Advanced Synthesis of 7-Amino-6-Nitrobenzodifuroxan: A Safe and Scalable Route for High-Energy Materials
Advanced Synthesis of 7-Amino-6-Nitrobenzodifuroxan: A Safe and Scalable Route for High-Energy Materials
The development of high-performance energetic materials is a critical frontier in defense and aerospace engineering, driving the demand for precursors with superior density and detonation velocity. Patent CN115746021A introduces a groundbreaking synthesis method for 7-amino-6-nitrobenzodifuroxan, also known as CL-18, and its isomers, addressing long-standing safety and efficiency challenges in the industry. This compound is renowned for its exceptional crystal density of approximately 1.936 g/cm³ and a theoretical detonation velocity reaching 8370 m/s, making it a superior candidate for next-generation munitions. The disclosed technology shifts away from hazardous traditional intermediates, offering a streamlined pathway that enhances operational safety without compromising the structural integrity required for high-energy applications. For R&D directors and procurement specialists, this patent represents a pivotal opportunity to secure a more reliable supply chain for advanced energetic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzofuroxan-based energetic compounds has been plagued by significant safety hazards and complex processing requirements that hinder large-scale production. Conventional routes frequently utilize pentanitroaniline as a key intermediate, a substance characterized by extreme sensitivity to impact and friction, thereby posing unacceptable risks during industrial handling and storage. Alternative pathways starting from trifluorobenzene or dichloroaniline often suffer from excessively long reaction sequences and poor overall yields, rendering them economically unviable for commercial scale-up. These legacy methods not only increase the cost of goods sold due to low efficiency but also introduce substantial variability in the impurity profile of the final product. Furthermore, the necessity to isolate unstable intermediates in solid form creates bottlenecks in the supply chain, as specialized containment and handling protocols are mandatory to prevent accidental initiation.
The Novel Approach
The innovative methodology outlined in the patent circumvents these critical bottlenecks by employing a cascade reaction strategy starting from 3,5-dihalogenated anisole derivatives. This approach fundamentally alters the risk profile of the synthesis by avoiding the isolation of the highly sensitive 2,4,6-trinitro-3,5-diazido anisole intermediate as a solid product. Instead, the process utilizes a liquid-phase transition where the diazido species is generated and immediately consumed in subsequent cyclization steps, drastically mitigating the potential for explosive accidents. The route is characterized by its operational simplicity, requiring standard nitration and phase-transfer catalysis conditions that are easily adaptable to existing reactor infrastructure. By eliminating the need for complex purification of hazardous intermediates, the novel approach ensures a smoother workflow that is inherently safer and more conducive to engineering amplification. This strategic shift allows manufacturers to achieve acceptable yields while maintaining rigorous safety standards essential for energetic material production.
Mechanistic Insights into Nitration and Azidation Cascade
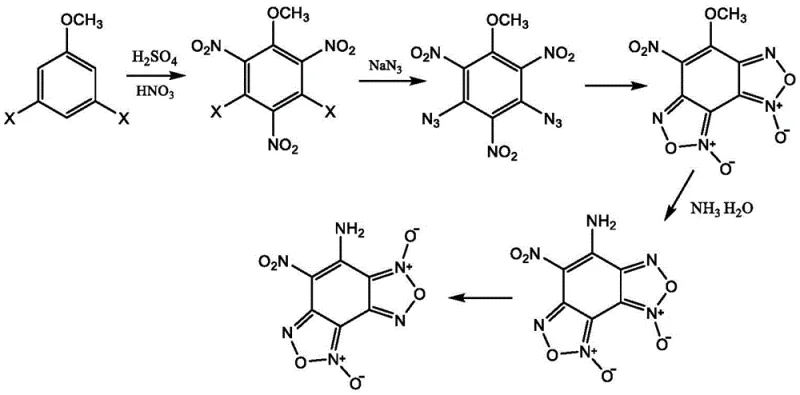
The core of this synthesis lies in a meticulously orchestrated sequence of electrophilic aromatic substitution and nucleophilic displacement reactions that construct the dense furoxan framework. The process initiates with the nitration of 3,5-dihalogenated anisole under controlled ice-water bath conditions, followed by heating to 90-100°C to install three nitro groups on the aromatic ring. Subsequently, a phase-transfer catalyzed azidation replaces the halogen atoms with azide groups using sodium azide and tetrabutylammonium bromide, a step critical for setting up the subsequent ring closure. The resulting trinitro-diazido intermediate undergoes thermal cyclization in solvents like glacial acetic acid or toluene at elevated temperatures, forming the methoxy-substituted benzodifuroxan core. This cyclization is driven by the intramolecular interaction of the azide and nitro groups, creating the fused heterocyclic system that defines the energetic properties of the molecule.
Following the formation of the core structure, the methoxy group is converted to an amino group through ammonolysis, yielding a specific isomer of 7-amino-6-nitrobenzofuroxan. This isomer is then subjected to a final thermal isomerization step in acidic media to obtain the thermodynamically stable CL-18 product. The control of isomerization is vital, as the arrangement of the furoxan rings significantly influences the crystal packing and detonation performance. The patent details specific solvent systems and temperature ranges, such as heating in propionic acid at 120°C, to ensure complete conversion to the desired isomer. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters for optimal purity and yield.
How to Synthesize 7-Amino-6-Nitrobenzodifuroxan Efficiently
Implementing this synthesis requires precise control over reaction temperatures and stoichiometry to manage the exothermic nature of nitration and the sensitivity of azide intermediates. The protocol begins with the careful addition of halogenated anisole to mixed acid, ensuring thermal runaway is prevented during the initial nitration phase. Operators must strictly adhere to the specified reaction times, such as maintaining the azidation at room temperature for 2 to 20 hours depending on the solvent choice, to ensure complete conversion before proceeding to cyclization. The detailed standardized synthesis steps below outline the specific workup procedures, including quenching, filtration, and recrystallization techniques necessary to isolate high-purity intermediates and the final product.
- Nitration of 3,5-dihalogenated anisole using mixed acid to form trinitro intermediates.
- Phase-transfer catalyzed azidation followed by thermal cyclization to form the benzodifuroxan core.
- Ammonolysis to generate the amino-isomer, followed by thermal isomerization to the final CL-18 product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative advantages by decoupling high-performance material production from high-risk operational constraints. The elimination of solid isolation for the diazido intermediate removes the need for expensive blast-proof facilities and specialized transport logistics, directly translating to reduced overhead costs. Additionally, the use of commodity chemicals like anisole derivatives and sodium azide ensures a robust and resilient supply base, minimizing the risk of raw material shortages that often plague specialty chemical markets. The simplified process flow reduces the number of unit operations required, which in turn lowers energy consumption and waste generation, aligning with increasingly stringent environmental regulations. These factors collectively enhance the economic viability of producing CL-18, making it a more accessible option for large-scale energetic formulations.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis significantly lowers operational expenditures by removing complex purification stages associated with hazardous intermediates. By avoiding the isolation of sensitive solids, the process reduces the need for specialized containment equipment and minimizes material loss during transfer operations. The use of common solvents and catalysts further drives down raw material costs, while the improved yield stability ensures consistent output volumes. Consequently, manufacturers can achieve a more favorable cost structure compared to legacy routes that suffer from low efficiency and high waste disposal fees.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials such as dihalogenated anisoles ensures that production schedules are not disrupted by niche supplier constraints. The robustness of the reaction conditions allows for flexible manufacturing planning, as the process is less susceptible to minor variations in reagent quality. This reliability is crucial for maintaining continuous supply lines to downstream formulators who depend on steady deliveries of energetic intermediates. Furthermore, the reduced safety risk profile simplifies regulatory compliance, accelerating the approval processes required for scaling up production capacity.
- Scalability and Environmental Compliance: The inherent safety of the liquid-phase processing facilitates easier scale-up from laboratory to pilot and commercial plant scales without extensive re-engineering. The reduction in hazardous waste generation, particularly the avoidance of solid explosive byproducts, simplifies effluent treatment and lowers the environmental footprint of the facility. This alignment with green chemistry principles not only mitigates regulatory risks but also enhances the corporate sustainability profile of the manufacturer. As global standards for energetic material production tighten, this scalable and compliant process positions suppliers as preferred partners for long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 7-amino-6-nitrobenzodifuroxan. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their existing product portfolios. The answers highlight the balance between performance optimization and process safety that defines this new manufacturing approach.
Q: Why is the new synthesis route for CL-18 considered safer than traditional methods?
A: Traditional methods often rely on pentanitroaniline, which is highly sensitive and dangerous. The new route avoids isolating the unstable diazido intermediate as a solid, significantly reducing explosion risks during manufacturing.
Q: What are the key raw materials required for this synthesis?
A: The process starts with readily available 3,5-dihalogenated anisole derivatives. Key reagents include nitric acid, sulfuric acid, sodium azide, and ammonia water, all of which are standard industrial chemicals.
Q: How does this process impact the purity of the final energetic material?
A: The method utilizes specific recrystallization steps and controlled isomerization conditions, ensuring high purity of the final 7-amino-6-nitrobenzodifuroxan product, which is critical for consistent detonation performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Amino-6-Nitrobenzodifuroxan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing high-quality energetic intermediates that meet the rigorous demands of modern defense and aerospace applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 7-amino-6-nitrobenzodifuroxan delivers the expected performance characteristics. Our commitment to safety and quality makes us an ideal partner for organizations seeking to leverage advanced synthesis technologies for their next-generation formulations.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this novel synthesis route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your operational constraints. Let us collaborate to drive innovation and efficiency in the field of high-energy materials together.
