Revolutionizing Polysubstituted Thiazole Production: A Cost-Effective FeCl3 Catalytic Route for Global Supply Chains
Revolutionizing Polysubstituted Thiazole Production: A Cost-Effective FeCl3 Catalytic Route for Global Supply Chains
The synthesis of polysubstituted thiazole compounds represents a critical challenge in modern organic chemistry, particularly given their ubiquity as core structural units in bioactive molecules ranging from antimalarial agents to anti-cancer therapeutics. Recent advancements documented in patent CN108358865B introduce a transformative methodology that leverages inexpensive enamines and elemental sulfur under iron catalysis to construct these valuable heterocycles. This innovation addresses long-standing inefficiencies in thiazole manufacturing by replacing complex, multi-step precursor preparations with a direct, one-pot oxidative cyclization strategy. For global procurement and R&D teams, this development signals a pivotal shift towards more sustainable and economically viable production models for high-value pharmaceutical intermediates. The protocol utilizes anhydrous ferric chloride as a robust catalyst within a dimethyl sulfoxide solvent system, operating effectively under ambient air conditions without the need for rigorous inert atmosphere controls. By streamlining the synthetic pathway and utilizing commodity chemicals like sulfur, this technology offers a compelling value proposition for scaling up the production of complex thiazole derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the thiazole ring has relied heavily on methodologies that involve expensive, hazardous, or difficult-to-prepare starting materials, creating significant bottlenecks in supply chain continuity and cost management. Traditional approaches often utilize alpha-p-toluenesulfonyloxy ketones or alkynyl iodonium salts coupled with thiourea, processes that require multi-step synthesis of the precursors themselves, thereby inflating the overall cost of goods and extending lead times. Furthermore, methods employing Lawesson's reagent, while effective for thionation, introduce severe handling challenges due to the reagent's toxicity, unpleasant odor, and high cost, making it unsuitable for large-scale industrial applications where worker safety and environmental compliance are paramount. Other existing routes may depend on precious metal catalysts or harsh reaction conditions that demand specialized equipment and extensive purification steps to remove trace metal impurities, which is a critical quality attribute for pharmaceutical intermediates. These legacy techniques often suffer from limited substrate scope and poor atom economy, generating substantial chemical waste that complicates disposal and increases the environmental footprint of the manufacturing process.
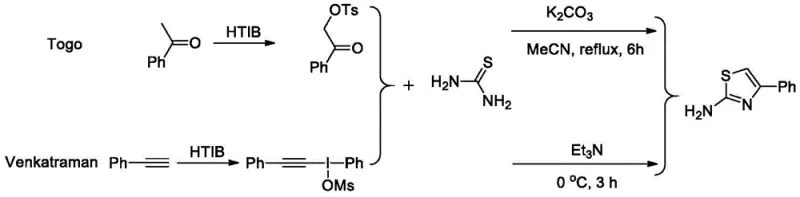
The Novel Approach
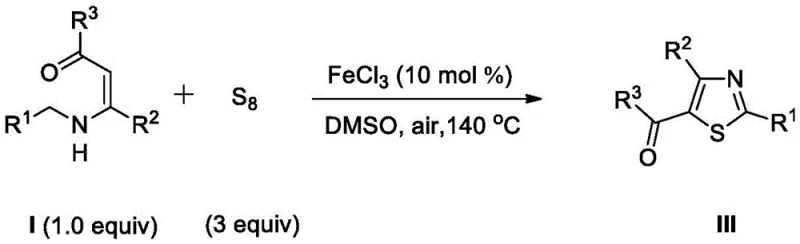
In stark contrast to these cumbersome legacy protocols, the novel methodology disclosed in the patent utilizes readily available enamines and elemental sulfur as the primary building blocks, fundamentally simplifying the retrosynthetic analysis and material sourcing strategy. This approach eliminates the need for pre-functionalized sulfur donors or expensive thionating agents, relying instead on the direct insertion of sulfur atoms from cheap S8 powder into the organic framework. The reaction proceeds through an efficient oxidative cyclization mechanism catalyzed by iron(III) chloride, a base metal catalyst that is not only abundant and low-cost but also avoids the regulatory scrutiny associated with heavy metal residues in final drug substances. By conducting the reaction in DMSO at elevated temperatures under air, the process achieves high conversion rates and excellent yields without the necessity for glovebox techniques or nitrogen purging, drastically reducing operational complexity. This streamlined workflow translates directly into reduced manufacturing cycles and lower capital expenditure requirements, positioning this technology as a superior alternative for the commercial scale-up of complex polysubstituted thiazoles.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between the iron catalyst, the enamine substrate, and elemental sulfur, facilitating a sequence of oxidation and cyclization events that construct the thiazole core with high precision. Initially, the anhydrous ferric chloride acts as a Lewis acid and oxidant, promoting the activation of the enamine double bond and facilitating its oxidation to an imine intermediate in the presence of air. This in situ generation of the reactive imine species is crucial, as it serves as the electrophilic center for the subsequent nucleophilic attack by the sulfur species derived from S8. The reaction environment in DMSO likely stabilizes polar intermediates and assists in the solubilization of elemental sulfur, ensuring homogeneous reaction kinetics throughout the process. Following the sulfur attack, a series of intramolecular cyclization and deprotonation steps occur, driven by the thermodynamic stability of the aromatic thiazole ring system. The iron catalyst plays a dual role in both initiating the oxidation and potentially mediating the final aromatization step, ensuring that the reaction proceeds to completion with minimal formation of partially cyclized byproducts.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of side products commonly associated with harsher thionation reagents. The use of elemental sulfur reduces the risk of phosphorus-containing impurities that are typical of Lawesson's reagent pathways, simplifying the downstream purification profile. Furthermore, the oxidative nature of the reaction ensures that any reduced sulfur species are converted or removed during the workup, leading to a cleaner crude product mixture. The robustness of the FeCl3 catalyst against air and moisture means that the reaction is less prone to stalling due to environmental leaks, a common issue in sensitive transition metal catalysis. This reliability ensures consistent batch-to-batch quality, a critical factor for maintaining stringent purity specifications required by regulatory bodies for pharmaceutical intermediates. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters such as temperature and catalyst loading to optimize yield and minimize waste generation.
How to Synthesize Polysubstituted Thiazoles Efficiently
The practical implementation of this synthesis route is designed for ease of execution in standard laboratory and pilot plant settings, requiring only common glassware and heating equipment. The protocol begins with the precise weighing of the enamine substrate and elemental sulfur, followed by their suspension in anhydrous DMSO to ensure proper mixing before catalyst addition. The addition of the iron catalyst initiates the reaction, which is then maintained at a controlled temperature of 140 °C to drive the oxidative cyclization to completion over a period of approximately 7 hours. Monitoring the reaction progress via thin-layer chromatography allows operators to determine the exact endpoint, preventing over-reaction or degradation of the product. Upon completion, the workup procedure involves standard liquid-liquid extraction techniques using ethyl acetate and brine, followed by drying and solvent removal, yielding a crude product that can be purified via standard silica gel chromatography to afford the target thiazole in high purity.
- Charge a reactor with enamine substrate (Compound I), elemental sulfur (S8), and dimethyl sulfoxide (DMSO) solvent.
- Add anhydrous ferric chloride (FeCl3) catalyst (10 mol%) to the mixture under air atmosphere.
- Heat the reaction mixture to 140 °C in an oil bath for approximately 7 hours until TLC indicates completion.
- Perform workup by extraction with ethyl acetate and saturated brine, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this FeCl3-catalyzed sulfur insertion technology presents a multitude of strategic benefits that directly impact the bottom line and operational resilience. The primary driver of value is the drastic reduction in raw material costs, achieved by substituting expensive, specialized reagents with commodity chemicals like elemental sulfur and iron chloride, which are globally available and price-stable. This shift away from proprietary or hazardous reagents also mitigates supply chain risks associated with vendor lock-in or regulatory restrictions on specific chemical classes, ensuring a more secure and continuous flow of materials for production. Additionally, the simplified operational requirements, such as the ability to run reactions under air rather than inert gas, reduce the utility consumption and infrastructure maintenance costs associated with specialized reactor systems. These factors combine to create a manufacturing process that is not only cheaper per kilogram but also more agile and responsive to fluctuating market demands.
- Cost Reduction in Manufacturing: The elimination of expensive thionating agents like Lawesson's reagent and complex precursors results in substantial cost savings per batch, while the use of a base metal catalyst avoids the high expense and recovery costs associated with precious metals. The simplified workup and purification process further reduces solvent usage and labor hours, contributing to a leaner overall cost structure for thiazole intermediate production. By lowering the barrier to entry for synthesizing these complex scaffolds, manufacturers can offer more competitive pricing to downstream pharmaceutical clients without compromising on quality margins.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as enamines and sulfur powder ensures that production schedules are not disrupted by shortages of niche reagents, providing a stable foundation for long-term supply agreements. The robustness of the reaction conditions means that manufacturing can be easily transferred between different facilities or geographic regions without the need for highly specialized equipment, enhancing the flexibility of the global supply network. This reliability is crucial for meeting the just-in-time delivery expectations of major pharmaceutical partners who require consistent availability of key intermediates for their own drug synthesis pipelines.
- Scalability and Environmental Compliance: The absence of toxic phosphorus byproducts and the use of a non-precious metal catalyst simplify waste treatment protocols, making it easier to comply with increasingly stringent environmental regulations regarding heavy metal discharge and hazardous waste disposal. The reaction's tolerance to air and its operation in a high-boiling solvent like DMSO facilitate safe scale-up from gram to ton quantities, as the thermal profile is manageable and does not require cryogenic conditions. This scalability ensures that the technology can support commercial volumes needed for blockbuster drug production while maintaining a smaller environmental footprint compared to traditional thiazole synthesis methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel thiazole synthesis technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these details is essential for assessing the feasibility of integrating this route into existing manufacturing workflows and for optimizing the economic potential of the new methodology.
Q: What are the primary advantages of using elemental sulfur in this thiazole synthesis?
A: Elemental sulfur serves as a cheap, abundant, and safe sulfur atom donor, eliminating the need for expensive or toxic thionating reagents like Lawesson's reagent, thereby significantly reducing raw material costs.
Q: Is the FeCl3 catalytic system sensitive to air or moisture?
A: No, the reaction is notably insensitive to air and proceeds efficiently under aerobic conditions, which simplifies operational requirements and removes the need for inert gas protection systems.
Q: What is the substrate scope for this synthetic methodology?
A: The method demonstrates excellent compatibility with various substituents, including aryl, alkyl, and alkoxy groups on the enamine backbone, allowing for the synthesis of diverse polysubstituted thiazole libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Thiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this FeCl3-catalyzed synthesis route for producing high-quality polysubstituted thiazoles, and we are fully equipped to leverage this technology for our global partners. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of large-scale API manufacturing with consistency and precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of thiazole intermediate meets the exacting standards required for pharmaceutical applications, minimizing the risk of downstream processing issues. Our commitment to technological advancement allows us to offer cost-effective solutions that do not compromise on the quality or safety of the final chemical product.
We invite forward-thinking pharmaceutical and agrochemical companies to collaborate with us to explore the full potential of this efficient synthesis platform for their specific pipeline needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project volume, along with specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in the production of complex heterocyclic intermediates.
