Revolutionizing 5-Amino-2-Methyl Quinoline Production: A Technical Deep Dive into Scalable Synthetic Routes
The pharmaceutical industry constantly seeks robust synthetic pathways for critical heterocyclic intermediates, and the production of 5-amino-2-methyl quinoline stands as a prime example of process evolution. As detailed in patent CN101983961A, a novel preparation method has emerged that fundamentally shifts the paradigm from energy-intensive thermal cyclization to mild, Lewis acid-catalyzed construction. This technical breakthrough addresses long-standing inefficiencies in yield and purity that have plagued manufacturers for decades. By leveraging o-chloroaniline and crotonaldehyde as accessible starting materials, the new route achieves a streamlined three-step sequence that is inherently safer and more economically viable. For R&D directors and procurement specialists alike, understanding this shift is crucial, as it represents a move towards sustainable, high-yield manufacturing that aligns with modern green chemistry principles and stringent supply chain reliability standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-amino-2-methyl quinoline relied heavily on the condensation of aniline with methyl acetoacetate, a process fraught with thermodynamic and kinetic challenges. The most significant bottleneck in this traditional pathway is the cyclization step, which necessitates extremely high temperatures reaching approximately 250°C to drive the ring closure. Such harsh thermal conditions not only consume excessive energy but also promote side reactions, leading to a mediocre cyclization yield of roughly 50%. Furthermore, the subsequent nitration of the resulting 2-methylquinoline is notoriously unselective, generating a complex mixture of 5-nitro and 8-nitro isomers. Separating these isomers is technically demanding and costly, often requiring extensive chromatography or recrystallization, which further erodes the overall process yield to as low as 30%. These factors combined make the conventional route unsuitable for large-scale industrial production where cost efficiency and consistent quality are paramount.
The Novel Approach
In stark contrast, the innovative methodology disclosed in the patent utilizes a strategic retrosynthetic disconnection that bypasses these thermal and selectivity hurdles entirely. By employing o-chloroaniline instead of plain aniline, the synthesis leverages the electronic properties of the chlorine substituent to direct subsequent reactions favorably. The initial cyclization with crotonaldehyde proceeds under the influence of a Lewis acid catalyst at significantly milder temperatures, ranging from ambient conditions to reflux, eliminating the need for extreme heat. This approach not only boosts the initial ring-closure yield to nearly quantitative levels but also sets the stage for highly regioselective nitration. The chlorine atom effectively blocks the 8-position and directs the nitro group to the desired 5-position, virtually eliminating the isomer contamination that plagues older methods. This structural elegance translates directly into simplified downstream processing and superior product quality.
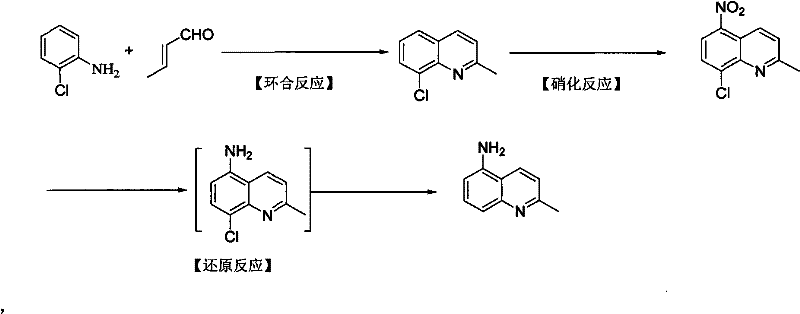
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization and Regioselective Nitration
The core of this synthetic advancement lies in the precise orchestration of the cyclization mechanism mediated by Lewis acids such as zinc chloride or ferric chloride. In this step, the Lewis acid activates the carbonyl group of the crotonaldehyde, facilitating a nucleophilic attack by the amino group of o-chloroaniline. This interaction promotes the formation of an imine intermediate which subsequently undergoes an intramolecular electrophilic aromatic substitution to close the quinoline ring. The presence of the chlorine atom at the ortho position is critical; it stabilizes the transition state and prevents polymerization of the aldehyde, a common side reaction in Skraup-type syntheses. The reaction is conducted in a mixed solvent system, typically involving propanol and isopropyl ether or THF, which optimizes solubility and heat transfer, ensuring that the reaction proceeds smoothly to afford 8-chloro-2-methylquinoline with exceptional purity exceeding 99%.
Following the ring construction, the nitration step demonstrates remarkable regiocontrol driven by the electronic nature of the quinoline scaffold. When 8-chloro-2-methylquinoline is subjected to a mixture of sulfuric and nitric acid at controlled temperatures between 25°C and 50°C, the electrophilic nitronium ion attacks the ring. Due to the deactivating yet ortho/para-directing nature of the chlorine atom (relative to its position) and the inherent electron density distribution of the quinoline nucleus, substitution occurs almost exclusively at the 5-position. This avoids the formation of the 8-nitro isomer, which is a major impurity in non-chlorinated routes. The subsequent reduction phase utilizes a tandem strategy: initial reduction with iron powder in acidic ethanol converts the nitro group to an amine, followed by catalytic hydrogenation with 5% palladium carbon. This second reduction step is vital as it simultaneously removes the chlorine atom via hydrodechlorination, yielding the final target molecule, 5-amino-2-methyl quinoline, without requiring separate dehalogenation steps.
How to Synthesize 5-Amino-2-Methyl Quinoline Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process begins with the careful addition of crotonaldehyde to a mixture of o-chloroaniline and Lewis acid in a solvent blend, maintaining temperature control to prevent exothermic runaway. Once the chloro-quinoline intermediate is isolated, it is dissolved in sulfuric acid for nitration, where the rate of acid addition must be managed to maintain the temperature below 50°C, preventing oxidative degradation. Finally, the reduction sequence involves a heterogeneous reaction with iron powder followed by a pressurized hydrogenation step. While the chemical transformations are robust, the physical handling of reagents and the workup procedures—such as pH adjustment and filtration—are critical for obtaining the high-purity solid product required for pharmaceutical applications. Detailed standardized operating procedures for these steps are essential for technology transfer.
- Cyclize o-chloroaniline and crotonaldehyde using a Lewis acid catalyst like zinc chloride in a mixed solvent system to form 8-chloro-2-methylquinoline.
- Perform nitration on the quinoline ring using sulfuric and nitric acid at controlled temperatures (25-50°C) to obtain 5-nitro-8-chloro-2-methylquinoline.
- Execute a two-stage reduction using iron powder followed by palladium carbon catalytic hydrogenation to remove the chloro group and reduce the nitro group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits that extend beyond simple chemical yield. The elimination of high-temperature cyclization steps drastically reduces energy consumption, directly lowering the utility costs associated with manufacturing. Furthermore, the use of commodity chemicals like o-chloroaniline and crotonaldehyde ensures a stable and diversified supply base, mitigating the risk of raw material shortages that can disrupt production schedules. The high regioselectivity of the nitration step means that less solvent and fewer resources are wasted on purification processes like chromatography, which are often the most expensive part of fine chemical production. This efficiency allows for a more predictable cost structure and shorter lead times, enabling suppliers to respond more agilely to market demand fluctuations without compromising on margin.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the workflow and the reduction of waste. By avoiding the formation of difficult-to-separate isomers, the need for complex purification infrastructure is minimized, leading to significant capital expenditure savings. Additionally, the mild reaction conditions reduce the wear and tear on reactor vessels and lower the safety overhead associated with high-pressure or high-temperature operations. The ability to recover and reuse catalysts, such as the palladium carbon in the final step, further contributes to a leaner cost profile, making the final API intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the chemical steps, which tolerate minor variations in conditions better than the finicky high-temperature cyclization of the past. The high yields reported in the patent examples, often exceeding 90% for the first two steps, mean that less starting material is required to produce a given amount of product, effectively increasing the throughput of existing facilities. This efficiency creates a buffer against supply shocks, ensuring that delivery commitments to downstream pharmaceutical partners can be met consistently. The simplicity of the post-treatment workups also accelerates the batch cycle time, allowing for more frequent production runs and tighter inventory management.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns well with modern regulatory expectations. The avoidance of extreme temperatures and the use of standard solvents simplify the engineering controls required for scale-up from pilot plant to commercial tonnage. The reduction in isomeric byproducts translates to a cleaner waste stream, reducing the burden on effluent treatment plants and lowering the environmental compliance costs. The process is inherently safer, with lower risks of thermal runaway, which facilitates easier permitting and insurance coverage for manufacturing sites. These factors combined make the technology highly attractive for long-term commercial partnerships focused on sustainable growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 5-amino-2-methyl quinoline using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis provided in the underlying patent literature, offering clarity on why this route is superior for industrial application. Understanding these nuances helps stakeholders make informed decisions about sourcing and process adoption.
Q: Why is the traditional aniline-based synthesis method considered inefficient for industrial scale?
A: Traditional methods often require extreme temperatures around 250°C for cyclization, resulting in low yields (approx. 50%) and significant energy costs. Furthermore, the subsequent nitration step produces difficult-to-separate isomers (5-nitro and 8-nitro), drastically reducing the overall purity and final yield to as low as 30%.
Q: How does the novel Lewis acid catalyzed route improve regioselectivity during nitration?
A: By utilizing o-chloroaniline as the starting material, the chlorine atom acts as a directing group that favors substitution at the 5-position of the quinoline ring. This strategic structural design minimizes the formation of unwanted isomers, allowing for much simpler purification and significantly higher isolated yields compared to unsubstituted aniline routes.
Q: What are the specific advantages of the reductive dechlorination step in this process?
A: The process employs a combination of iron powder reduction and palladium carbon catalytic hydrogenation. This dual approach ensures complete removal of the chlorine atom while simultaneously reducing the nitro group to an amine under mild conditions (40-50°C), avoiding the harsh conditions that typically degrade sensitive quinoline structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Amino-2-Methyl Quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields seen in patent literature are replicated faithfully on an industrial scale. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of 5-amino-2-methyl quinoline meets the exacting standards required for pharmaceutical intermediates. Our commitment to quality assurance means that clients can rely on us for consistent supply without the variability often associated with complex heterocyclic synthesis.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume requirements. We encourage potential clients to contact us directly to索取 specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal standards. Let us collaborate to secure a stable, high-quality supply of this critical intermediate for your next-generation therapeutic developments.
