Advanced Room-Temperature Synthesis of 9-Aryl Fluorene for Commercial OLED Production
Advanced Room-Temperature Synthesis of 9-Aryl Fluorene for Commercial OLED Production
The rapid evolution of the organic light-emitting diode (OLED) industry demands intermediates that combine high purity with scalable manufacturing processes. Patent CN103724151A introduces a groundbreaking method for synthesizing 9-aryl fluorene compounds, which serve as critical building blocks for high-performance photoelectric materials. This technology utilizes a novel cascade reaction involving 2-aryl benzaldehyde and aromatic hydrocarbons, catalyzed by trifluoromethanesulfonic acid and acetic anhydride. Unlike traditional methods that rely on harsh conditions or expensive transition metals, this approach operates efficiently at room temperature, offering a pathway to substantial cost reduction in electronic chemical manufacturing. The structural versatility of these compounds allows for the fine-tuning of thermal stability and luminescent efficiency, making them indispensable for next-generation display technologies. 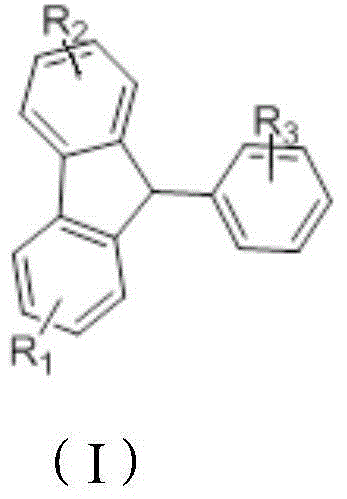
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9-aryl fluorene derivatives has been plagued by significant operational hurdles that hinder industrial adoption. Conventional routes, such as those utilizing Grignard reagents, necessitate strictly anhydrous and oxygen-free environments, which drastically increase infrastructure costs and safety risks. Furthermore, the post-treatment of Grignard reaction waste liquids is complex and environmentally burdensome due to the toxicity of byproducts. Alternative methods involving palladium catalysis require temperatures as high as 130°C and utilize expensive catalysts like palladium acetate, which not only inflate raw material costs but also introduce challenges in removing trace heavy metal impurities. These factors collectively render many existing processes unsuitable for the rigorous demands of commercial scale-up of complex polymer additives and electronic materials.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by employing a one-pot cascade reaction that bypasses the need for intermediate isolation. By leveraging trifluoromethanesulfonic acid and acetic anhydride, the reaction proceeds smoothly at room temperature, typically between 20-30°C, for a duration of 1 to 30 hours. This mild condition eliminates the energy expenditure associated with heating and cooling cycles, directly contributing to cost reduction in OLED material manufacturing. The process is robust across a wide range of substrates, including various substituted benzaldehydes and aromatic hydrocarbons like benzene or anthracene. The simplicity of the work-up, involving vacuum distillation followed by column chromatography, ensures that the final product meets stringent purity specifications without the need for complex purification trains.
Mechanistic Insights into Acid-Catalyzed Cascade Cyclization
The core of this synthetic innovation lies in its elegant mechanistic pathway, which avoids the formation of unstable intermediates that often degrade yield in traditional routes. The reaction initiates with the condensation of 2-aryl benzaldehyde with acetic anhydride to form an acetal compound in situ. This activated species then undergoes an intermolecular Friedel-Crafts reaction with the aromatic hydrocarbon component, generating a diaryl carbinol ester. Subsequently, an intramolecular Friedel-Crafts cyclization occurs, closing the fluorene ring system to yield the target 9-aryl fluorene. This seamless transition between reaction stages within a single vessel minimizes material loss and exposure to environmental contaminants, ensuring high reproducibility. The use of trifluoromethanesulfonic acid as a superacid catalyst enhances the electrophilicity of the intermediates, driving the reaction to completion even at ambient temperatures.
From a quality control perspective, this mechanism offers superior impurity management compared to metal-catalyzed alternatives. The absence of transition metals like palladium means there is no risk of metal leaching into the final product, a critical parameter for R&D directors focusing on the purity and杂质 profile of electronic chemicals. The reaction byproducts are primarily organic acids and anhydrides, which are easier to separate via distillation than inorganic metal salts. This cleanliness of the reaction profile simplifies the analytical validation process, allowing for faster batch release times. Furthermore, the ability to tune the substituents on the starting benzaldehyde allows for precise control over the electronic properties of the final fluorene derivative, facilitating the customization of materials for specific red, green, or blue emission requirements.
How to Synthesize 9-Phenylfluorene Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and mixing sequences to maximize the efficiency of the cascade process. The patent specifies that trifluoromethanesulfonic acid should be added at approximately 10% of the molar amount of the aldehyde, while acetic anhydride is used in a twofold molar excess. The reaction mixture is stirred briefly before the addition of the anhydride to ensure proper catalyst distribution. Detailed standardized synthesis steps see the guide below.
- Mix 2-aryl benzaldehyde and aromatic hydrocarbon in an organic solvent with trifluoromethanesulfonic acid catalyst.
- Add acetic anhydride to the mixture and stir at room temperature for 1 to 30 hours to complete the cascade reaction.
- Purify the crude product via vacuum distillation and column chromatography to obtain high-purity 9-aryl fluorene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthesis method offers tangible strategic benefits beyond mere technical feasibility. The elimination of expensive transition metal catalysts and the reduction of energy-intensive heating steps translate directly into a more favorable cost structure. The reliance on readily available commodity chemicals like acetic anhydride and common organic solvents ensures supply chain reliability, reducing the risk of production stoppages due to raw material shortages. Additionally, the mild reaction conditions enhance operational safety, potentially lowering insurance and compliance costs associated with high-pressure or high-temperature operations.
- Cost Reduction in Manufacturing: The removal of palladium catalysts and the avoidance of high-temperature reflux conditions significantly lower the variable costs per kilogram of production. By eliminating the need for specialized equipment capable of withstanding extreme conditions, capital expenditure is also optimized. The simplified purification process reduces solvent consumption and waste disposal fees, contributing to substantial cost savings over the lifecycle of the product. This economic efficiency makes the material more competitive in price-sensitive markets without compromising on quality.
- Enhanced Supply Chain Reliability: The use of stable, non-hazardous reagents that do not require special storage conditions like inert atmosphere gloveboxes simplifies logistics and inventory management. The robustness of the reaction against minor variations in temperature or moisture content ensures consistent batch-to-batch quality, reducing the rate of rejected lots. This reliability is crucial for maintaining continuous supply to downstream OLED manufacturers who operate on tight production schedules. Reducing lead time for high-purity electronic chemicals becomes achievable through streamlined processing.
- Scalability and Environmental Compliance: The one-pot nature of the reaction minimizes the generation of intermediate waste streams, aligning with increasingly stringent environmental regulations. The process is inherently scalable from laboratory benchtop to multi-ton reactors without significant re-engineering of the process parameters. This scalability ensures that supply can be ramped up quickly to meet surging demand in the display industry. The reduced environmental footprint also supports corporate sustainability goals, enhancing the brand value of the supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation.
Q: What are the advantages of this synthesis method over traditional Grignard routes?
A: Unlike Grignard reactions requiring anhydrous and oxygen-free conditions, this method operates at room temperature with mild conditions, significantly simplifying post-treatment and reducing toxic waste.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the one-pot cascade reaction eliminates intermediate isolation steps, reduces energy consumption, and uses readily available raw materials, making it highly scalable for commercial manufacturing.
Q: What is the typical yield range for 9-aryl fluorene derivatives using this patent?
A: The patent data indicates yields ranging from 28% to over 99%, depending on the specific substituents and aromatic hydrocarbons used, with many examples achieving greater than 99% yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Aryl Fluorene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of final electronic devices. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 9-aryl fluorene meets the exacting standards required for OLED applications. Our commitment to technical excellence allows us to support your R&D efforts with materials that enable superior device efficiency and longevity.
We invite you to collaborate with us to optimize your supply chain for photoelectric materials. Contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your product roadmap. Let us help you secure a stable supply of high-performance intermediates for your next-generation display projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
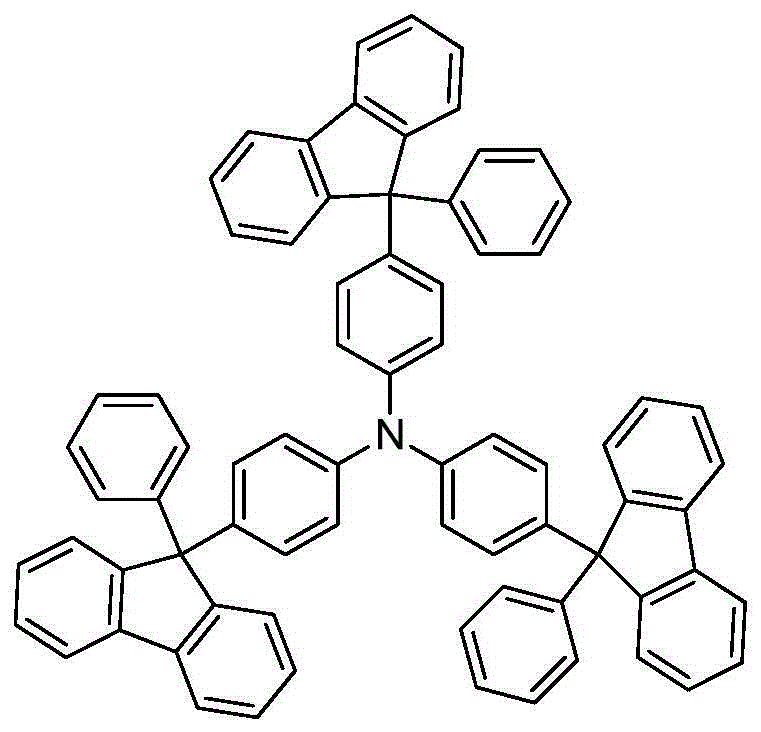
Understanding the downstream applications of these intermediates further highlights their value. For instance, 9-phenylfluorene can be oxidized and reacted to form tris[4-(9-phenylfluoren-9-yl)phenyl]amine (TFTPA), a material known for its excellent thermal and electrochemical stability.