Advanced Synthesis of 2-Fluoro-3-Bromo-Benzylamine for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for key intermediates that balance high purity with economic viability. Patent CN113372223B introduces a transformative method for the preparation of 2-fluoro-3-bromo-benzylamine, a critical building block in the synthesis of various bioactive molecules. This innovation addresses long-standing challenges in the field by utilizing 2-fluoro-3-bromobenzonitrile as a starting material, which is both cost-effective and readily available on the global market. The core of this technological breakthrough lies in the strategic application of borane dimethyl sulfide (BH3.Me2S) as a reducing agent, coupled with a meticulously controlled temperature profile and a unique quenching protocol. By shifting away from traditional high-pressure hydrogenation or multi-step functional group interconversions, this patent outlines a pathway that significantly streamlines the manufacturing process. For R&D directors and procurement specialists, understanding the nuances of this method is essential, as it represents a shift towards more sustainable and efficient chemical production. The detailed procedural steps provided in the patent data suggest a high degree of reproducibility, which is a cornerstone for reliable supply chain management in the competitive landscape of pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-fluoro-3-bromo-benzylamine has been plagued by inefficiencies that drive up costs and complicate supply chains. One prominent prior art method, described in U.S. Patent US8067408 B2, relies on the reduction of 3-bromo-2-fluorobenzamide. This route is inherently flawed due to its multi-step nature, requiring the initial formation of benzoyl chloride followed by amidation before the final reduction can occur. The cumulative yield of such a sequence is often disappointing, with reported reduction yields hovering around 46%, which is economically unsustainable for large-scale operations. Furthermore, the handling of acid chlorides introduces significant safety hazards and requires stringent corrosion-resistant equipment, adding to the capital expenditure. Another approach involves the conversion of aldehydes to oximes followed by reduction, as seen in PCT patent WO2018/15879. While this method achieves better yields, it necessitates the use of ten equivalents of reducing agent and subsequent acidic hydrolysis, generating substantial chemical waste and complicating the downstream purification process. These conventional pathways often involve dangerous reagents or harsh conditions that pose risks to operational safety and environmental compliance, making them less attractive for modern green chemistry initiatives.
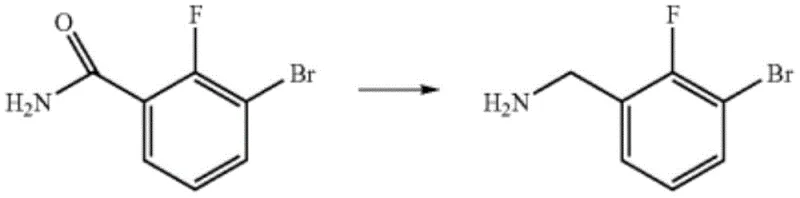
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach detailed in CN113372223B offers a streamlined, direct reduction strategy that bypasses unnecessary intermediate isolation steps. By starting directly from 2-fluoro-3-bromobenzonitrile, the process eliminates the need for pre-functionalization into amides or oximes, thereby reducing the overall number of unit operations required. The selection of BH3.Me2S as the reducing agent is particularly strategic; it provides a potent source of hydride ions capable of efficiently reducing the nitrile group to the corresponding amine under mild conditions. The patent specifies a reaction temperature range of -5°C to 5°C during the addition phase, which is critical for controlling the exothermic nature of the borane reaction and preventing the formation of side products. Following the addition, the reaction is allowed to warm to room temperature and stir for 8 to 15 hours, ensuring complete conversion without the need for energy-intensive heating. This methodology not only improves the atomic economy of the synthesis but also enhances the safety profile by avoiding high-pressure hydrogen gas and pyrophoric metal catalysts like Raney nickel, which are common in alternative nitrile reduction protocols.
Mechanistic Insights into BH3.Me2S-Catalyzed Nitrile Reduction
The mechanistic underpinning of this synthesis relies on the nucleophilic attack of the borane species on the electrophilic carbon of the nitrile group. When BH3.Me2S is introduced to the solution of 2-fluoro-3-bromobenzonitrile, the boron atom coordinates with the nitrogen lone pair, activating the triple bond towards hydride transfer. This coordination complex is sensitive to temperature fluctuations, which explains the patent's strict requirement for cooling the reaction mixture to between -5°C and 5°C during the dropwise addition. Maintaining this low temperature prevents the rapid evolution of heat that could lead to runaway reactions or the decomposition of the sensitive borane complex. Once the addition is complete, the mixture is stirred at room temperature, allowing the kinetic energy to facilitate the full reduction of the intermediate imine-borane complex to the amine-borane adduct. The use of an inert atmosphere, preferably nitrogen or argon, is crucial throughout this stage to prevent the oxidation of the borane reagent by atmospheric moisture or oxygen, which would otherwise deactivate the reducing agent and lower the overall yield. This precise control over reaction conditions ensures that the fluorine and bromine substituents on the aromatic ring remain intact, preserving the structural integrity required for downstream pharmaceutical applications.
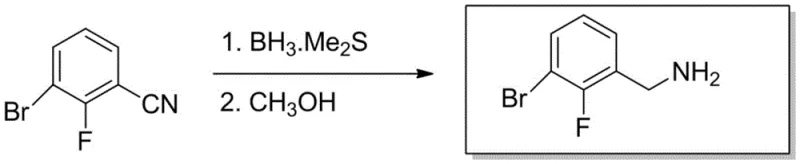
Impurity control is another critical aspect where this mechanism excels, particularly through the innovative quenching and workup procedure. After the reduction is complete, the reaction mixture contains residual boron species that must be removed to achieve high purity. The patent describes a specific quenching step where methanol is added dropwise until no more hydrogen gas is evolved. This step serves a dual purpose: it safely decomposes any excess borane and converts the amine-borane complex into the free amine and volatile borate esters. Following solvent removal, the concentrate is dissolved in methanol and refluxed for 2 to 5 hours. This reflux step is a distinctive feature of the invention, designed to ensure the complete hydrolysis of any stubborn boron-nitrogen bonds that might persist after the initial quench. Finally, purification via an alumina column using ethyl acetate as the eluent effectively separates the target amine from any remaining organic impurities or inorganic salts. This rigorous purification protocol is what allows the process to consistently achieve purity levels of ≥99%, meeting the stringent specifications required for pharmaceutical intermediates where trace impurities can impact drug safety and efficacy.
How to Synthesize 2-Fluoro-3-Bromo-Benzylamine Efficiently
Implementing this synthesis route in a production environment requires adherence to the specific parameters outlined in the patent to ensure optimal results. The process begins with the dissolution of the nitrile starting material in a suitable organic solvent such as tetrahydrofuran (THF) or dichloromethane (DCM), with concentration levels optimized between 0.05 and 0.5 g/mL to balance reaction kinetics and mixing efficiency. The subsequent addition of the borane reagent must be performed slowly under inert gas protection to maintain the thermal stability of the system. Once the reaction period is concluded, the careful addition of methanol is paramount for safety, as it releases hydrogen gas that must be vented appropriately. The final isolation involves standard solvent evaporation and chromatographic techniques that are well-established in industrial settings. For a comprehensive breakdown of the standardized operating procedures and safety checks required for this synthesis, please refer to the detailed guide below.
- Dissolve 2-fluoro-3-bromobenzonitrile in an organic solvent such as THF or DCM and cool the solution to between -5°C and 5°C under inert gas protection.
- Slowly add BH3.Me2S solution dropwise while maintaining low temperature, then allow the reaction to proceed at room temperature for 8 to 15 hours.
- Quench the reaction carefully with methanol until hydrogen evolution ceases, concentrate the mixture, and purify the residue via alumina column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis method offers profound benefits for procurement managers and supply chain leaders looking to optimize their sourcing strategies for fine chemical intermediates. The primary advantage lies in the significant reduction of raw material costs, driven by the use of 2-fluoro-3-bromobenzonitrile, which is generally more affordable and accessible than the benzoyl chloride or aldehyde precursors required by older methods. By eliminating the need for expensive transition metal catalysts and high-pressure hydrogenation equipment, the capital expenditure required for setting up production lines is drastically lowered. This cost efficiency is passed down through the supply chain, allowing for more competitive pricing structures without compromising on quality. Furthermore, the simplified operational workflow reduces the labor hours and energy consumption associated with multi-step syntheses, contributing to a leaner and more agile manufacturing process. These factors combined create a robust value proposition for buyers seeking reliable partners who can deliver high-quality intermediates at a sustainable cost point.
- Cost Reduction in Manufacturing: The elimination of costly metal catalysts and the reduction in reaction steps directly translate to lower operational expenditures. By avoiding the use of precious metals that require complex recovery and recycling processes, the overall cost of goods sold is significantly optimized. Additionally, the high yield reported in the patent examples, often exceeding 93%, means that less raw material is wasted per kilogram of finished product, further enhancing the economic efficiency of the process. This lean approach to synthesis ensures that resources are utilized maximally, providing a distinct financial advantage in a margin-sensitive market.
- Enhanced Supply Chain Reliability: The reliance on stable, liquid reagents like BH3.Me2S instead of gaseous hydrogen or unstable intermediates improves the predictability of the supply chain. Gaseous reactants often require specialized storage and transport infrastructure, which can be a bottleneck in global logistics. In contrast, the reagents used in this method are standard chemicals available from multiple suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate room temperature stirring for the majority of the cycle, also means that production schedules are less susceptible to disruptions caused by equipment failure or utility fluctuations, ensuring consistent on-time delivery for clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of extreme pressure or temperature requirements. The reaction can be safely conducted in standard glass-lined or stainless steel reactors without the need for specialized high-pressure vessels. Moreover, the avoidance of heavy metal catalysts simplifies waste treatment and disposal, aligning with increasingly strict environmental regulations regarding heavy metal residues in pharmaceutical waste streams. The use of common organic solvents like THF and ethyl acetate, which are easily recoverable and recyclable, further supports sustainability goals, making this method an environmentally responsible choice for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-fluoro-3-bromo-benzylamine using this advanced methodology. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their own development pipelines. For more specific technical data or custom synthesis requests, our team is prepared to provide detailed documentation and support.
Q: What are the primary advantages of using BH3.Me2S over traditional reducing agents for this synthesis?
A: Using BH3.Me2S avoids the need for hazardous gaseous reactants and expensive metal catalysts, resulting in higher yields exceeding 93% and simplifying the purification process significantly.
Q: How does this patented method improve impurity control compared to prior art?
A: The method employs a specific methanol reflux step post-reaction to completely quench boron complexes, followed by alumina chromatography, ensuring product purity levels consistently above 99%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process operates at mild temperatures and avoids high-pressure hydrogenation, making it inherently safer and more scalable for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-3-Bromo-Benzylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development and commercialization. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-fluoro-3-bromo-benzylamine meets the highest industry standards. We understand that reliability is just as important as quality, which is why our supply chain is designed to be resilient and responsive to the dynamic demands of the global market. Partnering with us means gaining access to a team of dedicated chemists and engineers committed to optimizing your production processes.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new developments, we are here to assist. Our goal is to build long-term relationships based on trust, transparency, and technical excellence. Reach out to us today to learn more about our capabilities and how we can help you accelerate your path to market with confidence and efficiency.
