Revolutionizing Vitamin A Intermediate Production via One-Step Phosphonate Coupling Technology
Revolutionizing Vitamin A Intermediate Production via One-Step Phosphonate Coupling Technology
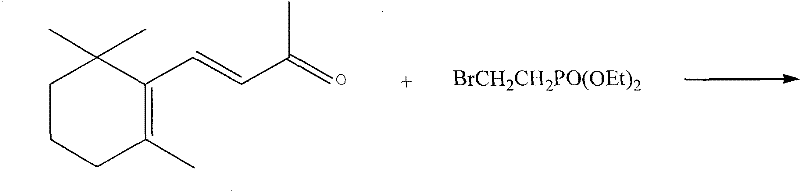
The global demand for Vitamin A and its derivatives, such as retinyl acetate and retinyl palmitate, continues to drive innovation in the fine chemical sector, particularly regarding the efficiency of key intermediate synthesis. Patent CN101492471B introduces a transformative methodology for producing 3-methyl-5-(2,6,6-trimethyl-1-cyclohexene-1-nyl)-2,4-pentadienyl phosphonic acid ethyl ester, a critical C15 building block in the Wittig-Horner synthesis of Vitamin A alkyl esters. Unlike traditional multi-step pathways that plague the industry with yield losses and operational complexity, this invention leverages a direct condensation strategy between beta-ionone and 1-phosphonate group-2-bromoethane. By integrating the coupling and dehydration stages into a seamless one-pot operation, this technology addresses the longstanding bottlenecks in retinoid manufacturing, offering a robust solution for reliable vitamin A intermediate supplier networks seeking to optimize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
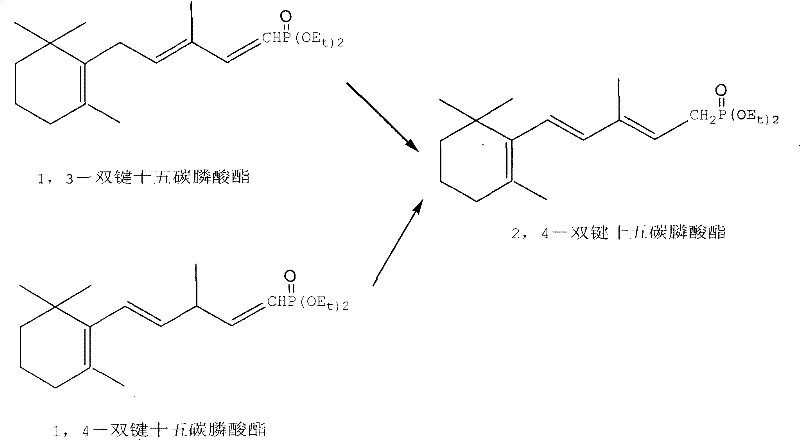
Historically, the synthesis of the requisite 2,4-diene 15-carbon phosphonate has been fraught with inefficiencies, primarily relying on circuitous routes that begin with beta-ionone but require extensive manipulation to achieve the correct double bond positioning. Conventional strategies often involve the initial formation of 1,3- or 1,4-diene isomers through Wittig-Horner reactions with gem-diphosphonates, followed by energetically demanding isomerization processes to shift the double bonds into the thermodynamically favored 2,4-conjugated system. Alternatively, some prior art methods utilize Grignard reagents at cryogenic temperatures to add carbon fragments, a technique that not only incurs high energy costs for cooling but also suffers from moderate yields, reported around 78.4% in specific instances, due to side reactions and the difficulty of controlling selectivity. These fragmented approaches necessitate multiple isolation and purification steps, significantly inflating the cost of goods sold and complicating the supply chain for high-purity retinoid intermediates.
The Novel Approach
In stark contrast to these laborious legacy methods, the technology disclosed in CN101492471B streamlines the entire synthesis into a singular, cohesive reaction sequence that bypasses the need for pre-formed aldehyde intermediates or difficult isomerization steps. The core innovation lies in the direct nucleophilic attack of an organozinc species, generated in situ from 1-phosphonate group-2-bromoethane and active zinc, onto the carbonyl group of beta-ionone. This coupling event generates a tertiary alcohol phosphonate intermediate which, rather than being isolated, is immediately subjected to acid-catalyzed dehydration within the same reaction vessel. This telescoped process eliminates the need for intermediate workups, reduces solvent consumption, and avoids the thermal stress associated with distilling sensitive allylic alcohols, thereby preserving the integrity of the conjugated diene system and ensuring a higher overall throughput for commercial scale-up of complex fine chemicals.
Mechanistic Insights into Zinc-Mediated Condensation and Dehydration
The chemical elegance of this process is rooted in the reactivity of the zinc-mediated coupling, which proceeds under mild thermal conditions ranging from 10°C to 50°C, avoiding the extreme cold required by Grignard counterparts. The active zinc surface, often enhanced as a zinc-copper couple, facilitates the oxidative insertion into the carbon-bromine bond of the phosphonate bromide, generating a reactive organometallic nucleophile that attacks the ketone functionality of beta-ionone with high regioselectivity. Following the formation of the beta-hydroxy phosphonate intermediate, the addition of a mineral acid such as sulfuric acid or hydrochloric acid triggers an E1 or E2 elimination mechanism. This dehydration step is driven by the thermodynamic stability gained from extending the conjugation of the pi-system, naturally favoring the formation of the 2,4-diene geometry over other potential isomers without the need for external equilibration catalysts.
Furthermore, the impurity profile of the resulting product is significantly cleaner due to the avoidance of harsh basic conditions often used in traditional Wittig olefinations, which can lead to epimerization or degradation of the sensitive cyclohexenyl ring. The acid-catalyzed dehydration occurs smoothly at temperatures between 20°C and 50°C, ensuring that the delicate polyene chain remains intact while water is eliminated to form the final double bond. This mechanistic pathway inherently suppresses the formation of saturated byproducts or over-reacted species, resulting in a crude product with high gas chromatography purity, often exceeding 90% before final distillation. Such inherent selectivity reduces the burden on downstream purification units, allowing for more efficient vacuum distillation protocols to achieve the stringent purity specifications required for pharmaceutical grade Vitamin A precursors.
How to Synthesize 2,4-Double-Bond 15-Carbon Phosphonate Efficiently
The practical execution of this synthesis relies on careful control of the zinc activation state and the sequential addition of reagents to manage the exothermic nature of the organometallic formation. Operators must ensure the zinc surface is highly active, typically achieved through sodium-naphthalene activation or copper plating, to initiate the coupling efficiently in solvents like tetrahydrofuran or dimethylformamide. Once the condensation is complete, indicated by the disappearance of the starting ketone via thin-layer chromatography, the reaction mixture is treated directly with aqueous acid to effect dehydration, followed by a standard extractive workup and vacuum distillation to isolate the target phosphonate. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Prepare active zinc-copper couple or activated zinc powder and suspend in anhydrous THF under inert atmosphere.
- Add 1-phosphonate group-2-bromoethane and beta-ionone sequentially, maintaining temperature between 10°C to 50°C for condensation.
- Without isolating the intermediate tertiary alcohol, add dilute sulfuric acid directly to the reaction mixture and stir at 20-50°C to effect dehydration and obtain the target diene phosphonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-step synthesis represents a strategic opportunity to de-risk the sourcing of critical Vitamin A intermediates while simultaneously driving down manufacturing overheads. By collapsing a multi-step sequence into a single operational unit, the technology drastically reduces the man-hours required for production, minimizes the inventory of hazardous intermediates that need to be stored and transported, and lowers the overall solvent intensity of the process. This consolidation translates directly into a more resilient supply chain, as the reliance on specialized cryogenic equipment and complex multi-stage purification trains is eliminated, allowing for faster batch turnover and improved responsiveness to market fluctuations in the nutraceutical and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps and the removal of expensive transition metal catalysts or cryogenic cooling requirements lead to substantial cost savings in utility and material consumption. The process utilizes commodity chemicals like beta-ionone and simple mineral acids, avoiding the premium pricing associated with specialized organolithium or Grignard reagents, thereby optimizing the raw material cost structure for cost reduction in pharmaceutical manufacturing.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route reduces the number of potential failure points in the production line, ensuring a more consistent and reliable output of high-purity retinoid intermediates. The robustness of the zinc-mediated chemistry allows for flexible scheduling and easier troubleshooting compared to sensitive low-temperature reactions, effectively reducing lead time for high-purity retinoid intermediates and securing continuity of supply for downstream Vitamin A esterification plants.
- Scalability and Environmental Compliance: The use of common organic solvents and the absence of heavy metal catalysts simplify waste stream management and effluent treatment, aligning with increasingly stringent environmental regulations. The process is inherently scalable from pilot batches to multi-ton production without requiring fundamental changes to the reactor infrastructure, facilitating the commercial scale-up of complex fine chemicals with a reduced environmental footprint and lower disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel phosphonate synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the operational benefits and chemical principles involved. Stakeholders are encouraged to review these points to assess the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary limitations of conventional 15-carbon phosphonate synthesis?
A: Conventional methods typically involve multi-step sequences starting from beta-ionone, often requiring the synthesis of 1,3- or 1,4-diene isomers which then necessitate complex isomerization or difficult selective reduction steps to achieve the thermodynamically stable 2,4-diene configuration. These routes suffer from low overall yields due to the accumulation of losses over three or more distinct reaction stages and often require harsh cryogenic conditions for Grignard reagents.
Q: How does the novel zinc-mediated route improve process efficiency?
A: The novel approach described in patent CN101492471B consolidates the synthesis into a single operational sequence where beta-ionone couples directly with 1-phosphonate group-2-bromoethane using active zinc. Crucially, the intermediate tertiary alcohol phosphonate is not isolated; instead, it undergoes immediate in-situ acid-catalyzed dehydration, eliminating purification steps between the coupling and elimination phases and drastically reducing solvent usage and processing time.
Q: Is this technology suitable for large-scale industrial production?
A: Yes, the process is highly amenable to industrial scale-up because it utilizes commercially available raw materials like beta-ionone and avoids the need for expensive transition metal catalysts or extremely low-temperature equipment. The use of common solvents like THF and standard mineral acids for dehydration simplifies waste treatment and allows for robust process control in large reactor vessels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Double-Bond 15-Carbon Phosphonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the global Vitamin A value chain, and we possess the technical expertise to translate this patented methodology into commercial reality. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with international quality standards. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced GC-MS and NMR instrumentation to guarantee that every batch of 2,4-diene phosphonate meets the exacting requirements for pharmaceutical and food-grade applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the economic benefits specific to your operation, along with specific COA data and route feasibility assessments that demonstrate our commitment to delivering superior value and supply security for your Vitamin A production needs.
