Advanced Manufacturing of 2-Fluoro-3-Trifluoromethylaniline: A Scalable Route for Global Pharmaceutical Supply Chains
The global demand for fluorinated pharmaceutical intermediates continues to surge, driven by the critical role these molecules play in enhancing the metabolic stability and bioavailability of modern drug candidates. A pivotal development in this sector is detailed in patent CN114920655A, which discloses a robust and economically viable preparation method for 2-fluoro-3-trifluoromethylaniline. This specific aniline derivative serves as a cornerstone building block for a wide array of therapeutic agents, including tyrosine kinase inhibitors and androgen receptor antagonists like proclutamide, which has shown promise in clinical trials for viral infections. The patented technology addresses long-standing inefficiencies in traditional synthesis by introducing a streamlined three-step sequence: regioselective nitration, nucleophilic fluorination, and catalytic hydrogenation. By shifting the synthetic strategy to utilize abundant chlorinated precursors rather than scarce fluorinated ones, this innovation offers a compelling value proposition for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios. The technical breakthrough lies not just in the chemical transformation itself, but in the holistic improvement of process safety, waste reduction, and overall cost structure, positioning it as a preferred route for cost reduction in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-fluoro-3-trifluoromethylaniline has been plagued by significant economic and operational bottlenecks that hinder efficient commercial scale-up of complex fluorinated intermediates. As illustrated in prior art such as patents DE3737986 and EP318704, traditional routes often rely on 2-fluoro-5-chlorotrifluorotoluene as the starting material. This precursor is inherently expensive due to the difficulty of introducing the fluorine atom early in the synthesis chain, creating a high baseline cost that propagates through the entire production lifecycle. Furthermore, alternative pathways described in literature, such as those involving low-temperature lithiation followed by carboxylation and Hoffmann rearrangement, introduce severe safety hazards and operational complexities. These legacy methods frequently require stringent anhydrous and anaerobic conditions, cryogenic temperatures, and the handling of unstable acyl chloride intermediates, all of which escalate capital expenditure and increase the risk of batch failure. The accumulation of toxic by-products and the generation of substantial "three wastes" (waste water, gas, and residue) further complicate regulatory compliance, making these conventional approaches increasingly untenable for modern, sustainability-focused chemical enterprises.
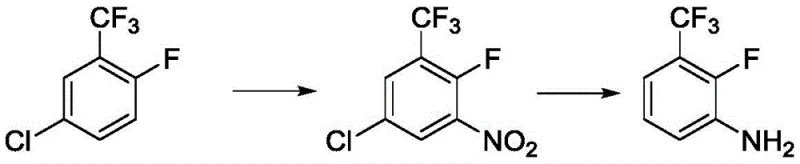
The Novel Approach
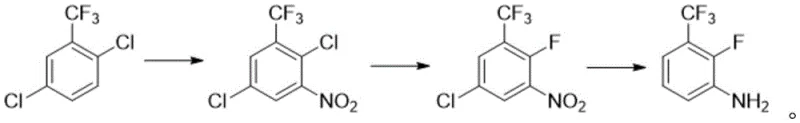
In stark contrast to these cumbersome legacy processes, the methodology outlined in CN114920655A represents a paradigm shift towards efficiency and accessibility. The novel approach initiates with 2,5-dichlorobenzotrifluoride, a commodity chemical that is significantly cheaper and more readily available on the global market than its fluorinated counterparts. The process elegantly installs the nitro group first, leveraging the directing effects of the existing substituents to achieve high regioselectivity, followed by a nucleophilic aromatic substitution (SNAr) to introduce the fluorine atom. This strategic reversal of functional group installation allows for milder reaction conditions and eliminates the need for hazardous rearrangement reactions. The final step involves a clean catalytic reduction to yield the target aniline. This streamlined workflow not only simplifies the operational protocol but also drastically improves the atom economy of the synthesis. By avoiding the pitfalls of expensive raw materials and dangerous reaction conditions, this new route provides a clear pathway for reducing lead time for high-purity intermediates, ensuring a more resilient and responsive supply chain for downstream pharmaceutical manufacturers.
Mechanistic Insights into Regioselective Nitration and SNAr Fluorination
The success of this synthetic strategy hinges on the precise control of regiochemistry during the initial nitration step and the efficiency of the subsequent nucleophilic fluorination. In the first stage, 2,5-dichlorobenzotrifluoride is subjected to electrophilic aromatic substitution using a nitrating agent such as fuming nitric acid in a strong acid medium like concentrated sulfuric acid. The electron-withdrawing nature of the trifluoromethyl and chloro groups deactivates the ring, necessitating careful control of temperature (0-45°C) and stoichiometry to prevent over-nitration or oxidation side reactions. The chlorine atom at the 2-position and the trifluoromethyl group at the 1-position direct the incoming nitro group predominantly to the 3-position, yielding 3-nitro-2,5-dichlorobenzotrifluoride with high purity (>98.5% HPLC). This high degree of selectivity is crucial for minimizing impurity profiles that could be difficult to remove in later stages, thereby ensuring the production of high-purity pharmaceutical intermediates that meet stringent quality specifications required by global regulatory bodies.
Following nitration, the process employs a nucleophilic aromatic substitution (SNAr) mechanism to replace the activated chlorine atom at the 2-position with a fluorine atom. The presence of the strongly electron-withdrawing nitro group ortho to the leaving chlorine significantly activates the ring towards nucleophilic attack, allowing the reaction to proceed with alkali metal fluorides such as potassium fluoride or cesium fluoride. The reaction is typically conducted in polar aprotic solvents like DMSO or DMF at elevated temperatures (60-120°C) to overcome the activation energy barrier. The choice of fluoride source and solvent system is critical; for instance, using cesium fluoride can enhance reaction rates due to its higher solubility and weaker lattice energy compared to potassium fluoride. The final reduction step utilizes heterogeneous catalysis, typically with Pd/C or Raney Nickel under hydrogen pressure (1.5-5 atm), to convert the nitro group to an amine without affecting the sensitive carbon-fluorine or carbon-chlorine bonds. This chemoselective reduction is vital for maintaining the structural integrity of the fluorinated scaffold while achieving the desired aniline functionality.
How to Synthesize 2-Fluoro-3-Trifluoromethylaniline Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process is designed to be robust, tolerating slight variations in conditions while maintaining high performance, which is essential for transfer from laboratory to pilot and production scales. Operators must focus on the precise control of the nitration exotherm and the efficient removal of inorganic salts generated during the fluorination workup. The simplicity of the isolation procedures—primarily involving filtration, water washing, and recrystallization—makes this route particularly attractive for facilities aiming to minimize downtime between batches. For a detailed breakdown of the specific reagent quantities, temperature profiles, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Dissolve 2,5-dichlorobenzotrifluoride in a strong acid solvent and react with a nitrating agent at 0-45°C to obtain 3-nitro-2,5-dichlorobenzotrifluoride.
- Perform nucleophilic aromatic substitution by reacting the nitro-intermediate with a fluoride source (e.g., KF, CsF) in a polar aprotic solvent at 60-120°C.
- Reduce the nitro group to an amine using a metal catalyst (Pd/C, Raney Ni) and hydrogen gas under mild pressure (1.5-5 atm) to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond simple unit price considerations. The fundamental shift from expensive, specialized fluorinated starting materials to commodity chlorinated chemicals creates a buffer against raw material price volatility, ensuring more stable costing models for long-term contracts. Furthermore, the reduction in synthetic steps and the elimination of hazardous unit operations like cryogenic reactions or Hoffmann rearrangements significantly lower the barrier to entry for contract manufacturing organizations (CMOs). This accessibility fosters a more competitive supplier landscape, giving buyers greater leverage and security of supply. The simplified process flow also means that production capacity can be ramped up more quickly in response to surging demand, as the equipment requirements are less specialized and more widely available in standard multipurpose chemical plants.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this route is the substitution of high-cost 2-fluoro-5-chlorotrifluorotoluene with inexpensive 2,5-dichlorobenzotrifluoride. By deferring the introduction of the fluorine atom to a later stage via nucleophilic substitution, the process leverages cheaper bulk chemicals while avoiding the premium pricing associated with pre-fluorinated aromatics. Additionally, the avoidance of complex purification steps required for removing by-products from rearrangement reactions further reduces utility consumption and waste disposal costs. The overall yield exceeding 68% ensures that material throughput is maximized, directly contributing to a lower cost of goods sold (COGS) without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available raw materials that are produced by multiple global vendors, reducing the risk of single-source bottlenecks. The mild reaction conditions (0-120°C) and ambient pressure hydrogenation allow for production in a broader range of facilities, diversifying the potential manufacturing base. This flexibility is crucial for mitigating risks associated with geopolitical disruptions or localized production shutdowns. Moreover, the robustness of the chemistry means that batch failure rates are minimized, leading to more predictable delivery schedules and improved on-time in-full (OTIF) performance for downstream customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a distinct advantage by generating fewer hazardous wastes and avoiding the use of toxic reagents common in older methodologies. The "three wastes" are markedly reduced, simplifying effluent treatment and lowering the environmental compliance burden. The elimination of pyrophoric reagents and extreme low-temperature operations enhances workplace safety, reducing insurance premiums and liability risks. These factors collectively make the process highly scalable, allowing for seamless transition from kilogram-scale development to multi-ton commercial production while adhering to increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-fluoro-3-trifluoromethylaniline using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process feasibility and procurement teams assessing supplier capabilities. The answers highlight the specific improvements in yield, purity, and operational safety that distinguish this route from conventional alternatives.
Q: What are the key advantages of the new synthesis route compared to traditional methods?
A: The new route utilizes inexpensive 2,5-dichlorobenzotrifluoride instead of costly fluorinated starting materials, avoids hazardous Hoffmann rearrangements, and achieves a total yield exceeding 68% with purity over 95%.
Q: How does this process address environmental and safety concerns in fluorinated intermediate production?
A: By eliminating toxic reagents and high-risk low-temperature anhydrous conditions, the process significantly reduces 'three wastes' and operational hazards, ensuring better compliance with modern green chemistry standards.
Q: Is this manufacturing method suitable for large-scale commercial production?
A: Yes, the process features mild reaction conditions (0-120°C), simple post-treatment operations like filtration and recrystallization, and uses readily available raw materials, making it highly scalable for industrial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-3-Trifluoromethylaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable, and compliant manufacturing processes in the modern pharmaceutical landscape. Our technical team has thoroughly analyzed the advancements presented in CN114920655A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative route to life. We are committed to delivering high-purity 2-fluoro-3-trifluoromethylaniline that meets the most rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Our facility is equipped to handle the specific solvent systems and catalytic hydrogenation requirements of this process, ensuring consistent batch-to-batch reproducibility and stringent purity specifications that exceed industry norms.
We invite global partners to collaborate with us to leverage these technological advantages for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to discuss specific COA data, route feasibility assessments, and how our implementation of this advanced synthesis method can drive value and security for your upcoming projects. Let us be your trusted partner in navigating the complexities of fluorinated intermediate manufacturing.
