Advanced Synthesis of Sila-Dehydroherbalone: Enabling Next-Generation Fragrance Intermediates
The fragrance and flavor industry is constantly evolving, driven by the demand for novel olfactory profiles and stable, synthetic alternatives to natural extracts. A significant breakthrough in this domain is documented in patent CN103524548A, which details the synthesis of Sila-Dehydroherbalone (1-(3-sila-3,3-dimethylcyclohex-1-enyl)ethanone). This compound represents a sophisticated application of the Si/C exchange strategy, where a quaternary carbon atom in a traditional fragrance skeleton is replaced by a silicon atom. This structural modification not only imparts a unique herbal and arborvitae aroma but also serves as a critical precursor for high-value organosilicon fragrances such as Herbal Ketone, Roman Musk, and Hevre Musk. The technology offers a robust pathway for creating stable, high-performance fragrance ingredients that overcome the seasonal dependencies and supply volatility associated with natural essential oils.
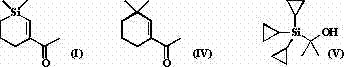
For R&D directors and procurement specialists, understanding the structural nuances is vital. The introduction of the silicon atom alters the electronic and steric properties of the molecule, potentially enhancing its diffusion and longevity in fragrance formulations. As a reliable synthetic flavors & fragrances supplier, leveraging this patented chemistry allows for the development of exclusive scent profiles that differentiate products in a crowded marketplace. The ability to synthesize this specific silicon-analog efficiently opens doors to a new class of fine chemicals that bridge the gap between traditional organic synthesis and advanced organometallic chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of gem-dimethylcyclohexene skeletons, which are ubiquitous in fragrance chemistry, often involves complex multi-step sequences requiring harsh conditions or expensive chiral auxiliaries. Conventional routes to analogs of Dehydroherbac may suffer from low atom economy, difficult purification processes due to closely related byproducts, and reliance on scarce natural precursors. Furthermore, achieving high stereochemical purity without resorting to costly resolution techniques has historically been a bottleneck. These limitations result in extended lead times and inflated production costs, making it challenging to maintain a consistent supply chain for high-volume fragrance manufacturing. The lack of efficient methods to introduce silicon into these specific frameworks has further restricted the exploration of silicon-based fragrance variants.
The Novel Approach
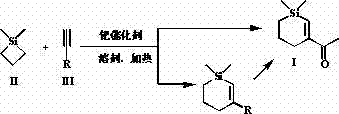
The methodology presented in CN103524548A revolutionizes this landscape by employing a palladium-catalyzed ring-expansion reaction. Instead of building the ring from scratch, this approach utilizes a strained four-membered silacyclobutane ring which undergoes insertion with a terminal alkyne. This strategy dramatically simplifies the synthetic route, reducing the number of operational steps and minimizing waste generation. By using commercially available 1,1-dimethylsilacyclobutane and simple terminal alkynes like 3-butyn-2-one, the process achieves high separation yields under relatively mild thermal conditions. This novel approach not only enhances cost reduction in synthetic flavors & fragrances manufacturing but also ensures a more predictable and scalable production workflow, addressing key pain points for supply chain managers.
Mechanistic Insights into Palladium-Catalyzed Ring Expansion
The core of this technology lies in the transition metal-catalyzed activation of the silicon-carbon bond within the silacyclobutane ring. Under the influence of palladium catalysts such as tetrakis(triphenylphosphine)palladium(0) or bis(triphenylphosphine)palladium(II) chloride, the strained Si-C bond undergoes oxidative addition. This generates a reactive palladium-silyl species that facilitates the insertion of the unsaturated terminal alkyne. The subsequent reductive elimination step closes the six-membered ring, effectively expanding the four-membered precursor into the desired cyclohexene derivative. This mechanism is highly advantageous because it tolerates various functional groups on the alkyne, allowing for the synthesis of diverse derivatives beyond just the ketone target.
From an impurity control perspective, the regioselectivity of the alkyne insertion is critical. The patent data suggests that the reaction conditions can be tuned to favor the formation of the desired 1,3-substituted product over other isomers. The use of specific ligands and solvents plays a pivotal role in stabilizing the catalytic cycle and preventing side reactions such as polymerization of the alkyne or decomposition of the silacycle. Understanding these mechanistic details allows process chemists to optimize catalyst loading (typically 0.5% to 10%) and reaction temperatures (ranging from 25°C to 200°C) to maximize throughput while maintaining the stringent purity specifications required for fragrance applications. This level of control is essential for ensuring batch-to-batch consistency in commercial production.
How to Synthesize Sila-Dehydroherbalone Efficiently
The patent outlines two primary strategies for executing this synthesis: a direct one-pot method and a two-step sequence involving an ester intermediate. The one-pot method is particularly attractive for large-scale operations due to its operational simplicity. It involves mixing the silacyclobutane and 3-butyn-2-one in a solvent like benzene or toluene, adding the catalyst, and heating until conversion is complete. Alternatively, the two-step route offers flexibility if different alkyne precursors are used, allowing for a Grignard modification in the second step to install the acetyl group. Both routes have been demonstrated to produce the target molecule with high purity (>98%) after standard silica gel chromatography.
- Mix 1,1-dimethylsilacyclobutane and a terminal alkyne (e.g., 3-butyn-2-one) in a dry organic solvent like benzene or toluene.
- Add a palladium catalyst such as Pd(PPh3)4 or (PPh3)2PdCl2 (0.5%-10% molar ratio) and heat the mixture to reflux (approx. 80-90°C).
- After reaction completion (1-40 hours), concentrate the mixture and purify via silica gel column chromatography to obtain the pure product with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits. The reliance on commodity chemicals like silacyclobutane and simple alkynes reduces dependency on specialized, hard-to-source intermediates. This shift significantly mitigates supply risk and stabilizes raw material costs. Furthermore, the robustness of the palladium-catalyzed system means that the process is less sensitive to minor fluctuations in reaction parameters, leading to higher overall equipment effectiveness (OEE) in a manufacturing setting. The simplified downstream processing, characterized by straightforward concentration and chromatography, reduces the burden on purification units and shortens the overall production cycle time.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the use of inexpensive, commercially available starting materials directly translate to lower variable costs. By avoiding the need for exotic reagents or cryogenic conditions, the process energy consumption is also optimized. The high isolated yields reported in the patent examples indicate efficient material utilization, minimizing waste disposal costs and maximizing the output per batch. This economic efficiency is crucial for maintaining competitive pricing in the global fragrance market.
- Enhanced Supply Chain Reliability: Sourcing 1,1-dimethylsilacyclobutane and terminal alkynes is significantly more reliable than sourcing complex natural extracts or highly specialized chiral building blocks. The chemical stability of these reagents allows for long-term storage and bulk purchasing, enabling better inventory management. Additionally, the scalability of the reaction from gram to kilogram scales without significant re-optimization ensures that supply can be ramped up quickly to meet market demand spikes, reducing lead time for high-purity fragrance intermediates.
- Scalability and Environmental Compliance: The reaction conditions described, such as heating in toluene or THF, are compatible with standard stainless steel reactors found in most fine chemical plants. The process avoids the use of highly toxic heavy metals beyond the catalytic amount of palladium, which can be recovered and recycled, aligning with modern green chemistry principles. The reduction in solvent usage and waste generation compared to traditional routes supports corporate sustainability goals and simplifies regulatory compliance regarding environmental discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Sila-Dehydroherbalone. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this chemistry into their existing product portfolios.
Q: What are the optimal catalyst systems for this ring expansion?
A: The patent specifies several effective palladium systems including Pd(PPh3)4, (PPh3)2PdCl2, and Pd(OAc)2 combined with triphenylphosphine ligands, allowing flexibility based on cost and availability.
Q: Can this process be scaled for industrial fragrance production?
A: Yes, the method utilizes commercially available raw materials like 1,1-dimethylsilacyclobutane and operates under standard heating conditions (25-200°C), making it highly suitable for commercial scale-up.
Q: What is the purity profile of the synthesized Sila-Dehydroherbalone?
A: Experimental data indicates that following the purification steps, the final product achieves a purity greater than 98%, meeting stringent requirements for fine fragrance applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sila-Dehydroherbalone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of organosilicon chemistry in the fragrance sector. Our team of expert process chemists is well-versed in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN103524548A can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Sila-Dehydroherbalone meets the exacting standards required by top-tier fragrance houses. Our commitment to quality and consistency makes us a trusted partner for developing next-generation scent profiles.
We invite you to explore how this advanced synthesis route can enhance your product line. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and sourcing strategies. Let us collaborate to bring this unique herbal and arborvitae aroma to your market with speed and precision.
