Scalable Production of Ethynylnaphthalene Derivatives via Aqueous [4+2] Cyclization Technology
Scalable Production of Ethynylnaphthalene Derivatives via Aqueous [4+2] Cyclization Technology
The pharmaceutical and specialty chemical industries are constantly seeking robust, environmentally benign synthetic routes for complex polycyclic aromatic hydrocarbons. Patent CN113372219B introduces a groundbreaking methodology for the preparation of ethynylnaphthalene derivatives utilizing a [4+2] cyclization reaction where water serves as the primary solvent. This innovation represents a significant paradigm shift from traditional organic solvent-based systems, leveraging a heterogeneous Al-SBA-15 catalyst to drive the dehydration and subsequent cycloaddition in a seamless one-pot process. For R&D directors and process chemists, this technology offers a compelling solution to the challenges of waste generation and catalyst recovery, providing a streamlined pathway to high-value naphthalene scaffolds that are critical precursors for dyes, fluorescent materials, and bioactive molecules.
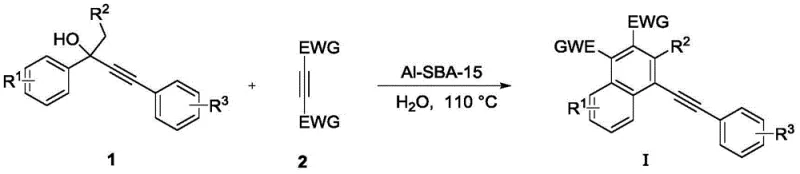
The core of this invention lies in its ability to merge two distinct chemical transformations—alkynol dehydration and Diels-Alder cycloaddition—into a single operational unit. By employing water not merely as a passive medium but as an active participant in the thermal management and phase separation of the reaction, the process achieves high atom economy. The resulting ethynylnaphthalene derivatives possess multiple reactive sites, making them versatile building blocks for the synthesis of indane compounds, furfural derivatives, and benzo[e][1,2]oxazinones. This versatility underscores the strategic value of mastering this synthesis route for any organization aiming to secure a reliable specialty chemical intermediates supplier capable of delivering complex fused-ring systems with high purity and consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted naphthalene derivatives has relied heavily on transition metal-catalyzed cyclizations or aromatic insertion reactions that often necessitate the use of volatile organic compounds (VOCs) such as dichloromethane, toluene, or DMF. These conventional protocols frequently suffer from significant drawbacks, including the requirement for stoichiometric amounts of expensive Lewis acids, difficult downstream processing to remove residual metal contaminants, and the generation of substantial hazardous waste streams. Furthermore, many traditional methods struggle with substrate tolerance, often failing when electron-withdrawing or sterically hindered groups are present on the aromatic rings. The reliance on homogeneous catalysts also complicates the recycling of catalytic species, leading to inflated operational expenditures and a larger environmental footprint that conflicts with modern green chemistry mandates.
The Novel Approach
In stark contrast, the methodology disclosed in CN113372219B utilizes a mesoporous aluminosilicate material, Al-SBA-15, which acts as a robust, reusable solid acid catalyst. This novel approach enables the reaction to proceed efficiently in water at elevated temperatures (90-130 °C), eliminating the need for toxic organic solvents during the critical cyclization step. The heterogeneous nature of the catalyst allows for simple filtration post-reaction, facilitating immediate catalyst recovery and reuse without significant loss of activity. This shift to an aqueous system not only drastically simplifies the workup procedure—requiring only phase separation and extraction—but also enhances the safety profile of the manufacturing process. By integrating dehydration and cyclization into a one-pot sequence, the new method reduces unit operations, minimizes solvent consumption, and delivers superior yields, exemplified by the 78% yield achieved in the synthesis of dimethyl 4-(phenylethynyl)naphthalene-1,2-dicarboxylate.
Mechanistic Insights into Al-SBA-15 Catalyzed [4+2] Cyclization
The mechanistic elegance of this transformation relies on the dual functionality of the Al-SBA-15 catalyst within the aqueous medium. Initially, the Lewis acid sites on the aluminosilicate surface promote the dehydration of the propargylic alcohol precursor (Compound 1), generating a reactive diene species in situ. This transient diene immediately undergoes a [4+2] cycloaddition with the electron-deficient alkyne dienophile (Compound 2, typically dimethyl or diethyl butynedioate). The hydrophobic effect of water likely plays a crucial role in accelerating this cycloaddition by forcing the organic reactants into closer proximity, thereby increasing the effective molarity and reaction rate. Following the formation of the non-aromatic cyclic intermediate, the catalyst further facilitates aromatization through dehydrogenation or elimination pathways, ultimately yielding the stable naphthalene core. This cascade mechanism ensures high selectivity and minimizes the formation of polymeric byproducts often seen in harsh acidic conditions.
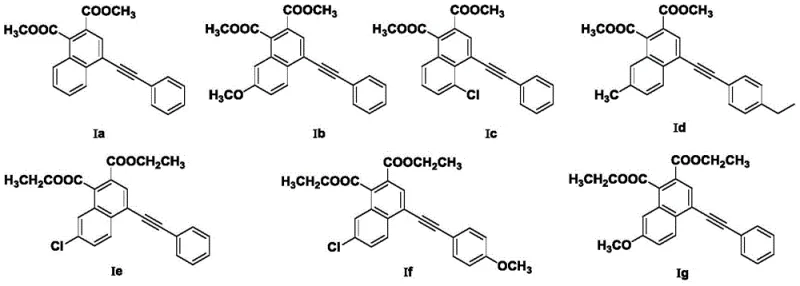
From an impurity control perspective, the use of a shape-selective mesoporous catalyst like Al-SBA-15 offers distinct advantages over homogeneous acids. The defined pore structure can restrict the formation of bulky side products, while the mild acidity prevents excessive decomposition of sensitive functional groups such as esters or halides. The patent data demonstrates excellent tolerance for various substituents (R1, R2, R3), including chloro, methoxy, and methyl groups, indicating that the electronic environment of the catalyst surface is well-balanced to accommodate diverse substrates without compromising the integrity of the final product. This robustness is essential for producing high-purity specialty chemical intermediates where trace impurities can derail downstream applications in electronics or pharmacology.
How to Synthesize Ethynylnaphthalene Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for generating these valuable scaffolds. It begins with the preparation of the alkynol precursor via a base-catalyzed addition of phenylacetylene to acetophenone derivatives in DMSO, followed by the key cyclization step in water. The operational simplicity of filtering the catalyst and separating the phases makes this method particularly attractive for scale-up. For detailed procedural specifics regarding reagent ratios, temperature profiles, and purification techniques, please refer to the standardized synthesis guide below which encapsulates the critical process parameters.
- Preparation of Alkynol Precursor: React phenylacetylene with substituted acetophenone using Bu4NOH/DMSO at 5°C to form the alkynol intermediate.
- Cyclization Reaction: Mix the alkynol intermediate with dimethyl butynedioate in water containing Al-SBA-15 catalyst at 110°C.
- Workup and Purification: Filter the heterogeneous catalyst for reuse, extract the organic phase, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous catalytic system translates into tangible economic and logistical benefits. The primary driver for cost optimization is the elimination of expensive, anhydrous organic solvents and the reduction in waste disposal fees associated with halogenated solvents. By switching to water, the process inherently lowers the raw material costs and mitigates the regulatory burdens associated with VOC emissions. Additionally, the recyclability of the Al-SBA-15 catalyst means that the cost of catalytic materials is amortized over multiple batches, leading to a significant reduction in the cost of goods sold (COGS) over the lifecycle of the product. This efficiency allows for more competitive pricing strategies in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The transition to a water-based solvent system removes the necessity for complex solvent recovery distillation columns and reduces energy consumption related to solvent evaporation. Since the catalyst is heterogeneous, there is no need for costly metal scavenging resins or extensive washing protocols to meet strict residual metal specifications required by pharmaceutical customers. This streamlining of the downstream processing directly correlates to lower operational expenditures and improved margin potential for high-volume production runs.
- Enhanced Supply Chain Reliability: Utilizing water as a solvent significantly improves the safety profile of the manufacturing facility, reducing the risk of fire and explosion hazards associated with flammable organic solvents. This safety enhancement leads to fewer regulatory inspections and interruptions, ensuring a more consistent and reliable supply of critical intermediates. Furthermore, the starting materials, such as substituted acetophenones and phenylacetylenes, are commodity chemicals with stable global supply chains, minimizing the risk of raw material shortages that could impact production schedules.
- Scalability and Environmental Compliance: The one-pot nature of the reaction reduces the number of isolation steps, which simplifies equipment requirements and shortens the overall cycle time per batch. This efficiency is crucial for scaling from pilot plant to commercial tonnage production. Moreover, the aqueous effluent generated is easier to treat compared to mixed organic waste streams, facilitating compliance with increasingly stringent environmental regulations. This green chemistry alignment future-proofs the supply chain against evolving sustainability mandates from end-user industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of the Al-SBA-15 catalytic system.
Q: What are the primary advantages of using Al-SBA-15 in water for this synthesis?
A: The use of Al-SBA-15 in water eliminates the need for toxic organic solvents and allows for easy catalyst recovery via filtration, significantly reducing waste disposal costs and environmental impact compared to traditional homogeneous metal catalysis.
Q: Can this process be scaled for industrial production of dye intermediates?
A: Yes, the protocol utilizes a heterogeneous catalyst and water solvent, which are inherently safer and easier to handle on a large scale, making it highly suitable for the commercial manufacturing of specialty chemical intermediates.
Q: What is the substrate scope for the R1 and R3 positions?
A: The method tolerates a wide range of substituents including hydrogen, methyl, methoxy, chlorine, and bromine at various positions, allowing for the synthesis of diverse functionalized naphthalene derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethynylnaphthalene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient, green synthetic routes for complex aromatic systems. Our technical team has extensively evaluated the Al-SBA-15 catalyzed cyclization pathway and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of ethynylnaphthalene derivatives meets the exacting standards required for downstream synthesis of dyes and pharmaceuticals.
We invite you to collaborate with us to leverage this innovative technology for your specific project requirements. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how this aqueous process can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to drive efficiency and innovation in your supply chain.
