Advanced Iridium-Catalyzed Synthesis of Chiral Tetrahydroquinazoline Derivatives for Commercial Scale
Introduction to High-Enantioselective Tetrahydroquinazoline Synthesis
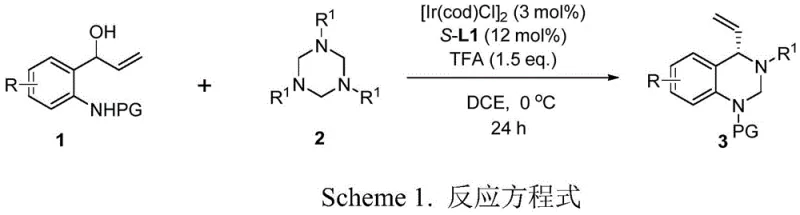
The development of efficient synthetic routes for chiral heterocycles remains a cornerstone of modern pharmaceutical process research, particularly for scaffolds found in bioactive small molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN116375653A, which discloses a robust method for synthesizing chiral tetrahydroquinazoline derivatives with exceptional enantioselectivity. This technology leverages an asymmetric [4+2]-cycloaddition strategy, utilizing a sophisticated iridium catalyst system combined with a chiral ligand to transform readily available racemic 2-aminophenyl allyl alcohols and 1,3,5-triazine compounds into high-value chiral intermediates. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate supplier partnerships, this methodology represents a paradigm shift from traditional, restrictive syntheses to a more scalable and versatile platform. The process operates under mild conditions, typically at 0°C, and demonstrates remarkable tolerance for various substituents, thereby addressing critical challenges in the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the synthesis of chiral tetrahydroquinazoline derivatives has been notably constrained by substrate availability and reaction efficiency. As highlighted in the background of the patent, earlier approaches, such as the copper-catalyzed asymmetric [4+2]-cycloaddition reported in 2018, relied heavily on 4-ethynyl-3,1-benzoxazin-2-one substrates. These precursors are notoriously difficult to synthesize, often requiring multi-step sequences that inflate raw material costs and extend lead times for high-purity intermediates. Furthermore, these conventional methods frequently suffered from low reaction yields and poor stereoselectivity, limiting their utility in the production of clinical-grade materials. The narrow substrate scope of these older methodologies meant that introducing structural diversity required extensive re-optimization, creating bottlenecks in the drug discovery pipeline and hindering cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the novel iridium-catalyzed protocol described in CN116375653A utilizes racemic 2-aminophenyl allyl alcohols, which are significantly more accessible and commercially viable starting materials. By employing a specific combination of an iridium dimer catalyst and a chiral phosphoramidite ligand (S-L1), this method achieves high regioselectivity and enantioselectivity simultaneously. The reaction conditions are remarkably mild, proceeding effectively at 0°C in dichloroethane (DCE) with trifluoroacetic acid (TFA) as an additive. This approach not only simplifies the operational complexity but also expands the chemical space accessible to medicinal chemists, allowing for the incorporation of diverse functional groups such as halogens, alkyls, and alkoxy groups without compromising optical purity. This technological leap facilitates the rapid generation of diverse libraries for biological screening while ensuring a streamlined path to commercial production.
Mechanistic Insights into Iridium-Catalyzed Asymmetric [4+2]-Cycloaddition
The core of this transformative synthesis lies in the precise orchestration of the iridium catalytic cycle. The reaction initiates with the coordination of the iridium precursor, typically [Ir(cod)Cl]2, with the chiral ligand S-L1 to form the active cationic iridium species. This chiral catalyst then activates the racemic 2-aminophenyl allyl alcohol, likely through the formation of a pi-allyl iridium intermediate, which serves as the electrophilic partner in the cycloaddition. The 1,3,5-triazine compound acts as the nucleophile, attacking the activated allyl system in a highly stereocontrolled manner dictated by the chiral environment of the ligand. The presence of the TFA additive is crucial, as it likely assists in the protonation steps necessary for the turnover of the catalytic cycle and the final aromatization or stabilization of the tetrahydroquinazoline core. This mechanistic pathway ensures that even though the starting alcohol is racemic, the dynamic kinetic resolution or specific enantioconvergent process leads to a single dominant enantiomer of the product.
Understanding the impurity profile is vital for regulatory compliance, and this catalytic system excels in minimizing side reactions. The high regioselectivity observed prevents the formation of unwanted isomeric byproducts that often plague cycloaddition reactions. The specific interaction between the bulky chiral ligand and the substrate sterically hinders alternative reaction pathways, ensuring that the [4+2]-cyclization occurs exclusively at the desired position. This intrinsic selectivity reduces the burden on downstream purification processes, such as chromatography or recrystallization, which are often the most costly and time-consuming steps in process chemistry. By controlling the stereochemistry at the bond-forming step, the process inherently delivers high-purity intermediates, aligning perfectly with the stringent quality requirements of global pharmaceutical supply chains.
How to Synthesize Chiral Tetrahydroquinazoline Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to maximize yield and enantiomeric excess. The procedure involves the in situ generation of the active catalyst followed by the sequential addition of substrates under an inert atmosphere. The reaction is sensitive to moisture and oxygen, necessitating the use of argon protection and anhydrous solvents. While the general protocol is robust, fine-tuning the stoichiometry of the triazine component and the acid additive can further enhance performance for specific substrate classes. For a detailed breakdown of the standardized operating procedures, please refer to the technical guide below.
- Prepare the catalytic system by dissolving [Ir(cod)Cl]2 (3 mol%) and chiral ligand S-L1 (12 mol%) in DCE under argon protection.
- Add racemic 2-aminophenyl allyl alcohol, 1,3,5-triazine compound, and TFA additive (1.5 eq.) to the reaction mixture.
- Stir the reaction at 0°C for 24 hours, then quench and purify the crude product to obtain the chiral tetrahydroquinazoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this iridium-catalyzed methodology offers compelling advantages that directly impact the bottom line and supply chain resilience. The shift from complex, custom-synthesized benzoxazinones to commodity-grade allylic alcohols drastically simplifies the raw material procurement landscape. This change reduces dependency on niche suppliers and mitigates the risk of supply disruptions, ensuring enhanced supply chain reliability for long-term projects. Furthermore, the mild reaction conditions eliminate the need for energy-intensive heating or cryogenic cooling beyond standard chillers, contributing to substantial cost savings in utility consumption and reactor occupancy time.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the accessibility of starting materials and the efficiency of the catalytic system. By utilizing racemic 2-aminophenyl allyl alcohols, which are easier to source and synthesize than previous precursors, manufacturers can significantly lower the cost of goods sold (COGS). Additionally, the high catalytic efficiency allows for low metal loading (3 mol% Ir), reducing the expense associated with precious metal recovery and waste treatment. The elimination of complex purification steps due to high selectivity further drives down processing costs, making this route economically superior for large-scale production.
- Enhanced Supply Chain Reliability: The broad substrate scope demonstrated in the patent data implies that this chemistry is not fragile; it can accommodate various electronic and steric environments on the aromatic rings. This robustness means that supply chain managers do not need to qualify multiple distinct synthetic routes for different analogues within a drug series. The ability to use a single, unified platform for generating diverse tetrahydroquinazoline derivatives streamlines vendor qualification and inventory management, reducing lead time for high-purity intermediates and accelerating time-to-market for new drug candidates.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is facilitated by the use of standard solvents like dichloroethane and the absence of hazardous reagents. The reaction proceeds at 0°C to room temperature, which is easily manageable in standard glass-lined or stainless steel reactors without specialized equipment. Moreover, the high atom economy of the [4+2]-cycloaddition and the minimal formation of byproducts align with green chemistry principles, simplifying waste disposal and environmental compliance reporting. This ease of scale-up ensures a smooth transition from process development to commercial manufacturing without the typical teething problems associated with complex asymmetric syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and claims within CN116375653A, providing clarity for technical teams evaluating this route for their specific projects. Understanding these nuances is essential for effective technology transfer and risk assessment.
Q: What are the key advantages of this iridium-catalyzed method over previous copper-catalyzed routes?
A: Unlike previous copper-catalyzed methods requiring difficult-to-synthesize 4-ethynyl-3,1-benzoxazin-2-one substrates with limited scope, this iridium-catalyzed protocol utilizes readily available racemic 2-aminophenyl allyl alcohols. It offers significantly broader substrate applicability, milder reaction conditions (0°C), and superior enantioselectivity (up to 99% ee).
Q: What is the typical catalyst loading and reaction temperature for this synthesis?
A: The optimized protocol employs a low loading of the iridium dimer [Ir(cod)Cl]2 at 3 mol% paired with 12 mol% of the chiral phosphoramidite ligand S-L1. The reaction proceeds efficiently at mild temperatures, typically maintained at 0°C to room temperature, ensuring high stereocontrol without requiring cryogenic conditions.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates excellent functional group tolerance. The patent data confirms successful synthesis with various substituents including halogens (Cl, Br), electron-donating groups (methoxy, methyl), and different nitrogen protecting groups (Ts, Ms, Ns), making it highly versatile for generating diverse pharmaceutical intermediate libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Tetrahydroquinazoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain a competitive edge in the pharmaceutical industry. Our team of expert process chemists has thoroughly analyzed the iridium-catalyzed route described in CN116375653A and is fully equipped to translate this academic innovation into robust commercial processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are designed to handle sensitive organometallic chemistry with precision, supported by rigorous QC labs that guarantee stringent purity specifications for every batch delivered.
We invite you to collaborate with us to leverage this advanced technology for your next drug development program. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this route can optimize your budget. We encourage you to contact us today to request specific COA data for similar scaffolds and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
