Advanced Cucurbituril Cyclodextrin Dual-Host Compounds for Scalable Pharmaceutical and Catalytic Applications
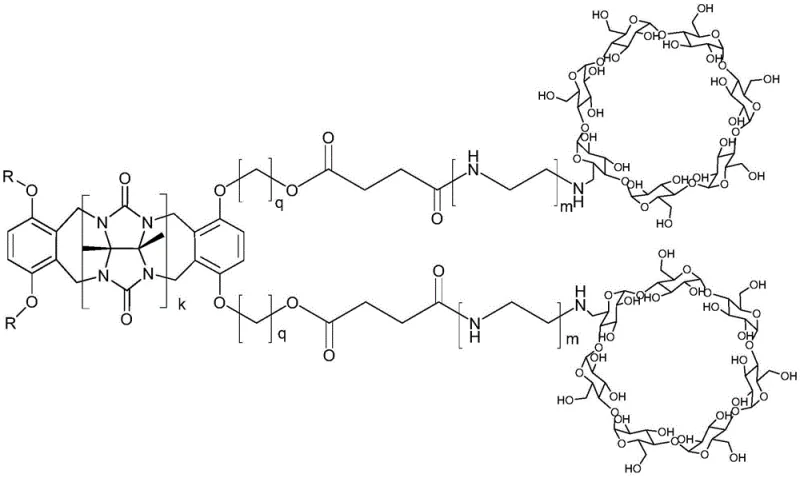
The landscape of supramolecular chemistry is undergoing a significant transformation with the introduction of advanced dual-host systems that overcome the limitations of traditional single-cavity molecules. As detailed in patent CN112125985A, a novel class of ring-opening cucurbituril cyclodextrin dual-host compounds has been developed, representing a breakthrough in molecular recognition and carrier technology. These sophisticated macromolecules are engineered by linking halogen-modified ring-opening cucurbituril with carboxylated amino-cyclodextrin through robust ester bonds, creating a linear architecture that possesses two distinct types of cavities within a single molecular framework. This unique structural arrangement allows the compound to exhibit the beneficial properties of both cyclodextrin, known for its biocompatibility and solubility enhancement, and open-ring cucurbituril, recognized for its ability to bind hydrophobic cations and facilitate pi-pi interactions. The integration of these two diverse supramolecular hosts into one entity creates a synergistic effect that significantly broadens the scope of potential applications in pharmaceuticals, fine chemicals, and catalysis.
For procurement managers and supply chain directors, the implications of this technology extend far beyond mere academic interest, offering tangible pathways for cost reduction in pharmaceutical intermediates manufacturing. The synthesis route described eliminates the need for expensive transition metal catalysts often required in complex coupling reactions, relying instead on straightforward nucleophilic substitution and esterification processes using readily available alkali bases and organic solvents. This simplification of the chemical process directly translates to a more robust supply chain, as the raw materials such as sodium carbonate, potassium carbonate, and common solvents like tetrahydrofuran and acetone are globally sourced commodities with stable pricing and availability. Furthermore, the reaction conditions are remarkably mild, operating effectively within a temperature range of 30°C to 60°C, which reduces energy consumption and minimizes the thermal stress on equipment, thereby lowering the total cost of ownership for production facilities. The ability to produce high-purity materials through simple precipitation and recrystallization steps also reduces the burden on waste treatment systems, aligning with increasingly stringent environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional supramolecular carriers typically rely on single-host molecules such as unmodified cyclodextrins or standard cucurbiturils, which inherently possess limitations regarding their binding specificity and loading capacity. Single-cavity hosts often struggle to simultaneously accommodate guest molecules with differing physicochemical properties, leading to suboptimal encapsulation efficiency and restricted utility in complex formulation environments. For instance, while cyclodextrins are excellent for improving the water solubility of hydrophobic drugs, they lack the specific cation-binding capabilities required for certain catalytic processes or ion-transport applications. Conversely, standard cucurbiturils may offer strong binding but often suffer from poor solubility in aqueous media, necessitating complex derivatization steps that increase production costs and reduce overall yield. Additionally, conventional synthesis methods for modifying these macrocycles frequently involve harsh reaction conditions, toxic reagents, or multi-step protection and deprotection strategies that generate significant chemical waste and complicate the scale-up process for commercial manufacturing.
The Novel Approach
The novel approach presented in this patent fundamentally addresses these deficiencies by constructing a hybrid molecular architecture that combines the strengths of two distinct supramolecular families into a single, linear dual-host system. By covalently linking an open-ring cucurbituril unit with a cyclodextrin unit via a flexible ester linkage, the resulting compound creates a continuous binding environment that can interact with a wider variety of guest molecules through multiple interaction mechanisms simultaneously. This dual-cavity design not only enhances the overall loading capacity for active pharmaceutical ingredients or catalytic substrates but also improves the stability of the host-guest complexes formed. The synthetic strategy employs a direct condensation reaction between functionalized precursors, bypassing the need for intricate multi-step syntheses and allowing for greater control over the final molecular weight and polydispersity. This streamlined methodology ensures that the final product maintains high purity levels suitable for sensitive applications in the food and pharmaceutical industries while remaining economically viable for large-scale industrial production.
Mechanistic Insights into Esterification-Linked Dual-Host Assembly
The core chemical mechanism driving the formation of these dual-host compounds is a nucleophilic substitution reaction followed by esterification, which serves as the critical linkage between the two macrocyclic domains. In this process, the halogen-modified ring-opening cucurbituril acts as the electrophile, where the halogen atom (such as bromine or chlorine) attached to the aromatic rim is displaced by the nucleophilic oxygen of the carboxylated cyclodextrin derivative. This reaction is facilitated by the presence of a base, such as sodium carbonate or potassium bicarbonate, which deprotonates the carboxylic acid groups on the cyclodextrin, increasing their nucleophilicity and driving the reaction forward. The choice of solvent system, typically a mixture of polar aprotic solvents like dimethyl sulfoxide or tetrahydrofuran with water-miscible alcohols, is crucial for ensuring that both the hydrophobic cucurbituril derivative and the hydrophilic cyclodextrin derivative remain in solution long enough to react effectively. The resulting ester bond is chemically stable under physiological conditions yet can be tuned for biodegradability depending on the specific chain lengths and substituents used, offering versatility for different end-use requirements.
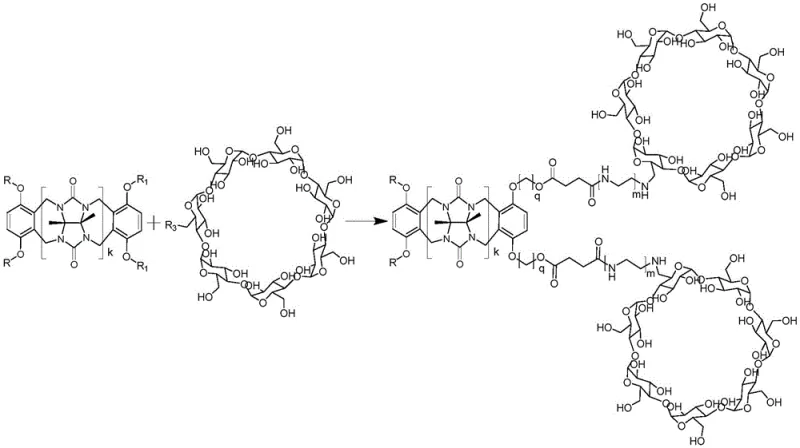
Impurity control is a paramount concern in the synthesis of such complex supramolecular architectures, particularly when intended for pharmaceutical or food-grade applications. The reaction design inherently minimizes the formation of side products by utilizing stoichiometric ratios of reactants that favor the formation of the desired mono- or di-substituted products over cross-linked polymers. For example, maintaining a molar ratio of halogen-modified cucurbituril to carboxyl-modified cyclodextrin between 5:6 and 5:11 helps to prevent excessive cross-linking which could lead to insoluble aggregates. Post-reaction purification is achieved through a selective precipitation strategy where the reaction mixture is poured into a non-solvent like acetone or ethanol, causing the high molecular weight dual-host compound to precipitate while smaller unreacted starting materials and inorganic salts remain in the supernatant. Subsequent recrystallization from water and organic solvent mixtures further refines the product, removing any trace impurities and ensuring that the final material meets stringent purity specifications required by regulatory bodies for use in human health applications.
How to Synthesize Ring-Opening Cucurbituril Cyclodextrin Dual-Host Compound Efficiently
The synthesis of these advanced dual-host compounds follows a reproducible protocol that balances reaction efficiency with ease of operation, making it accessible for both laboratory research and pilot plant operations. The process begins with the precise weighing and mixing of the functionalized macrocyclic precursors in a reactor equipped with temperature control and stirring capabilities to ensure homogeneous reaction conditions throughout the vessel. Operators must carefully monitor the reaction temperature, keeping it within the optimal window of 30°C to 60°C to maximize conversion rates without degrading the sensitive macrocyclic structures. Following the reaction period, the workup procedure involves a series of phase separations and crystallizations that are designed to recover the maximum amount of product while minimizing solvent usage and waste generation. Detailed standardized synthesis steps see the guide below.
- Preparation of halogen-modified ring-opening cucurbituril and carboxyl-modified cyclodextrin precursors with specific functional groups.
- Esterification reaction conducted in a mixed solution of alkali and organic solvent at controlled temperatures between 30°C and 60°C.
- Precipitation using anti-solvents, followed by filtration, water dissolution, and recrystallization to isolate the high-purity dual-host compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this dual-host compound technology offers substantial benefits that align with the goals of cost optimization and supply chain resilience. The elimination of precious metal catalysts from the synthesis route removes a significant variable cost driver and mitigates the risk associated with the price volatility of rare earth elements or noble metals. This shift towards base-metal or metal-free catalysis not only reduces the direct material costs but also simplifies the downstream processing requirements, as there is no need for expensive and time-consuming metal scavenging steps to meet residual metal limits in the final product. Furthermore, the use of commodity chemicals as solvents and reagents ensures that the supply chain is not dependent on niche suppliers, thereby reducing the risk of disruption due to geopolitical issues or single-source bottlenecks. The robustness of the synthesis process also means that production yields are consistent and predictable, allowing for more accurate inventory planning and reduced safety stock requirements.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway significantly lowers operational expenditures by reducing the number of unit operations required to achieve high purity. By avoiding complex protection group chemistry and utilizing direct esterification, the process saves on reagent costs and reduces the volume of hazardous waste that requires disposal. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and reduced utility bills. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials, improving the overall atom economy of the process and maximizing the return on investment for raw material procurement.
- Enhanced Supply Chain Reliability: The reliance on widely available chemical feedstocks ensures a stable and continuous supply of raw materials, which is critical for maintaining uninterrupted production schedules. Since the synthesis does not require specialized or hard-to-source reagents, procurement teams can leverage multiple vendors to negotiate better pricing and service terms. The scalability of the process means that production volumes can be ramped up quickly to meet surges in demand without the need for significant capital investment in new equipment or infrastructure. This flexibility allows manufacturers to respond agilely to market dynamics and secure long-term contracts with confidence, knowing that the supply of this key intermediate can be sustained over time.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction parameters that are easily transferable from laboratory flasks to industrial reactors. The use of aqueous workups and recyclable organic solvents aligns with green chemistry principles, facilitating compliance with environmental regulations and reducing the burden on waste treatment facilities. The solid product is easily isolated by filtration, which is a unit operation that scales linearly and efficiently, avoiding the complexities associated with distillation or chromatography on a large scale. This environmental compatibility not only reduces regulatory risk but also enhances the brand reputation of companies adopting this technology as leaders in sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this dual-host technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these aspects is crucial for assessing the feasibility of integrating this material into existing product lines or development pipelines.
Q: What distinguishes this dual-host compound from traditional single-host systems?
A: This compound integrates two distinct cavities: the hydrophobic cone of cyclodextrin and the cation-binding C-shaped cavity of open-ring cucurbituril. This linear arrangement allows for simultaneous recognition of different guest molecules, enhancing loading capacity and catalytic versatility compared to single-component hosts.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the preparation method utilizes mild reaction conditions (30°C to 60°C) and common organic solvents like tetrahydrofuran and acetone. The straightforward workup involving precipitation and recrystallization avoids complex purification steps, making it highly amenable to commercial scale-up.
Q: What are the primary commercial applications for this supramolecular material?
A: Due to its excellent molecular recognition and solubility properties, it is ideal for pharmaceutical drug delivery systems, supramolecular catalysis, and as a functional additive in the food and fragrance industries where encapsulation of active ingredients is required.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ring-Opening Cucurbituril Cyclodextrin Dual-Host Compound Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM is uniquely positioned to support the commercialization of this cutting-edge supramolecular technology, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature controls required for the synthesis of these sensitive dual-host compounds, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. We understand the critical nature of supply continuity for our clients and have established robust procurement channels for all necessary precursors to guarantee uninterrupted delivery schedules. Our technical team works closely with partners to optimize the process for their specific volume requirements, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless and efficient.
We invite you to engage with our technical procurement team to discuss how this innovative dual-host compound can enhance your product portfolio and drive value through improved performance and cost efficiency. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this superior material for your specific application needs. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements, allowing you to make informed decisions with confidence. Let us collaborate to bring the next generation of supramolecular materials to market, combining scientific excellence with commercial viability.
