Revolutionizing FDCA Production: Mild Single-Atom Catalysis for Commercial Scale-Up of Complex Polymer Additives
The chemical industry is currently witnessing a paradigm shift towards sustainable manufacturing, particularly in the production of bio-based platform chemicals like 2,5-Furandicarboxylic Acid (FDCA). A groundbreaking approach detailed in patent CN111138392B introduces a highly efficient method for converting furfural directly into FDCA using transition metal single-atom catalysts. This technology addresses critical bottlenecks in existing supply chains by utilizing molecular oxygen and carbon dioxide as benign reagents under remarkably mild conditions. For R&D directors and procurement specialists seeking a reliable polymer additive supplier, this innovation represents a significant leap forward in process intensification. By leveraging illumination-assisted catalyst preparation and low-temperature oxidative carbonylation, the process achieves yields exceeding 85% while minimizing environmental impact. This report analyzes the technical feasibility and commercial implications of adopting this novel synthetic route for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of FDCA has been plagued by severe thermodynamic and kinetic barriers that hinder cost-effective production. The most common pathway involves the oxidation of 5-hydroxymethylfurfural (HMF), which, despite high conversion rates, suffers from complex catalyst requirements and difficult product separation issues that compromise overall process integration. Alternative routes involving furoic acid often rely on the Henkel reaction or carbonylation processes that demand extreme temperatures ranging from 260°C to 285°C. Such harsh operating conditions not only result in excessive energy consumption but also lead to significant safety risks and equipment degradation over time. Furthermore, traditional methods frequently utilize stoichiometric oxidants or generate substantial amounts of hazardous waste, creating a heavy burden on downstream purification and waste treatment facilities. These inefficiencies make it challenging to achieve the economies of scale necessary for FDCA to effectively compete with petroleum-derived terephthalic acid in the global polymer market.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN111138392B utilizes a transition metal single-atom catalyst supported on graphene to facilitate direct conversion at temperatures as low as 90-120°C. This approach fundamentally alters the reaction landscape by activating molecular oxygen and carbon dioxide simultaneously, thereby eliminating the need for harsh thermal activation or toxic oxidizing agents. The use of single-atom catalysis ensures maximum atom efficiency, as every metal atom participates in the reaction, drastically reducing the required catalyst loading compared to bulk metal counterparts. Additionally, the process operates at moderate pressures of 0.1-1.0 MPa, which simplifies reactor design and lowers capital expenditure for high-pressure containment systems. By streamlining the synthesis into a direct oxidative carbonylation step, this novel approach offers a streamlined pathway that is inherently safer, more energy-efficient, and environmentally superior to conventional multi-step syntheses.
Mechanistic Insights into Single-Atom Catalyzed Oxidative Carbonylation
The core of this technological breakthrough lies in the unique electronic structure of the transition metal single-atom catalysts, such as Pd, Ni, or Cu, anchored on a graphene support. The illumination-assisted preparation method ensures that metal ions are reduced and dispersed at the atomic level, creating highly active sites that facilitate the adsorption and activation of small gas molecules like O2 and CO2. During the reaction, the aldehyde group of furfural is oxidized while simultaneously undergoing carbonylation, a dual transformation that is kinetically challenging under standard conditions. The graphene support plays a crucial role in stabilizing these single atoms and preventing aggregation, thereby maintaining high catalytic activity over extended reaction times of 15-20 hours. This mechanism allows for the precise insertion of carboxyl groups into the furan ring without disrupting the aromatic stability of the heterocycle, ensuring high selectivity towards the desired 2,5-isomer.
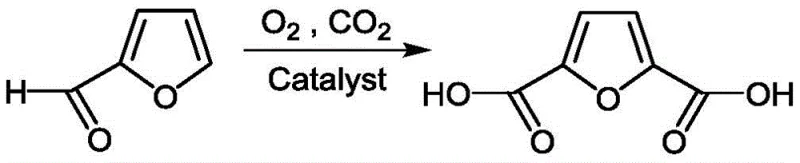
Furthermore, the impurity profile of the resulting FDCA is significantly improved due to the mild reaction environment which suppresses side reactions such as ring opening or polymerization that are common at higher temperatures. The specific interaction between the single metal atoms and the reactants lowers the activation energy barrier, enabling the reaction to proceed efficiently at 90-120°C rather than the 260°C required by traditional disproportionation methods. This precise control over the reaction pathway minimizes the formation of by-products, simplifying the subsequent purification steps and enhancing the overall purity of the final crystalline product. For quality control teams, this means a more consistent product specification with fewer unknown impurities, which is critical for applications in high-performance polymers and pharmaceutical intermediates where trace contaminants can affect material properties.
How to Synthesize 2,5-Furandicarboxylic Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing high-purity FDCA suitable for commercial applications. The process begins with the photochemical preparation of the catalyst, followed by a one-pot oxidative carbonylation in an autoclave system. Detailed operational parameters regarding temperature, pressure, and gas ratios are critical to achieving the reported yields of over 85%. The following guide summarizes the standardized procedure derived from the patent examples, ensuring reproducibility and safety during scale-up operations. Please refer to the structured steps below for the complete technical workflow.
- Prepare the transition metal single-atom catalyst by dispersing graphene in water, adding transition metal salt, and irradiating with a light source followed by freeze-drying.
- Load the catalyst and furfural into an autoclave, introduce oxygen and carbon dioxide (2: 1 ratio), and react at 90-120°C and 0.1-1.0 MPa for 15-20 hours.
- Dissolve the product in ethyl acetate, filter to recover the catalyst, and purify the filtrate via reduced pressure rectification to obtain high-purity FDCA.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this single-atom catalytic route offers transformative benefits that extend beyond mere technical novelty. The ability to operate at significantly lower temperatures translates directly into reduced utility costs and lower maintenance requirements for reactor vessels, addressing the critical need for cost reduction in polymer additive manufacturing. Moreover, the use of abundant and inexpensive feedstocks like furfural, combined with gaseous reagents like oxygen and carbon dioxide, mitigates the risk associated with volatile liquid reagent pricing. The simplicity of the workup procedure, which involves filtration and distillation, reduces the complexity of the production line and shortens the batch cycle time. These factors collectively enhance the reliability of the supply chain by making the production process less susceptible to disruptions caused by equipment failure or raw material scarcity.
- Cost Reduction in Manufacturing: The elimination of high-temperature requirements and the use of earth-abundant transition metals significantly lower the operational expenditure associated with energy and catalyst procurement. By avoiding the need for expensive noble metal catalysts in bulk form or complex multi-step synthesis sequences, the overall cost of goods sold is drastically simplified. The high atom economy of using CO2 and O2 means that waste disposal costs are minimized, contributing to substantial cost savings in environmental compliance and waste management. This economic efficiency makes the process highly competitive against established petrochemical routes.
- Enhanced Supply Chain Reliability: Utilizing furfural, a biomass-derived platform chemical, diversifies the raw material base away from fossil fuel dependency, ensuring greater long-term stability in sourcing. The robust nature of the graphene-supported catalyst allows for easy recovery and potential reuse, reducing the frequency of catalyst replenishment orders. This resilience against raw material volatility is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream customers in the packaging and textile industries.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of hazardous solvents during the reaction phase simplify the engineering requirements for scaling up from pilot to commercial production. The process aligns perfectly with green chemistry principles by utilizing non-toxic gases and generating minimal waste, facilitating easier regulatory approval in regions with strict environmental standards. This sustainability profile enhances the marketability of the final FDCA product to eco-conscious brands seeking bio-based alternatives for their polymer supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative carbonylation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide clarity on process capabilities. Understanding these details is essential for evaluating the feasibility of integrating this method into existing manufacturing infrastructures.
Q: What are the key advantages of the single-atom catalyst method over traditional FDCA synthesis?
A: Unlike traditional routes requiring harsh temperatures up to 260°C or complex multi-step sequences, this method operates at mild conditions (90-120°C) with high atom economy using O2 and CO2, significantly reducing energy consumption and waste generation.
Q: Can the catalyst be recovered and reused in this process?
A: Yes, the solid catalyst can be easily recovered by simple filtration after the reaction mixture is dissolved in solvent, allowing for potential recycling and further reducing the cost of goods sold.
Q: What yields can be expected from this oxidative carbonylation route?
A: Experimental data indicates that yields consistently exceed 85%, with specific examples demonstrating yields between 85.6% and 90.1% depending on the specific transition metal used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Furandicarboxylic Acid Supplier
As the global demand for bio-based polymers continues to surge, securing a stable supply of high-quality monomers like FDCA is paramount for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging advanced catalytic technologies to deliver superior chemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for high-performance applications in plastics and pharmaceuticals.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your strategic goals for sustainable growth and operational excellence.
