Advanced Synthesis of Indoxacarb Key Intermediates: Overcoming Azeotropic Limitations for Commercial Scale
The chemical manufacturing landscape for high-value agrochemical intermediates is constantly evolving, driven by the dual imperatives of regulatory compliance and cost efficiency. A pivotal advancement in this domain is documented in patent CN111269194B, which details a novel preparation method for a key intermediate of indoxacarb, specifically 7-chloroindeno[1,2-e][1,3,4]oxadiazine-2,4a(3H,5H)-dicarboxylic acid-4a-methyl ester-2-benzyl ester. This technology addresses longstanding inefficiencies in the cyclization step of indoxacarb synthesis, a critical pathway for producing one of the world's most effective oxadiazine insecticides. By substituting traditional diethoxymethane reagents with paraformaldehyde substances, the disclosed method fundamentally alters the reaction thermodynamics and byproduct profile. This shift not only enhances the chemical purity of the final intermediate but also resolves complex solvent separation issues that have historically plagued large-scale production facilities. For global supply chain leaders and R&D directors, understanding this mechanistic pivot is essential for evaluating next-generation sourcing strategies that prioritize both yield optimization and environmental sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
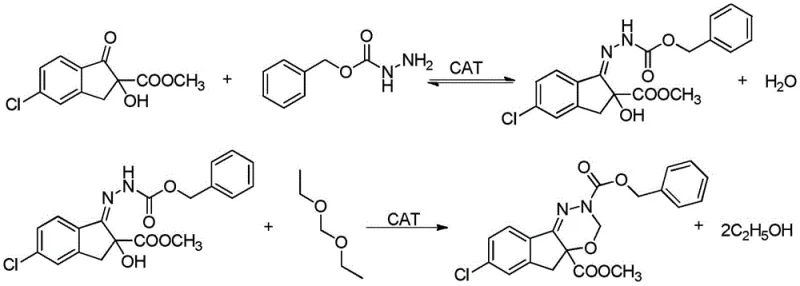
Historically, the industrial synthesis of this critical indoxacarb precursor has relied heavily on a two-step sequence involving an initial condensation followed by a cyclization using diethoxymethane. While chemically feasible, this legacy approach suffers from significant thermodynamic and kinetic drawbacks that compromise overall process efficiency. As illustrated in the reaction scheme, the initial condensation between 5-chloro-2-methoxycarbonyl-2-hydroxy-1-indenone and hydrazinoformate benzyl ester is reversible, requiring rigorous water removal to drive equilibrium toward the intermediate. However, the subsequent cyclization step introduces a more severe complication: the generation of ethanol as a stoichiometric byproduct. In the acidic environment required for catalysis, this ethanol does not remain inert; instead, it actively participates in transesterification side reactions with the methyl ester moiety of the product. This leads to the formation of ethyl ester impurities, which are structurally similar to the target molecule and notoriously difficult to separate via standard crystallization techniques. Furthermore, the presence of ethanol creates a complex solvent matrix when combined with unreacted diethoxymethane.
The formation of these ethyl ester impurities is not merely a quality control nuisance; it represents a substantial yield loss mechanism that directly impacts the cost of goods sold. The patent data indicates that under conventional conditions, the content of the desired intermediate hovers around 94%, with yields struggling to exceed 85%. Beyond the chemical impurities, the physical separation of solvents presents a formidable engineering challenge. Ethanol and diethoxymethane form a tight azeotropic mixture that is extremely difficult to break using standard distillation columns. This azeotrope cannot be easily recycled back into the process, forcing manufacturers to treat large volumes of mixed solvent as hazardous waste. This not only inflates raw material costs due to the inability to recover valuable reagents but also imposes a heavy burden on wastewater treatment facilities. Consequently, the conventional route is characterized by high operational expenditure, significant environmental footprint, and a ceiling on achievable purity that limits its viability for premium-grade agrochemical applications.
The Novel Approach
In stark contrast to the legacy methodology, the innovative process described in CN111269194B leverages paraformaldehyde as the methylene source for the cyclization reaction, effectively decoupling the ring-closing step from ethanol generation. By utilizing paraformaldehyde, which depolymerizes in situ to release formaldehyde, the reaction pathway bypasses the formation of alcohol byproducts entirely. This strategic substitution eliminates the root cause of the transesterification side reaction, thereby preserving the integrity of the methyl ester group on the indoxacarb scaffold. The result is a dramatic suppression of ethyl ester impurities, with patent data demonstrating a reduction in impurity levels from approximately 5% down to below 0.1%. This leap in chemical selectivity translates directly to a product content exceeding 97% and yields surpassing 95%, representing a significant upgrade in process performance. Moreover, the solvent system is simplified to a single organic phase, typically toluene, which facilitates straightforward water removal via azeotropic distillation without generating unrecoverable mixed solvent wastes.
Mechanistic Insights into Paraformaldehyde-Mediated Cyclization
The core innovation of this synthesis lies in the controlled depolymerization of paraformaldehyde within the reaction medium. Paraformaldehyde serves as a stable, solid polymeric source of formaldehyde (CH2O)n. Upon introduction into the heated toluene suspension, typically maintained between 95°C and 100°C, the polymer chains undergo thermal depolymerization to release monomeric formaldehyde gas directly into the solution. This in situ generation ensures a steady, controlled concentration of the electrophilic methylene species, which then reacts with the hydrazone nitrogen of the condensation intermediate. The absence of ethoxy groups in the reagent structure is the critical differentiator; unlike diethoxymethane, which cleaves to release ethanol upon reacting, paraformaldehyde releases only water as the oxygenated byproduct. This water is continuously removed from the reaction system through azeotropic distillation with toluene, driving the equilibrium forward according to Le Chatelier's principle without introducing nucleophilic alcohols that could attack the ester functionality. This mechanistic elegance ensures that the reaction proceeds with high atom economy regarding the carbon skeleton while minimizing side reactions.
From an impurity control perspective, this mechanism offers a robust defense against the formation of analogues that complicate downstream processing. In the conventional route, the ethyl ester impurity possesses polarity and solubility characteristics very similar to the target methyl ester, often requiring multiple recrystallizations or expensive chromatographic steps to remove. By preventing the formation of this impurity at the source, the new process simplifies the purification train significantly. The patent specifies that after the cyclization is complete and the acid catalyst is washed out, the crude product can be recrystallized directly from solvents like methyl tert-butyl ether (MTBE) or methanol to achieve high purity. The stability of the reflux temperature during the cyclization phase, maintained steadily at 95-100°C, further contributes to batch-to-batch consistency. This thermal stability is crucial for industrial implementation, as it prevents localized hot spots that could lead to decomposition or alternative degradation pathways, ensuring that the impurity profile remains predictable and manageable across multi-ton scales.
How to Synthesize 7-chloroindeno[1,2-e][1,3,4]oxadiazine Derivatives Efficiently
Implementing this advanced synthesis route requires precise control over reaction parameters, particularly regarding the preparation of the paraformaldehyde suspension and the management of water removal. The process begins with the formation of the condensation intermediate, where stoichiometry and water separation are paramount. Following this, the handling of paraformaldehyde demands attention to particle size and suspension stability to ensure consistent depolymerization rates. The dropwise addition of the paraformaldehyde-toluene suspension into the refluxing intermediate solution must be carefully timed, typically over a period of 2 to 5 hours, to match the rate of water removal. This synchronization prevents the accumulation of unreacted formaldehyde while maintaining the driving force for cyclization. The detailed operational protocol involves specific temperature ramps, vacuum levels for solvent recovery, and crystallization conditions that are critical for maximizing yield. For a comprehensive understanding of the exact procedural nuances required to replicate these high-yield results in a GMP environment, please refer to the standardized synthesis guide below.
- Condense 5-chloro-2-methoxycarbonyl-2-hydroxy-1-indenone with benzyl hydrazinoformate in toluene using an acid catalyst like oxalic acid, removing water via azeotropic distillation.
- Prepare a suspension of low-polymerization degree paraformaldehyde (n=5-8) in toluene at 50-60°C to ensure uniform dispersion before addition.
- Slowly add the paraformaldehyde suspension to the refluxing condensation intermediate at 95-100°C, continuously separating generated water until the system is dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this paraformaldehyde-based route offers tangible strategic advantages that extend beyond mere chemical yield. The primary economic driver is the drastic simplification of solvent management and waste disposal. In the conventional process, the generation of the ethanol-diethoxymethane azeotrope creates a waste stream that is costly to dispose of and impossible to recycle, representing a direct loss of raw material value. By eliminating this mixed solvent issue, the new method allows for the near-complete recovery and reuse of the toluene solvent. This closed-loop solvent capability significantly reduces the recurring cost of purchasing fresh solvents and lowers the volume of hazardous waste requiring third-party treatment. Furthermore, the enhancement in product purity from roughly 94% to over 97% reduces the burden on quality control laboratories and minimizes the risk of batch rejection due to out-of-specification impurity profiles. This reliability is invaluable for maintaining continuous production schedules in high-volume agrochemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of ethanol byproducts removes the need for complex separation technologies designed to break azeotropes, thereby lowering capital expenditure on distillation infrastructure. Additionally, the suppression of ethyl ester impurities means that less material is lost to mother liquors during crystallization, effectively increasing the mass balance of the process. The ability to use recovered toluene without extensive purification further drives down variable costs, making the overall cost of goods sold significantly more competitive compared to legacy methods. The reduction in impurity levels also implies a potential reduction in the number of recrystallization steps required, saving both time and energy utilities.
- Enhanced Supply Chain Reliability: Sourcing paraformaldehyde is generally more stable and logistically simpler than managing volatile liquid reagents like diethoxymethane, which may be subject to stricter transportation regulations due to flammability and volatility. The robustness of the reaction conditions, operating at standard reflux temperatures without the need for extreme vacuum or cryogenic cooling, ensures that the process is less susceptible to equipment failures or utility fluctuations. This operational resilience translates to more predictable lead times and a lower risk of production delays. For global buyers, this means a more secure supply of high-purity indoxacarb intermediates, mitigating the risk of shortages that can disrupt the formulation of finished pesticide products.
- Scalability and Environmental Compliance: As environmental regulations regarding volatile organic compounds (VOCs) and wastewater discharge become increasingly stringent, this process offers a clear compliance advantage. The reduction in mixed solvent waste and the ability to recycle toluene align perfectly with green chemistry principles and corporate sustainability goals. The process generates minimal aqueous waste, primarily consisting of the water produced during the reaction and the wash water for catalyst removal, which is far easier to treat than solvent-laden effluent. This environmental friendliness facilitates smoother permitting processes for new manufacturing lines and reduces the long-term liability associated with hazardous waste storage and disposal, making it an ideal candidate for sustainable commercial scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the technology's capabilities. Understanding these specifics is crucial for technical teams evaluating the feasibility of technology transfer or for procurement specialists negotiating supply agreements based on quality specifications. The answers highlight the distinct advantages of the paraformaldehyde route over traditional methods, focusing on purity, yield, and operational safety.
Q: Why is paraformaldehyde preferred over diethoxymethane for indoxacarb intermediate synthesis?
A: Paraformaldehyde eliminates the generation of ethanol as a byproduct, thereby preventing transesterification side reactions that form difficult-to-remove ethyl ester impurities. Additionally, it avoids the formation of ethanol-diethoxymethane azeotropes, simplifying solvent recovery.
Q: What represents the critical quality attribute in this synthesis route?
A: The critical quality attribute is the suppression of ethyl ester impurities. By avoiding ethanol generation, the process maintains product content above 97% compared to roughly 94% in conventional methods, significantly reducing downstream purification burdens.
Q: How does this process impact environmental compliance and waste management?
A: The process drastically reduces environmental pressure by eliminating mixed solvent waste streams. Unlike the prior art which produces unrecoverable ethanol-diethoxymethane azeotropes, this method allows for the complete recycling of the toluene solvent after simple phase separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-chloroindeno[1,2-e][1,3,4]oxadiazine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO specializing in complex agrochemical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific thermal and separation requirements of the paraformaldehyde cyclization process, ensuring that the theoretical benefits of patent CN111269194B are fully realized in commercial batches. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every shipment of indoxacarb intermediate meets the highest standards of quality, free from the ethyl ester impurities that plague older manufacturing methods. Our commitment to process excellence ensures that our clients receive a product that not only meets but exceeds industry benchmarks for consistency and reliability.
We invite forward-thinking agrochemical companies to collaborate with us to leverage this advanced technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this greener, more efficient route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments tailored to your production needs. Let us help you secure a sustainable, high-quality supply of indoxacarb intermediates that positions your business for long-term success in the global market.
