Revolutionizing L-Pyroglutamine Production: A Deep Dive into Solvent-Free Thermal Cyclization Technology
Revolutionizing L-Pyroglutamine Production: A Deep Dive into Solvent-Free Thermal Cyclization Technology
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective pathways for synthesizing critical chiral intermediates. Patent CN101298431B introduces a groundbreaking solvent-free one-step reaction synthesis method for L-pyroglutamine, a vital building block in the production of nootropic drugs, cosmetics, and agrochemicals. This technology represents a significant departure from traditional multi-step esterification processes, offering a streamlined approach that utilizes L-glutamic acid and urea as primary feedstocks. By eliminating organic solvents entirely, this method not only addresses growing environmental compliance pressures but also fundamentally alters the economic landscape of manufacturing this valuable amino acid derivative. For R&D directors and procurement specialists, understanding the mechanistic nuances and commercial implications of this patent is essential for optimizing supply chains and reducing overall production expenditures in the competitive global market.
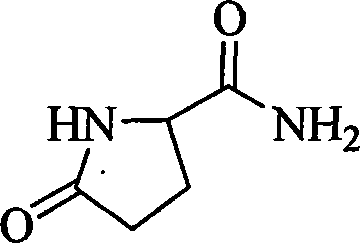
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of L-pyroglutamine has relied heavily on convoluted pathways starting from L-sodium glutamate or L-pyroglutamic acid methyl esters. These conventional methodologies typically necessitate a sequence of acidification, followed by esterification, and finally cyclization, each step requiring distinct reaction conditions and extensive purification protocols. The reliance on organic solvents in these traditional routes introduces significant logistical burdens, including the need for solvent recovery systems, explosion-proof infrastructure, and complex waste treatment facilities to handle volatile organic compounds (VOCs). Furthermore, the use of esterification agents often results in lower atom economy and generates substantial salt waste, driving up both the raw material costs and the environmental footprint of the manufacturing process. These inefficiencies create bottlenecks in scaling production, making it difficult for manufacturers to respond agilely to fluctuating market demands without incurring prohibitive operational expenses.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a direct thermal condensation between L-glutamic acid and urea under solvent-free conditions. This innovative strategy simplifies the synthetic route into a single pot operation where the reactants are heated in two distinct temperature stages to drive the cyclization and amidation reactions simultaneously. The absence of solvent not only removes the associated safety hazards and recovery costs but also increases the effective concentration of reactants, thereby accelerating reaction kinetics and improving overall space-time yield. By utilizing urea as both a nitrogen source and a reaction promoter, the process achieves high conversion rates while generating minimal by-products, primarily ammonia and carbon dioxide, which are easily managed. This streamlined methodology offers a robust platform for cost reduction in amino acid derivative manufacturing, allowing producers to achieve superior economic efficiency without compromising on the stereochemical integrity of the final product.
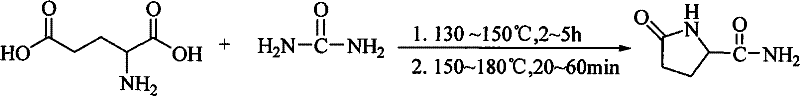
Mechanistic Insights into Solvent-Free Thermal Cyclization
The core of this technological advancement lies in the precise thermal management of the reaction mixture, which facilitates a complex cascade of dehydration and cyclization events. Initially, heating the mixture of L-glutamic acid and urea to the range of 130°C to 150°C promotes the formation of an intermediate ureide species through nucleophilic attack of the amino group on the carbonyl carbon of urea. As the temperature is subsequently elevated to the 150°C to 180°C range, intramolecular cyclization occurs, driven by the elimination of water and ammonia molecules to form the stable five-membered pyrrolidone ring characteristic of L-pyroglutamine. The solvent-free environment plays a critical role here by shifting the equilibrium towards product formation through the continuous removal of volatile by-products, effectively acting as a driving force for the reaction completion. This thermal protocol ensures that the chiral center at the C-5 position remains intact, preserving the optical activity required for downstream pharmaceutical applications where enantiomeric purity is paramount.
Impurity control in this system is achieved through a combination of stoichiometric optimization and post-reaction purification strategies. The patent specifies a molar ratio of glutamic acid to urea between 1:1.0 and 1:1.5, which is carefully calibrated to minimize the formation of biuret or other urea-derived side products while ensuring complete consumption of the more expensive amino acid. Following the thermal reaction, the crude product is subjected to an aqueous workup involving dissolution and treatment with activated carbon. This step is crucial for adsorbing colored impurities and trace organic by-products that may have formed during the high-temperature phase. The subsequent filtration and vacuum evaporation yield a product with exceptional purity, often exceeding 99%, demonstrating that the simplicity of the reaction conditions does not come at the expense of product quality. This rigorous control over the impurity profile makes the process highly attractive for high-purity L-pyroglutamine production standards.
How to Synthesize L-Pyroglutamine Efficiently
The synthesis of L-pyroglutamine via this patented solvent-free method offers a straightforward yet highly effective protocol for laboratory and industrial scale production. The process begins with the precise weighing and mixing of L-glutamic acid and urea, followed by a controlled heating regimen that transitions through two specific temperature plateaus to maximize yield and minimize degradation. The simplicity of the equipment requirements—essentially a heated reactor with stirring capability—makes this route accessible for rapid deployment. For detailed operational parameters and safety considerations, the standardized synthesis steps are outlined below to guide technical teams in replicating these results.
- Mix L-glutamic acid and urea in a molar ratio of 1: 1.0 to 1:1.5 in a reaction vessel.
- Heat the mixture to 130°C-150°C for 2-5 hours, then increase temperature to 150°C-180°C for 20-60 minutes.
- Dissolve the crude product in water, treat with activated carbon, filter, and evaporate to obtain pure L-pyroglutamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free technology translates into tangible strategic advantages that extend beyond mere chemical efficiency. The elimination of organic solvents removes a major variable from the supply chain, mitigating risks associated with solvent price volatility and availability disruptions. Furthermore, the simplified workflow reduces the number of unit operations required, leading to shorter batch cycles and increased throughput capacity within existing infrastructure. This operational leanness allows manufacturers to maintain lower inventory levels of raw materials while responding more quickly to customer orders, thereby enhancing overall supply chain resilience. The ability to source commodity chemicals like urea and glutamic acid, which are produced on a massive global scale, ensures a stable and reliable pharmaceutical intermediate supplier network that is less susceptible to geopolitical or logistical shocks compared to specialized reagent supply chains.
- Cost Reduction in Manufacturing: The most significant economic benefit arises from the complete removal of organic solvents, which eliminates the capital and operational expenditures associated with solvent storage, recovery distillation columns, and VOC abatement systems. Additionally, the high atom economy of the reaction means that a greater proportion of the input mass is converted into saleable product, reducing the cost per kilogram of the final API intermediate. The energy consumption is also optimized due to the shorter reaction times and the absence of energy-intensive solvent evaporation steps, contributing to substantial utility savings. These cumulative factors result in a drastically simplified cost structure that provides a competitive edge in pricing negotiations with downstream pharmaceutical clients.
- Enhanced Supply Chain Reliability: By relying on L-glutamic acid and urea, both of which are bulk commodities with mature and diversified global supply bases, manufacturers can secure long-term supply contracts at stable prices. This contrasts sharply with traditional methods that might require specialized esters or coupling agents subject to tighter market constraints. The robustness of the reaction conditions also implies that the process is less sensitive to minor variations in raw material quality, further stabilizing production schedules. Consequently, this leads to reducing lead time for high-purity amino acid derivatives, ensuring that customers receive their orders consistently without unexpected delays caused by raw material shortages or complex synthesis failures.
- Scalability and Environmental Compliance: The solvent-free nature of this process inherently aligns with green chemistry principles, making it easier to obtain environmental permits and maintain compliance with increasingly stringent regulations regarding industrial emissions. Scaling up from pilot to commercial production is facilitated by the lack of solvent handling issues, such as heat transfer limitations in large vessels or explosion risks, allowing for seamless commercial scale-up of complex chiral intermediates. The reduced waste generation, particularly the absence of saline wastewater from neutralization steps common in older methods, lowers waste disposal costs and minimizes the environmental impact, enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solvent-free synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production lines.
Q: What are the primary advantages of the solvent-free method for L-pyroglutamine synthesis?
A: The solvent-free method eliminates the need for expensive organic solvents and complex recovery systems, significantly reducing environmental pollution and production costs while maintaining high yields above 90%.
Q: How does this process compare to traditional esterification-cyclization routes?
A: Traditional routes involve multiple steps including acidification and esterification which require harsh reagents. The new thermal cyclization method simplifies this to a one-pot reaction, drastically shortening reaction time and improving operational safety.
Q: What purity levels can be achieved with this patented technology?
A: Through optimized temperature staging and activated carbon treatment, the process consistently achieves purity levels exceeding 99%, suitable for high-grade pharmaceutical and cosmetic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Pyroglutamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the solvent-free synthesis route for L-pyroglutamine and have integrated similar advanced methodologies into our manufacturing portfolio. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our facilities are equipped with state-of-the-art reactors capable of precise temperature control required for the two-stage thermal process, and our stringent purity specifications guarantee that every batch meets the rigorous demands of the pharmaceutical industry. With our rigorous QC labs and commitment to continuous improvement, we deliver high-quality intermediates that support your drug development and commercialization goals.
We invite you to collaborate with us to leverage this efficient technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this solvent-free approach can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of L-pyroglutamine for your global operations.
