Scalable Production of Ammonium 3,5-Dinitroamino-1,2,4-Triazolate via Direct Nitration
The landscape of energetic material synthesis is undergoing a significant transformation driven by the need for safer, more efficient, and cost-effective manufacturing protocols. A pivotal development in this sector is detailed in patent CN114773281A, which discloses a novel preparation method for the ammonium salt of 3,5-dinitroamino-1,2,4-triazole, a high-energy compound with exceptional thermal stability and nitrogen content. This patent addresses the critical bottlenecks of traditional synthesis routes, which have historically been plagued by excessive step counts, hazardous operating conditions, and suboptimal yields. By leveraging a streamlined mixed-acid nitration system followed by a direct salt-forming reaction, this technology offers a robust pathway for producing high-purity energetic intermediates. For R&D directors and procurement strategists, understanding the nuances of this two-step process is essential for evaluating its potential to disrupt current supply chains and reduce the total cost of ownership for advanced energetic formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-dinitroamino-1,2,4-triazole derivatives has been a formidable challenge due to the complexity of introducing nitro groups onto the triazole ring without degrading the heterocyclic structure. As illustrated in the prior art, conventional pathways often involve a tedious five-step sequence that begins with the condensation of aminoguanidine or similar precursors, followed by cyclization, protection, nitration, and deprotection steps. This multi-stage approach not only consumes significant amounts of solvents and reagents but also introduces multiple points of failure where yield losses accumulate dramatically. Furthermore, the intermediate compounds in these long sequences often exhibit poor stability, necessitating stringent storage conditions and increasing the risk of safety incidents during scale-up. The cumulative effect of these inefficiencies is a final product with a prohibitively high cost basis and an impurity profile that is difficult to control, making it less attractive for large-scale industrial applications where consistency is paramount.
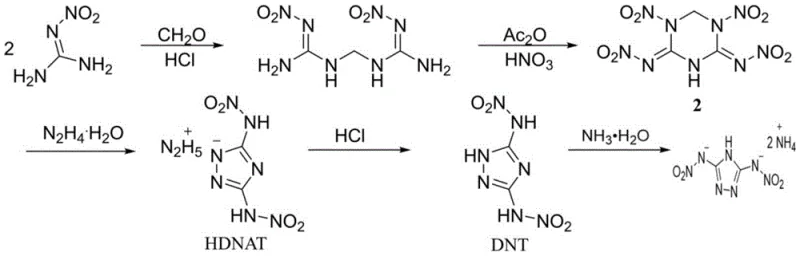
The Novel Approach
In stark contrast to the convoluted legacy methods, the invention described in CN114773281A introduces a remarkably concise two-step strategy that bypasses the need for protecting groups and intermediate isolations. The core innovation lies in the direct nitration of commercially available 3,5-diamino-1,2,4-triazole using a optimized mixture of nitric and sulfuric acids. This one-pot nitration effectively converts the amino groups into nitramino groups in a single operational unit, drastically simplifying the workflow. Following the nitration, the crude intermediate is extracted and immediately subjected to a neutralization reaction with an aqueous ammonium source. This telescoped approach not only accelerates the production timeline but also minimizes the exposure of unstable intermediates to harsh environments, thereby enhancing overall process safety. The result is a synthesis route that is not only chemically elegant but also industrially viable, offering a clear path toward commercial scalability.
Mechanistic Insights into Mixed Acid Nitration and Salt Formation
The chemical efficacy of this novel process is rooted in the precise control of electrophilic aromatic substitution dynamics within a highly acidic medium. The use of a mixed acid system, specifically comprising concentrated sulfuric acid and nitric acid in a volume ratio ranging from 1:4 to 3:4, creates a potent nitrating environment capable of generating the nitronium ion (NO2+) in sufficient concentration. When 3,5-diamino-1,2,4-triazole is introduced into this system at controlled low temperatures between -10°C and 10°C, the electron-rich amino groups undergo rapid nitration to form the 3,5-dinitroamino-1,2,4-triazole intermediate. The sulfuric acid acts as both a dehydrating agent and a catalyst, driving the equilibrium towards the formation of the nitronium species while stabilizing the transition state. Maintaining the temperature within this narrow window is critical; temperatures that are too high can lead to oxidative degradation of the triazole ring, while temperatures that are too low may result in incomplete conversion, both of which would compromise the purity of the final energetic material.
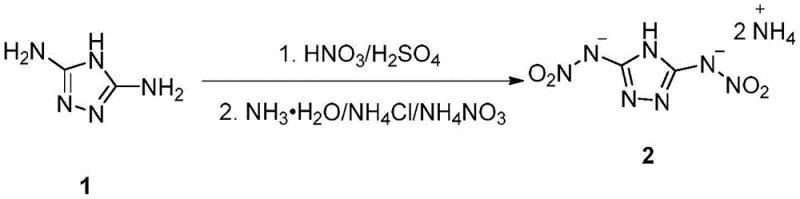
Following the nitration phase, the mechanism shifts to an acid-base neutralization and precipitation process. The reaction mixture is quenched with ice water to dilute the acid strength and precipitate the organic intermediate, which is then extracted into an organic phase such as ethyl acetate or dichloromethane. This extraction step serves a dual purpose: it isolates the desired dinitro compound from the bulk aqueous acid waste, and it provides a clean medium for the subsequent salt formation. Upon the slow addition of an ammonium source, such as ammonia water or ammonium chloride, the acidic protons on the nitramino groups are deprotonated, forming the stable ammonium salt. The low temperature maintained during this step (0°C to 10°C) ensures that the precipitation occurs in a controlled manner, yielding a crystalline product with high structural integrity and minimal inclusion of solvent impurities. This mechanistic understanding allows process engineers to fine-tune parameters like drip rates and stirring speeds to maximize crystal quality and filtration efficiency.
How to Synthesize Ammonium 3,5-Dinitroamino-1,2,4-Triazolate Efficiently
Implementing this synthesis protocol requires careful attention to the order of addition and thermal management to replicate the high yields reported in the patent examples. The process begins with the preparation of the nitrating agent, followed by the controlled addition of the triazole substrate, and concludes with the workup and salting-out procedure. Detailed operational parameters, including specific molar ratios and extraction frequencies, are critical for achieving the reported yields of over 80%. For laboratory and pilot plant teams looking to adopt this methodology, adhering to the standardized steps outlined below is essential for ensuring reproducibility and safety.
- Prepare a mixed acid system using concentrated sulfuric acid and nitric acid (volume ratio 1: 4 to 3:4) and cool to -10 to 10°C.
- Slowly add 3,5-diamino-1,2,4-triazole to the acid mixture under stirring, maintaining temperature control to ensure complete nitration to the dinitro intermediate.
- Quench the reaction with ice water, extract the intermediate with an organic solvent, and react with an ammonium source (e.g., ammonia water) at low temperature to precipitate the final ammonium salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this streamlined synthesis route offers profound advantages for procurement managers and supply chain directors tasked with optimizing the sourcing of energetic material precursors. The reduction from a five-step process to a two-step sequence fundamentally alters the cost structure of the final product by eliminating the need for multiple intermediate purifications, solvent swaps, and extended reaction times. This simplification translates directly into reduced utility consumption, lower labor costs, and decreased waste disposal fees, all of which contribute to a more competitive pricing model. Furthermore, the reliance on commodity chemicals such as sulfuric acid, nitric acid, and ammonia ensures that the raw material supply chain is robust and resistant to market volatility, unlike specialized reagents that may face availability constraints.
- Cost Reduction in Manufacturing: The primary driver of cost savings in this process is the drastic reduction in unit operations. By avoiding the isolation of unstable intermediates and eliminating three distinct synthetic steps, manufacturers can significantly reduce the consumption of solvents and energy required for heating and cooling cycles. The high overall yield reported in the patent examples, reaching up to 82.1%, means that less raw material is wasted, directly improving the material efficiency of the plant. Additionally, the use of inexpensive starting materials like 3,5-diamino-1,2,4-triazole and common mineral acids ensures that the variable cost per kilogram of product remains low, facilitating better margin management for downstream applications.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the simplicity of the reagent list. Since the process relies on bulk industrial chemicals that are widely produced and stocked globally, the risk of supply disruption due to vendor-specific issues is minimized. The short reaction time also implies a faster turnaround from raw material intake to finished goods, allowing for more agile inventory management and the ability to respond quickly to fluctuations in demand. This agility is crucial for industries where just-in-time delivery is preferred, as it reduces the need for holding large safety stocks of expensive intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions, specifically the low temperature range of -10°C to 10°C, make this process highly amenable to scale-up in standard stainless steel reactors without requiring exotic materials of construction. From an environmental standpoint, the reduction in step count inherently reduces the volume of hazardous waste generated per unit of product. The ability to recycle the aqueous acid phase or treat it more efficiently due to the lack of complex organic byproducts further enhances the environmental profile of the manufacturing process, aiding in compliance with increasingly stringent global regulations on chemical emissions and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring that stakeholders have accurate information for decision-making. Understanding these details is vital for assessing the feasibility of integrating this technology into existing production lines.
Q: What are the critical temperature controls for the nitration step?
A: The patent specifies a strict temperature range of -10°C to 10°C during the addition of the starting material and the subsequent stirring period to prevent decomposition and ensure high yield.
Q: Which ammonium sources are compatible with this synthesis?
A: The process is versatile and supports various ammonium sources including ammonia water (NH3·H2O), ammonium chloride (NH4Cl) solution, and ammonium nitrate (NH4NO3) solution.
Q: How does this method improve upon traditional five-step syntheses?
A: By utilizing a direct one-pot nitration strategy, this method eliminates three intermediate isolation steps, significantly reducing raw material loss, processing time, and waste generation compared to conventional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ammonium 3,5-Dinitroamino-1,2,4-Triazolate Supplier
As the demand for high-performance energetic materials continues to grow, partnering with a manufacturer that possesses deep technical expertise in nitration chemistry is critical for success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with advanced corrosion-resistant reactors and rigorous QC labs capable of handling the specific thermal and safety requirements of nitro-compound synthesis. We are committed to delivering products that meet stringent purity specifications, ensuring that every batch performs consistently in your final applications, whether for propellants, explosives, or gas generators.
We invite you to collaborate with us to explore how this innovative synthesis route can be tailored to your specific volume and quality requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this direct nitration method. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this critical energetic material intermediate.
