Advanced Catalytic Strategy for High-Purity Felodipine Intermediates and Commercial Scalability
Introduction to Advanced Intermediate Synthesis
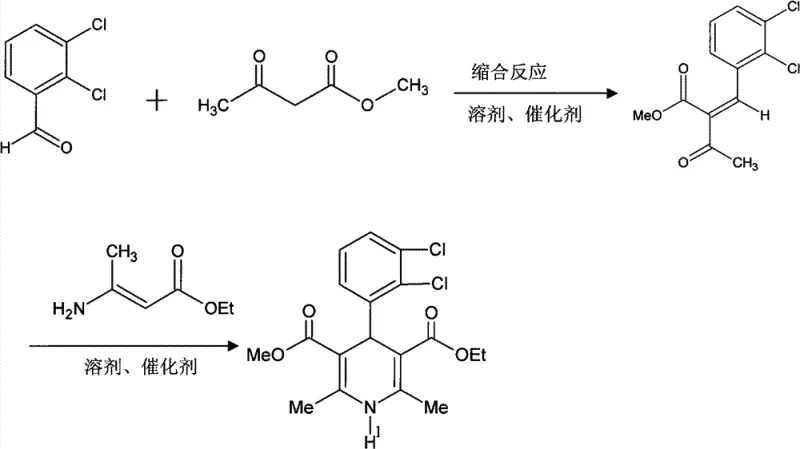
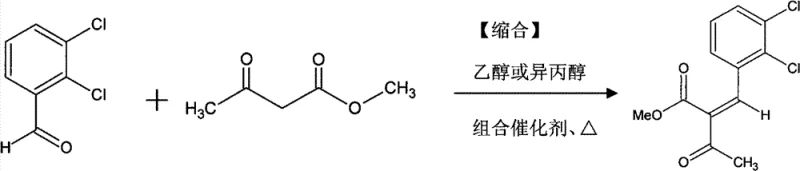
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for critical antihypertensive agents, particularly within the 1,4-dihydropyridine class. Patent CN101613280B introduces a groundbreaking preparation method for methyl 2-(2,3-dichlorobenzylidine)acetoacetate, a pivotal synthetic intermediate for felodipine and related drugs. This innovation centers on a novel combined catalyst system comprising a secondary amine and quinoline carboxylic acid, which fundamentally alters the reaction landscape compared to traditional single-catalyst approaches. By optimizing the condensation reaction between 2,3-dichlorobenzaldehyde and methyl acetoacetate, this technology achieves exceptional purity levels exceeding 98% while maintaining high yields. The strategic implementation of this catalytic duo not only suppresses unwanted side reactions but also streamlines the downstream purification process, addressing long-standing challenges in the manufacturing of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzylidene acetoacetate derivatives has been plagued by significant chemical inefficiencies when relying on standard acidic or basic catalysts. Traditional methods utilizing strong bases like piperidine or diethylamine often necessitate rigorous low-temperature controls to prevent the degradation of the sensitive ethylene linkage in the benzal structure. Furthermore, these conditions frequently lead to the formation of troublesome diadducts, where a second molecule of methyl acetoacetate reacts with the initial condensation product, creating difficult-to-remove impurities. Conversely, acid-catalyzed pathways are prone to generating aldol condensation by-products, which complicate the isolation of the target intermediate and drastically reduce overall process efficiency. Previous attempts to mitigate these issues, such as the use of piperidine acetate salts, have shown limited success, often failing to deliver both high yield and high purity simultaneously without extensive and costly recrystallization steps.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by employing a synergistic catalyst system that overcomes the inherent drawbacks of single-component catalysts. By combining a secondary amine with quinoline carboxylic acid, the reaction environment is finely tuned to promote the desired condensation while effectively suppressing the formation of diadducts and aldol by-products. This dual-catalyst approach allows the reaction to proceed smoothly at moderate temperatures ranging from 30°C to 60°C, eliminating the need for energy-intensive cryogenic conditions. The result is a process that not only delivers superior yields in the range of 80% to 85% but also produces intermediates with crystalline properties that facilitate easy filtration and washing. This technological advancement ensures that the primary precipitate already possesses high purity, significantly reducing the burden on subsequent purification stages and enhancing the overall economic feasibility of the synthesis.
Mechanistic Insights into Combined Amine-Quinoline Catalysis
The efficacy of this novel synthesis route lies in the unique mechanistic interplay between the secondary amine and the quinoline carboxylic acid within the alcoholic solvent medium. The secondary amine acts as the primary base to initiate the Knoevenagel-type condensation by activating the methylene group of the methyl acetoacetate, while the quinoline carboxylic acid serves as a crucial co-catalyst that modulates the reaction kinetics. This modulation prevents the excessive basicity that typically leads to polymerization or multiple additions, thereby steering the reaction pathway exclusively towards the formation of the mono-condensed benzylidene product. The specific choice of quinoline derivatives, such as 2-quinolinecarboxylic acid, provides a steric and electronic environment that stabilizes the transition state, ensuring high regioselectivity and minimizing the generation of structural isomers or oligomeric impurities.
Furthermore, the impurity control mechanism is intrinsically linked to the physical properties of the catalyst components, particularly the quinoline carboxylic acid. Unlike pyridine carboxylic acids used in prior art, quinoline carboxylic acids exhibit significantly lower solubility in cold water, a property that is ingeniously exploited for catalyst recovery and product purification. During the workup phase, simple water washing effectively removes the bulk of the catalyst from the organic phase without dissolving the product, thereby preventing catalyst contamination in the final API intermediate. This distinct solubility profile ensures that the reaction mixture remains clean throughout the process, allowing for the direct isolation of high-purity crystals upon cooling. The minimization of foreign bodies and by-products simplifies the analytical profile of the intermediate, providing R&D teams with a reliable and consistent starting material for the subsequent cyclization steps required to produce the final dihydropyridine drug substance.
How to Synthesize Methyl 2-(2,3-dichlorobenzylidine)acetoacetate Efficiently
The operational protocol for this synthesis is designed for straightforward execution in standard chemical reactors, requiring precise control over stoichiometry and temperature to maximize the benefits of the combined catalyst system. The process begins with the dissolution of 2,3-dichlorobenzaldehyde and methyl acetoacetate in a suitable alcohol solvent, such as absolute ethanol or isopropanol, followed by the sequential addition of the secondary amine and quinoline carboxylic acid. Maintaining the reaction temperature within the optimal window of 38°C to 47°C for a duration of 6 to 8 hours is critical to achieving complete conversion while preserving the integrity of the product. Upon completion, the reaction mixture is subjected to controlled cooling crystallization, which induces the precipitation of the target intermediate in a highly pure crystalline form, ready for filtration and drying. For a comprehensive, step-by-step technical guide including exact molar ratios and safety precautions, please refer to the standardized synthesis instructions below.
- Prepare the reaction mixture by combining 2,3-dichlorobenzaldehyde and methyl acetoacetate in an alcohol solvent such as ethanol or isopropanol.
- Add the novel combined catalyst system consisting of a secondary amine (e.g., piperidine) and quinoline carboxylic acid (e.g., 2-quinolinecarboxylic acid) under controlled temperature conditions.
- Maintain the reaction at 30-60°C for 4-10 hours, then cool the solution to crystallize the product, followed by filtration, washing, and drying to obtain high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for procurement managers and supply chain directors focused on cost optimization and reliability. The ability to recover and recycle the quinoline carboxylic acid catalyst through simple aqueous extraction significantly reduces the consumption of expensive reagents, leading to substantial cost savings in raw material expenditures over large production volumes. Additionally, the high purity of the crude product minimizes the need for multiple recrystallization cycles, which translates to reduced solvent usage, lower energy consumption for heating and cooling, and decreased waste disposal costs. These efficiencies collectively contribute to a more sustainable and economically attractive manufacturing process that aligns with modern green chemistry principles and regulatory expectations for environmental compliance.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the efficient recovery of catalysts directly lower the operational expenditure associated with producing high-quality pharmaceutical intermediates. By avoiding the use of harsh conditions and expensive specialty reagents required in older methods, manufacturers can achieve a leaner production model that maximizes output value while minimizing input costs. This economic efficiency is further amplified by the high yield of the reaction, which ensures that a greater proportion of raw materials are converted into saleable product rather than lost as waste.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures consistent batch-to-batch quality, which is essential for maintaining uninterrupted supply chains in the highly regulated pharmaceutical sector. The use of readily available and stable raw materials, combined with a process that is tolerant to minor variations in operating conditions, reduces the risk of production delays or batch failures. This reliability allows supply chain planners to forecast inventory needs with greater confidence and secure long-term contracts with downstream API manufacturers who demand stringent quality assurances.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to perform effectively from laboratory bench scales up to industrial production capacities without loss of efficiency or purity. The simplified workup procedure, which relies on basic physical separation techniques like filtration and crystallization rather than complex chromatographic methods, facilitates easy scale-up in multi-purpose reactors. Moreover, the reduced generation of hazardous by-products and the ability to recycle solvents and catalysts support stricter environmental regulations, positioning manufacturers as responsible partners in the global pharmaceutical supply network.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology for felodipine intermediates. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the method's practical benefits. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of the quinoline carboxylic acid catalyst system over traditional methods?
A: The novel combined catalyst system significantly improves reaction selectivity, minimizing by-products like diadducts and aldol condensation impurities common in acid or base-catalyzed processes. It also facilitates easier catalyst recovery due to the low solubility of quinoline carboxylic acid in cold water.
Q: What purity levels can be achieved with this synthesis method?
A: This method consistently yields intermediates with purity exceeding 98% directly from the primary crystallization. Further recrystallization of the mother liquor products can achieve purity levels greater than 99%, ensuring high quality for downstream pharmaceutical synthesis.
Q: Is this process scalable for industrial production of antihypertensive drug intermediates?
A: Yes, the process operates under mild temperatures (30-60°C) and uses common alcohol solvents, making it highly suitable for large-scale manufacturing. The efficient recovery and recycling of catalysts and unreacted raw materials further enhance its economic viability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 2-(2,3-dichlorobenzylidine)acetoacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antihypertensive medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic methods described in patent CN101613280B can be seamlessly translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of methyl 2-(2,3-dichlorobenzylidine)acetoacetate performs flawlessly in your downstream synthesis of felodipine and related 1,4-dihydropyridines.
We invite pharmaceutical partners to collaborate with us to leverage this cutting-edge technology for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive efficiency and quality in your antihypertensive drug production.
