resolution by replacing complex amide formation strategies with a streamlined diastereomeric salt formation process. By leveraging specific chiral organic secondary amines, manufacturers can achieve superior optical purity while drastically simplifying the operational workflow. This report analyzes the technical merits and commercial implications of this innovation for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for optically active 1-indanone-3-acetic acid derivatives typically rely on the formation of diastereomeric amides using expensive condensing agents. As illustrated in the prior art, the racemic starting material reacts with a chiral amine to form an amide blend, which necessitates multiple recrystallizations or column chromatography to achieve separation. 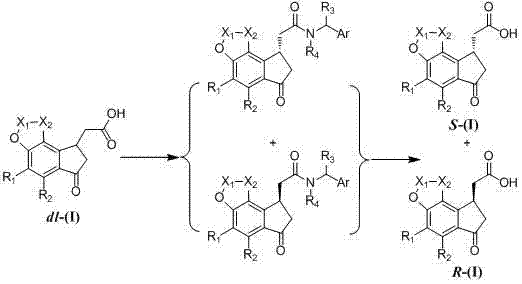 Furthermore, the subsequent recovery of the free acid requires hydrolysis under strongly acidic or alkaline conditions, which poses a severe risk of racemization and reduces the overall optical purity of the final product. These harsh conditions not only compromise quality but also generate significant chemical waste, increasing environmental compliance costs and complicating the purification process for large-scale operations.
Furthermore, the subsequent recovery of the free acid requires hydrolysis under strongly acidic or alkaline conditions, which poses a severe risk of racemization and reduces the overall optical purity of the final product. These harsh conditions not only compromise quality but also generate significant chemical waste, increasing environmental compliance costs and complicating the purification process for large-scale operations.
The Novel Approach
In stark contrast, the novel method disclosed in the patent utilizes a direct resolution strategy via diastereomeric ammonium salt formation, bypassing the need for covalent amide bond formation entirely. The process involves reacting the racemic 1-indanone-3-acetic acid with a chiral organic secondary amine in a suitable solvent to precipitate the desired diastereomeric salt directly. 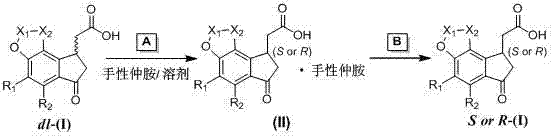 This salt is then subjected to a mild acid neutralization step to liberate the optically active acid, avoiding the destructive hydrolysis conditions of the conventional route. This approach significantly reduces the number of unit operations, eliminates the cost of condensing agents, and ensures that the stereochemical integrity of the molecule is preserved throughout the isolation process, resulting in consistently high enantiomeric excess.
This salt is then subjected to a mild acid neutralization step to liberate the optically active acid, avoiding the destructive hydrolysis conditions of the conventional route. This approach significantly reduces the number of unit operations, eliminates the cost of condensing agents, and ensures that the stereochemical integrity of the molecule is preserved throughout the isolation process, resulting in consistently high enantiomeric excess.
Mechanistic Insights into Chiral Secondary Amine Resolution
The core of this technological advancement lies in the specific selection of chiral organic secondary amines that form stable, crystalline salts with the target acid. The patent highlights the efficacy of amines such as N-benzyl-a-phenylethylamine and N,N'-bis-[(R)-1-styroyl] ethylenediamine, which possess bulky steric groups that enhance the solubility differences between the diastereomeric salts. 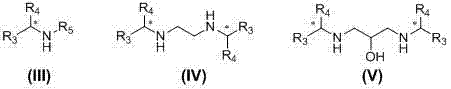 These structural features facilitate the preferential crystallization of one enantiomer over the other from solvents like methanol, acetone, or ethyl acetate. The mechanism relies on the reversible ionic interaction between the carboxylic acid and the amine, which is strong enough to allow for selective crystallization but weak enough to be easily broken by simple acidification. This reversible nature is key to preventing the thermal or chemical degradation often seen in covalent modification strategies.
These structural features facilitate the preferential crystallization of one enantiomer over the other from solvents like methanol, acetone, or ethyl acetate. The mechanism relies on the reversible ionic interaction between the carboxylic acid and the amine, which is strong enough to allow for selective crystallization but weak enough to be easily broken by simple acidification. This reversible nature is key to preventing the thermal or chemical degradation often seen in covalent modification strategies.
Furthermore, the impurity profile is tightly controlled through the crystallization of the intermediate salt, which acts as a powerful purification step before the final acid is released. By optimizing the molar ratio of the chiral amine to the racemic acid, typically between 0.5 to 1.2 equivalents, the process maximizes the yield of the desired isomer while minimizing the retention of the unwanted enantiomer in the crystal lattice. The mother liquor containing the undesired isomer can potentially be recycled after racemization, further enhancing the atom economy of the process. This level of control over the solid-state chemistry ensures that the final product meets the stringent purity specifications required for pharmaceutical applications without the need for extensive downstream processing.
How to Synthesize 1-Indanone-3-Acetic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this resolution technology in a production environment. The process is designed to be operationally simple, requiring standard reactor equipment and common organic solvents that are readily available in most chemical manufacturing facilities. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and quality control.
- React racemic 1-indanone-3-acetic acid with a chiral organic secondary amine in a suitable solvent to form a diastereomeric ammonium salt, followed by crystallization to isolate the desired stereoisomer.
- Neutralize the isolated diastereomeric salt with an aqueous acid to release the free optically active 1-indanone-3-acetic acid, then extract and purify the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution method offers tangible benefits in terms of cost structure and supply reliability. The elimination of condensing agents, which are often costly and require careful handling, directly reduces the raw material bill for every batch produced. Additionally, the simplified workflow reduces the consumption of solvents and energy associated with multiple recrystallization and hydrolysis steps, leading to substantial operational expenditure savings. The robustness of the process also minimizes the risk of batch failures due to racemization, ensuring a more predictable and consistent output of high-value intermediates.
- Cost Reduction in Manufacturing: The removal of expensive coupling reagents and the reduction in solvent usage significantly lower the variable cost per kilogram of the final product. By avoiding complex chromatographic separations and harsh hydrolysis conditions, the process reduces the load on waste treatment facilities and lowers the cost of regulatory compliance. These efficiencies translate into a more competitive pricing structure for the final API intermediate, allowing for better margin management in volatile markets.
- Enhanced Supply Chain Reliability: The reagents required for this method, such as chiral phenylethylamines and common solvents like acetone and methanol, are commercially available from multiple global suppliers, reducing dependency on single-source vendors. The simplicity of the reaction conditions means that the process can be easily transferred between manufacturing sites without significant re-engineering, ensuring continuity of supply even during regional disruptions. This flexibility is crucial for maintaining the production schedules of downstream pharmaceutical clients who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of hazardous hydrolysis byproducts make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The reduced generation of chemical waste aligns with modern green chemistry principles, simplifying the permitting process for new manufacturing lines. This environmental advantage not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing organization, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable basis for decision-making.
Q: Why is the chiral salt formation method superior to amide formation for this intermediate?
A: The chiral salt formation method eliminates the need for expensive condensing agents and avoids harsh hydrolysis conditions required for amide cleavage, thereby preventing racemization and reducing production costs significantly.
Q: What represents the key advantage in terms of optical purity for this process?
A: This process achieves exceptionally high optical purity, often exceeding 99% ee, because the mild acid neutralization step preserves the stereochemical integrity of the molecule unlike strong acid or base hydrolysis.
Q: Is this resolution method scalable for industrial production?
A: Yes, the method utilizes common solvents like methanol and acetone and avoids complex purification steps like column chromatography, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Indanone-3-Acetic Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced chiral resolution technologies to deliver high-quality pharmaceutical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102417497A are fully realized in practical manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-indanone-3-acetic acid meets the exacting standards required for the synthesis of complex therapeutic agents like Ramelteon.
We invite potential partners to engage with our technical procurement team to discuss how this optimized resolution route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines and volume needs.
