Advanced Purification Technology for Meldonium Dihydrate Manufacturing and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with operational efficiency, particularly for cardiovascular therapeutics like Meldonium. Patent CN109369447B introduces a transformative methodology for the preparation of 3-(2,2,2-trimethylhydrazinium) propionate dihydrate, addressing critical bottlenecks in the final purification stages. Traditionally, the isolation of this zwitterionic compound has been plagued by the formation of stubborn emulsions and complex inorganic double salts when using conventional gaseous acidification agents. This new intellectual property discloses a strategic shift towards using specific ammonium salts as acidifying agents, which fundamentally alters the precipitation dynamics of inorganic byproducts. By integrating this novel acidification step immediately following alkaline hydrolysis, manufacturers can bypass the cumbersome electrodialysis separation processes that have historically inflated production costs and extended lead times. For global supply chain stakeholders, this represents a pivotal opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering consistent quality without the volatility associated with complex gas-handling infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Meldonium dihydrate has relied heavily on the neutralization of hydrolyzed reaction mixtures using acidic gases such as carbon dioxide or sulfur dioxide. While theoretically sound, this approach presents severe practical deficiencies when scaled to commercial volumes. The introduction of these gases into the alkaline hydrolysate often results in the uncontrolled formation of mixed salt systems, specifically potassium carbonate or bicarbonate complexes, which act as potent emulsifiers. These emulsions create a milky, opaque mixture that resists standard filtration techniques, trapping the valuable active pharmaceutical ingredient within a chaotic matrix of inorganic salts and solvent. Furthermore, the precise control of pH required to precipitate the target molecule without co-precipitating impurities is exceptionally difficult to maintain during gas sparging, leading to inconsistent batch quality. In colder climates, the physical properties of the gas cylinders themselves can further retard the acidification rate, extending processing times to upwards of ten hours per batch and creating significant bottlenecks in winter production schedules.
The Novel Approach
The innovative process detailed in the patent data circumvents these physicochemical hurdles by substituting gaseous acids with solid ammonium salts, such as ammonium nitrate, ammonium bicarbonate, or ammonium sulfate. This substitution triggers a clean neutralization reaction where the ammonium cation reacts with residual hydroxide ions to release ammonia gas and water, effectively lowering the pH without introducing new anionic species that could form insoluble double salts. The result is a reaction system that remains clear and free of the persistent emulsions characteristic of the old method. Consequently, the inorganic salts generated during hydrolysis can be removed through simple, rapid filtration rather than requiring multi-stage electrodialysis or complex solvent extractions. This streamlined workflow not only simplifies the equipment requirements—eliminating the need for specialized gas scrubbing and electrodialysis stacks—but also ensures that the final crystalline product meets stringent pharmacopeia standards for sulfated ash content, typically achieving levels below 0.1 percent with minimal effort.
Mechanistic Insights into Ammonium Salt-Mediated Acidification
To fully appreciate the technical superiority of this route, one must examine the molecular interactions occurring during the critical work-up phase. The process begins with the alkaline hydrolysis of methyl 3-(2,2,2-trimethylhydrazinium) propionate, yielding the free acid form of Meldonium in a basic solution. In conventional methods, adding CO2 generates carbonate ions that compete with the Meldonium zwitterion for cationic partners, leading to the aforementioned double salt precipitation. However, the novel approach utilizes the unique acid-base properties of ammonium salts. When an ammonium salt (NH4)nY is introduced to the hydrolysate, the ammonium ion acts as a weak acid donor. It reacts stoichiometrically with the excess hydroxide ions from the hydrolysis step to form water and volatile ammonia, as depicted in the reaction scheme below. This mechanism ensures that the pH drops precisely to the isoelectric point of the target molecule without flooding the system with competing anions like carbonate or sulfite.
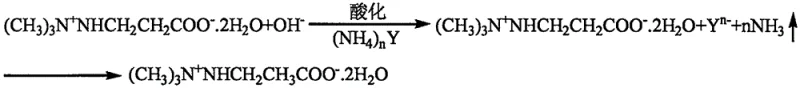
Furthermore, the presence of specific mixed solutions, such as aqueous sodium ethoxide or halide salts, prior to acidification plays a crucial role in breaking any potential micro-emulsions that might begin to form. These additives modify the surface tension and solubility parameters of the medium, forcing the inorganic salts to aggregate into large, filterable granules rather than colloidal suspensions. This control over the solid-liquid interface is the key to achieving the high purity levels required for cardiovascular drugs. By avoiding the entrapment of product within inorganic salt lattices, the overall yield is maximized, and the need for recrystallization is minimized. For R&D directors evaluating technology transfer, this mechanistic clarity offers a predictable and robust pathway that reduces the risk of batch failure due to unpredictable emulsion behavior.
How to Synthesize Meldonium Dihydrate Efficiently
Implementing this improved synthesis protocol requires strict adherence to temperature controls and reagent ratios to maximize the benefits of the ammonium salt acidification strategy. The process is designed to be operationally simple, relying on standard unit operations found in most multipurpose pharmaceutical plants. Operators must first ensure the complete hydrolysis of the methyl ester intermediate under mild conditions to prevent degradation of the hydrazine moiety. Following the removal of initial inorganic precipitates, the timing of the ammonium salt addition is critical; it must be dosed into the combined mother liquors to ensure uniform pH adjustment throughout the vessel. Detailed standardized operating procedures regarding the specific molar ratios of hydroxide to ammonium salt, as well as the optimal temperature ranges for crystallization, are essential for reproducibility. The following guide outlines the critical operational steps derived from the patent examples to assist technical teams in replicating this high-efficiency route.
- Perform alkaline hydrolysis of the methyl ester intermediate using potassium or sodium hydroxide in ethanol at controlled low temperatures (18-20°C).
- Filter off inorganic precipitates formed during hydrolysis and wash with ethanol to recover product trapped in the cake.
- Acidify the combined mother liquor using a solid ammonium salt (e.g., ammonium nitrate or bicarbonate) instead of gaseous acids to precipitate inorganic salts cleanly.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel manufacturing process translates directly into enhanced operational resilience and cost structure optimization. The elimination of electrodialysis, a capital-intensive and energy-consuming separation technology, significantly reduces the utility load per kilogram of produced API. Moreover, the removal of gaseous acid handling systems mitigates safety risks associated with high-pressure gas cylinders and toxic gas leaks, thereby lowering insurance and compliance overheads. The simplified workflow, which replaces a multi-hour gas sparging operation with a rapid solid addition and filtration sequence, drastically shortens the manufacturing cycle time. This acceleration allows for increased throughput within existing facility footprints, effectively expanding capacity without the need for new construction. Additionally, the robustness of the process against environmental variables, such as ambient temperature fluctuations that affect gas flow rates, ensures consistent delivery schedules year-round, safeguarding against the supply disruptions common in older, less stable technologies.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this new protocol is the complete removal of the electrodialysis purification step. Electrodialysis requires expensive membrane stacks, high electricity consumption, and frequent maintenance, all of which contribute substantially to the cost of goods sold. By achieving high purity through simple crystallization and filtration, the process eliminates these variable costs entirely. Furthermore, the use of commodity ammonium salts as acidifying agents is generally more cost-effective and logistically simpler than managing the supply and storage of industrial gases like sulfur dioxide. The reduction in processing time also lowers labor costs and increases asset utilization rates, allowing the manufacturing suite to produce more batches over the same period. These cumulative savings create a significant margin advantage that can be passed down the supply chain or reinvested into further process optimization.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of purification steps that are prone to failure or delay. The conventional gas acidification method is particularly vulnerable to equipment fouling from emulsions and the logistical challenges of gas delivery, especially in remote manufacturing locations. The new ammonium salt-based method relies on solid reagents that are easy to store, transport, and dose with high precision. This shift reduces the dependency on specialized infrastructure and minimizes the risk of batch hold-ups caused by filtration failures. Consequently, suppliers adopting this technology can offer more reliable lead times and maintain higher safety stock levels with confidence. The ability to consistently meet tight specifications for sulfated ash and content without rework ensures that downstream formulation partners receive materials that are ready for immediate use, smoothing the entire production pipeline.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies minor inefficiencies, but this novel route is inherently scalable due to its reliance on fundamental unit operations like mixing, filtration, and drying. The absence of complex emulsion management means that scaling from pilot plant to commercial tonnage does not require disproportionate increases in separation equipment. From an environmental perspective, the process generates less hazardous waste; the byproduct ammonia can be easily captured and scrubbed, and the avoidance of sulfur dioxide eliminates the generation of sulfite-rich wastewater streams that require extensive treatment. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site. Companies prioritizing sustainability in their vendor selection criteria will find this process highly attractive as it demonstrates a commitment to cleaner, safer, and more efficient chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved Meldonium synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is vital for technical teams assessing the feasibility of technology transfer and for procurement professionals evaluating supplier capabilities. The answers highlight the specific advantages of the ammonium salt method over traditional gas acidification, focusing on purity profiles, operational simplicity, and regulatory compliance. Stakeholders are encouraged to review these points to gain a comprehensive understanding of how this innovation impacts the overall value proposition of the supply chain.
Q: Why is ammonium salt acidification superior to CO2 gas acidification for Meldonium?
A: Ammonium salt acidification prevents the formation of complex double salts and stable emulsions that typically occur with carbonate/bicarbonate systems, thereby eliminating the need for energy-intensive electrodialysis purification steps.
Q: What is the achievable purity level using this improved process?
A: The process consistently yields Meldonium Dihydrate with a content greater than 99.5% and a sulfated ash content of less than 0.1%, meeting strict pharmacopeia standards without complex post-treatment.
Q: Does this method improve scalability for industrial production?
A: Yes, by replacing slow gas sparging (which can take 8-10 hours) with rapid solid addition and simple filtration, the cycle time is drastically reduced, making large-scale commercial manufacturing significantly more efficient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meldonium Dihydrate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN109369447B is essential for maintaining competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel acidification method are fully realized in large-scale manufacturing. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify that every batch of Meldonium Dihydrate meets the <0.1% sulfated ash requirement and >99.5% content standard. Our commitment to process excellence means we can offer a stable supply of high-quality cardiovascular intermediates that support your drug development and commercialization timelines without the risks associated with outdated purification technologies.
We invite you to collaborate with us to optimize your sourcing strategy for this critical therapeutic agent. Our experts are prepared to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this emulsion-free production method. We encourage you to contact our technical procurement team to request specific COA data from our recent batches and to discuss route feasibility assessments tailored to your specific volume requirements. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technologically advanced and resilient against the common pitfalls of traditional chemical synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
