Advanced Synthesis of Carbazole-Based Sulfonyl Chloride for High-Sensitivity Bioanalysis and Commercial Scale-Up
Introduction to Patent CN102702074B and Technological Breakthroughs
The pharmaceutical and analytical chemistry sectors are constantly seeking more robust and sensitive tools for trace analysis, particularly for detecting alcohols and amines in complex biological matrices. Patent CN102702074B introduces a significant advancement in this domain by disclosing a novel preparation method for a sulfonyl chloride compound utilizing carbazole as a fluorophore. This specific compound, identified as 3,6-dimethoxy-9-phenylcarbazole-1-sulfonyl chloride, represents a critical evolution in pre-column derivatization reagents used for high-performance liquid chromatography (HPLC). Unlike conventional markers that often suffer from instability or interference issues, this new molecular architecture integrates a carbazole parent ring with auxiliary methoxy and phenyl substituents to maximize conjugation and fluorescence intensity. For R&D directors and procurement specialists alike, understanding the synthesis and application of this reliable fluorescent probe supplier material is essential for developing next-generation diagnostic assays and quality control protocols in the pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the analysis of amines and alcohols lacking chromophoric groups has relied heavily on pre-column derivatization, yet existing technologies present substantial operational hurdles. Many traditional labeling reagents exhibit extreme sensitivity to atmospheric moisture, leading to premature hydrolysis before they can react with the target analyte. This hydrolysis not only wastes expensive reagents but generates by-products that co-elute with the desired derivatives, causing significant interference in chromatographic separation. Furthermore, the structural limitations of older reagents often result in insufficient detection sensitivity, rendering them incapable of quantifying trace compounds at the levels required for modern disease diagnosis or environmental monitoring. Additionally, the stability of the resulting derivative products is frequently compromised, leading to drift in quantitative results over time, while some reagents introduce toxicity concerns through the generation of corrosive gases during the labeling process.
The Novel Approach
The methodology outlined in the patent addresses these systemic failures through a rationally designed synthetic pathway that prioritizes both molecular stability and reaction specificity. By employing a carbazole backbone substituted with electron-donating groups, the new reagent achieves a level of fluorescence intensity that supports fmol-level detection limits, far surpassing the capabilities of many legacy compounds. The synthesis strategy specifically controls the substitution pattern to prevent the formation of multi-substituted by-products, a common issue in sulfonation reactions. Moreover, the derivatization reaction conditions are remarkably mild, requiring only a slight excess of the reagent to drive the reaction to completion without generating toxic waste streams. This approach ensures that any unreacted reagent hydrolyzes into products that are chromatographically distinct from the analytes, thereby eliminating background noise and ensuring the integrity of the analytical data for high-purity pharmaceutical intermediates.
Mechanistic Insights into Carbazole Functionalization and Derivatization
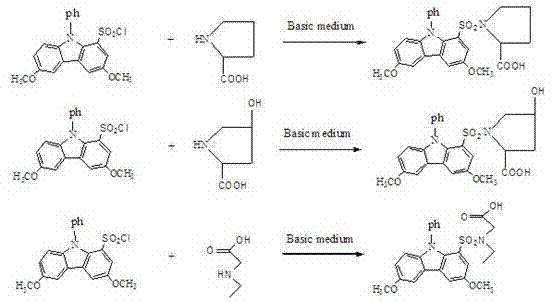
The synthesis of 3,6-dimethoxy-9-phenylcarbazole-1-sulfonyl chloride involves a sophisticated four-step cascade that exemplifies modern heterocyclic chemistry. The process initiates with the regioselective bromination of carbazole using N-bromosuccinimide (NBS) catalyzed by silica gel in dichloromethane, a choice that allows for precise control over the degree of substitution at room temperature. This is followed by a nucleophilic substitution where the bromine atoms are replaced by methoxy groups using sodium methoxide and cuprous iodide in DMF, establishing the electron-rich nature of the final fluorophore. The subsequent N-phenylation step utilizes iodobenzene and cesium carbonate in acetonitrile; the selection of acetonitrile is critical as its lower boiling point (81.1°C) facilitates reflux at milder temperatures, while the weak basicity of cesium carbonate minimizes side reactions. Finally, the introduction of the reactive chlorosulfonyl group is achieved via a two-stage process involving chlorosulfonic acid and thionyl chloride, carefully managed at a 1:1.5 molar ratio to ensure mono-substitution.
Once synthesized, the reagent functions as a highly efficient electrophile in derivatization reactions with nucleophilic amines. The mechanism involves the nucleophilic attack of the amine nitrogen on the sulfur atom of the sulfonyl chloride group, displacing the chloride ion to form a stable sulfonamide bond. This reaction is kinetically favorable under mild alkaline conditions (pH 10), allowing for rapid labeling of targets such as L-hydroxyproline, L-proline, and N-ethylglycine. The resulting sulfonamide derivatives retain the intense fluorescence of the carbazole core, which is further enhanced by the extended conjugation system. Crucially, the steric and electronic properties of the 3,6-dimethoxy-9-phenyl configuration protect the sulfonamide bond from hydrolytic cleavage, ensuring that the derivatives remain stable for weeks, a feature that is vital for batch processing in clinical laboratories.
How to Synthesize 3,6-Dimethoxy-9-phenylcarbazole-1-sulfonyl chloride Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and purification protocols defined in the patent to ensure the high purity necessary for analytical applications. The process is designed to be scalable, moving from gram-scale laboratory preparations to potential commercial production without losing yield or selectivity. Key to the success of this manufacturing process is the rigorous control of reaction temperatures and the use of specific catalysts like silica gel and cuprous iodide which can be recovered or managed effectively. The following guide outlines the standardized operational procedure derived from the patent claims, providing a roadmap for process chemists aiming to replicate this high-value intermediate.
- Bromination of carbazole using N-bromosuccinimide and silica gel catalyst in dichloromethane.
- Substitution to form 3,6-dimethoxycarbazole using sodium methoxide and cuprous iodide in DMF.
- N-phenylation using iodobenzene, cesium carbonate, and copper powder in acetonitrile.
- Final sulfonyl-chlorination using chlorosulfonic acid followed by thionyl chloride treatment.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical performance. The shift towards this specific carbazole derivative addresses critical pain points related to reagent stability and waste management, which directly impact the total cost of ownership for analytical laboratories. By utilizing a synthesis pathway that avoids harsh conditions and toxic by-products, manufacturers can significantly reduce the costs associated with hazardous waste disposal and specialized containment equipment. Furthermore, the robustness of the final product means less frequent re-ordering due to spoilage, enhancing inventory management efficiency. The ability to source a reliable fluorescent probe supplier who can deliver this specific intermediate ensures continuity in diagnostic testing workflows, which is paramount for time-sensitive medical applications.
- Cost Reduction in Manufacturing: The synthesis protocol eliminates the need for expensive transition metal catalysts that are difficult to remove, relying instead on more economical and manageable catalysts like silica gel and copper powder. The use of acetonitrile as a solvent in the phenylation step not only lowers energy consumption due to reduced reflux temperatures but also simplifies solvent recovery processes. By preventing the formation of polysubstituted by-products through precise molar control of chlorosulfonic acid, the process maximizes atom economy and reduces the burden on downstream purification steps like column chromatography. These cumulative efficiencies translate into substantial cost savings in the manufacturing of complex analytical reagents without compromising on the stringent purity specifications required for pharmaceutical analysis.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including carbazole, N-bromosuccinimide, and iodobenzene, are commodity chemicals with well-established global supply chains, mitigating the risk of raw material shortages. The synthetic route is robust and tolerant to minor variations in reaction conditions, which enhances the predictability of production schedules and lead times. Because the final product exhibits excellent shelf-life stability, distributors and end-users can maintain larger safety stocks without the fear of degradation, effectively buffering against supply chain disruptions. This reliability is crucial for maintaining the continuous operation of quality control labs in the pharmaceutical and food safety sectors where consistent reagent performance is non-negotiable.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as filtration, extraction, and recrystallization that are easily transferable from pilot plants to large-scale reactors. The avoidance of corrosive gases and toxic by-products during the derivatization application phase aligns with increasingly stringent environmental regulations regarding laboratory safety and waste emission. The recyclability of the silica gel catalyst in the initial step further underscores the green chemistry credentials of this method, appealing to organizations with strong sustainability mandates. Consequently, scaling the production of this carbazole-based sulfonyl chloride presents minimal environmental compliance hurdles, facilitating smoother regulatory approvals for commercial manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this carbazole sulfonyl chloride derivative. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this reagent for their specific analytical needs. Understanding these nuances is key to optimizing the integration of this fluorescent probe into existing HPLC workflows.
Q: What are the stability advantages of this carbazole sulfonyl chloride compared to traditional reagents?
A: Unlike many traditional labeling reagents that are highly sensitive to moisture and prone to hydrolysis, this carbazole-based derivative forms stable products that remain intact for several weeks. The hydrolysis products of the reagent itself do not interfere with the detection of the target analytes, ensuring high reproducibility in HPLC analysis.
Q: How does the synthesis process minimize toxic by-products?
A: The patented method utilizes silica gel as a heterogeneous catalyst for the initial bromination, which is non-toxic and recyclable. Furthermore, the use of acetonitrile as a solvent in the phenylation step allows for lower reaction temperatures due to its boiling point, and the use of cesium carbonate as a mild base prevents the formation of excessive side products often seen with stronger alkalis.
Q: What is the detection sensitivity achievable with this fluorescent probe?
A: The reagent enables ultra-trace analysis with a detection limit reaching the femtomole (fmol) level. This high sensitivity is attributed to the strong fluorescence of the carbazole parent ring enhanced by the electron-donating methoxy and phenyl groups, making it ideal for detecting trace amines in complex biological matrices like plasma.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,6-Dimethoxy-9-phenylcarbazole-1-sulfonyl chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance fluorescent probes play in advancing pharmaceutical research and environmental monitoring. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-purity intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications demanded by the analytical chemistry sector, including detailed impurity profiling and fluorescence validation. We are committed to delivering not just a chemical product, but a comprehensive solution that supports your R&D and quality assurance objectives with unmatched reliability.
We invite you to collaborate with our technical team to explore how this advanced carbazole derivative can enhance your analytical capabilities. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can support your project timelines and quality goals effectively.
