Optimizing Aspernigerin Production: A Technical Analysis of High-Yield Synthetic Routes for Pharmaceutical Applications
Introduction to Advanced Aspernigerin Synthesis Technology
The pharmaceutical industry is constantly seeking robust synthetic pathways for bioactive alkaloids, particularly those exhibiting potent anticancer properties. Patent CN102285915B introduces a groundbreaking synthesis method for Aspernigerin, a novel alkaloid secondary metabolite originally isolated from the endophytic fungus Aspergillus niger. This compound has demonstrated significant cytotoxic activity against various human cancer cell lines, including KB, Hela, and SW1116, positioning it as a critical lead compound in oncology drug discovery. The structural complexity of Aspernigerin, characterized by its unique bis-tetrahydroquinoline piperazine scaffold, has historically posed challenges for efficient manufacturing. However, the methodology detailed in this patent offers a transformative approach, shifting from low-yield natural extraction and cumbersome total synthesis to a streamlined, high-efficiency chemical construction.
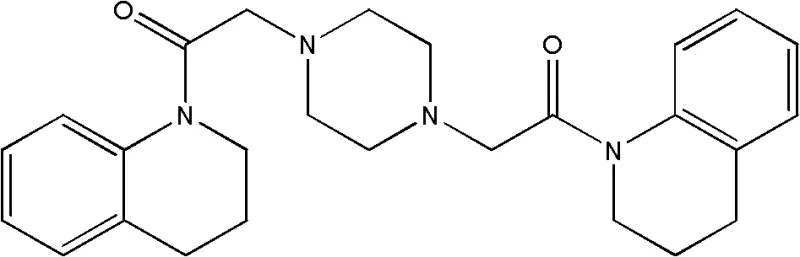
This technical insight report dissects the proprietary synthesis route disclosed in CN102285915B, evaluating its viability for commercial scale-up. By leveraging a two-step acylation and alkylation strategy, the process achieves exceptional purity levels exceeding 99% while drastically reducing operational complexity. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable supply of high-quality pharmaceutical intermediates. The following analysis provides a deep dive into the mechanistic advantages, operational parameters, and commercial implications of adopting this superior synthetic methodology for the production of anticancer agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN102285915B, the availability of Aspernigerin was severely restricted by the inefficiencies of existing production methods. The seminal work by the Tan Ren group, while pioneering, relied on a synthetic route that was inherently flawed for industrial application. As illustrated in the historical context of the patent, the conventional pathway involved multiple reaction steps that were not only chemically inefficient but also operationally burdensome. The most critical bottleneck was the overall yield, which hovered around a mere 47.3%, rendering the process economically unviable for large-scale manufacturing. Furthermore, the purification protocols were excessively tedious, necessitating repeated column chromatography separations.
From a process chemistry perspective, the reliance on column chromatography is a major red flag for scale-up, as it introduces significant bottlenecks in throughput and generates substantial solid waste. Additionally, the conventional method required frequent pH adjustments using acids and bases, leading to the generation of large volumes of saline wastewater, often referred to as 'three wastes' in industrial terminology. These factors combined to create a high-cost, low-output scenario that hindered the broader pharmacological evaluation and potential clinical development of Aspernigerin. The inability to produce gram-to-kilogram quantities efficiently meant that supply chain continuity for this valuable anticancer intermediate remained a persistent challenge for the global research community.
The Novel Approach
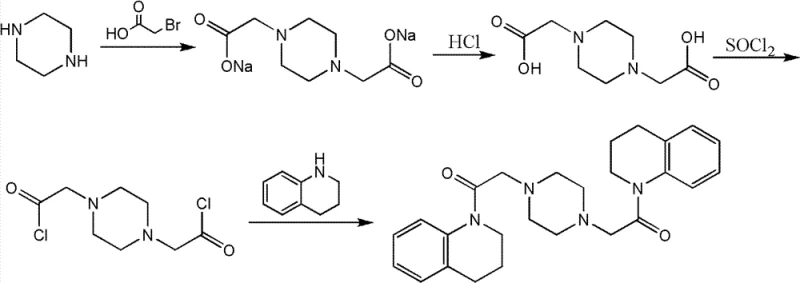
In stark contrast to the convoluted historical routes, the novel approach presented in CN102285915B simplifies the synthesis into a concise, two-step sequence that maximizes atom economy and operational simplicity. The core innovation lies in the strategic construction of the molecule using readily available starting materials: 1,2,3,4-tetrahydroquinoline, chloroacetyl chloride, and piperazine. By decoupling the synthesis into a distinct acylation phase followed by a coupling phase, the inventors have created a modular process that allows for rigorous quality control at each stage. The first step generates a stable intermediate, N-chloroacetyl-1,2,3,4-tetrahydroquinoline, which can be purified via vacuum distillation rather than chromatography, setting a high standard of purity before the final coupling even begins.
The second step involves the nucleophilic substitution of the chloro-intermediate with piperazine, a reaction that proceeds with high efficiency under controlled thermal conditions. Crucially, the final purification is achieved through a simple recrystallization process using toluene, completely eliminating the need for column chromatography. This shift from chromatographic purification to crystallization is a hallmark of mature process chemistry, signaling that the route is ready for ton-scale production. The result is a white crystalline product with a purity of greater than 99% as determined by HPLC, achieved with significantly reduced environmental impact and operational time. This novel approach effectively resolves the supply constraints that previously plagued the development of Aspernigerin-based therapeutics.
Mechanistic Insights into Nucleophilic Acyl Substitution and Alkylation
The chemical elegance of this synthesis rests on two fundamental organic transformations: nucleophilic acyl substitution and nucleophilic aliphatic substitution. In the first stage, 1,2,3,4-tetrahydroquinoline acts as a nucleophile, attacking the electrophilic carbonyl carbon of chloroacetyl chloride. This reaction is facilitated by an acid-binding agent, such as triethylamine or N,N-dimethylaniline, which scavenges the hydrogen chloride byproduct, driving the equilibrium forward. The reaction temperature is carefully maintained between -10°C and 40°C to prevent side reactions, such as polymerization or over-acylation, ensuring the selective formation of the mono-acylated intermediate. The use of solvents like dichloromethane or chloroform provides an ideal medium for this exothermic reaction, allowing for precise thermal control and efficient mixing.
The second mechanistic phase involves the displacement of the chloride ion in the intermediate by the nitrogen atoms of piperazine. This is a classic SN2-type reaction where the secondary amine of piperazine attacks the methylene carbon adjacent to the carbonyl group. The reaction requires higher temperatures, typically between 50°C and 150°C, to overcome the activation energy barrier, especially when using less polar solvents like toluene. The choice of base in this step is critical; inorganic bases like anhydrous potassium carbonate or organic bases like triethylamine are employed to neutralize the generated HCl and maintain the nucleophilicity of the piperazine. The symmetry of the piperazine molecule allows for the simultaneous formation of two amide bonds, constructing the final bis-substituted architecture of Aspernigerin in a single pot. This convergent synthesis strategy minimizes the number of isolation steps, thereby reducing material loss and maximizing the overall process yield.
Impurity control is intrinsically built into this mechanistic design. By isolating the intermediate via vacuum distillation at 162°C to 164°C under reduced pressure (130 Pa), volatile impurities and unreacted starting materials are effectively removed. This ensures that the subsequent coupling reaction starts with a high-purity substrate, which is essential for preventing the formation of complex byproduct mixtures that are difficult to separate later. Furthermore, the final recrystallization from toluene exploits the differential solubility of the product versus any remaining salts or oligomeric impurities. As the solution cools, the highly pure Aspernigerin crystallizes out, leaving impurities in the mother liquor. This thermodynamic purification method is far superior to kinetic separation methods like chromatography for large-scale manufacturing, ensuring consistent batch-to-batch quality and robust impurity profiles that meet stringent pharmaceutical standards.
How to Synthesize Aspernigerin Efficiently
Implementing this synthesis protocol requires precise adherence to the reaction parameters outlined in the patent to ensure optimal yield and safety. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors. The key to success lies in the careful control of addition rates and temperature profiles during the exothermic acylation step, as well as the efficient removal of solvent and salts during the workup phases. Operators must be trained to monitor reaction progress via thin-layer chromatography (TLC) to determine the exact endpoint, preventing over-reaction or degradation. The following guide summarizes the critical operational stages derived from the patent embodiments, serving as a foundational reference for process engineers aiming to replicate this high-efficiency route.
- Dissolve 1,2,3,4-tetrahydroquinoline in an organic solvent such as dichloromethane, cool to -10°C to 40°C, and react with chloroacetyl chloride in the presence of an acid-binding agent to form N-chloroacetyl-1,2,3,4-tetrahydroquinoline.
- Dissolve piperazine in a solvent like DMF or toluene with a base, then add the N-chloroacetyl intermediate dropwise at 50°C to 150°C to facilitate the coupling reaction.
- Purify the crude product by removing solvents and recrystallizing from toluene or mixed solvents to obtain white crystals with purity exceeding 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN102285915B represents a significant opportunity to optimize costs and secure supply continuity for anticancer intermediates. The transition from a low-yield, chromatography-dependent process to a high-yield, crystallization-based workflow fundamentally alters the cost structure of manufacturing. By eliminating the need for expensive silica gel and the labor-intensive processes associated with column chromatography, the direct material and operational costs are substantially reduced. Furthermore, the high yield of the intermediate step, reported at over 90%, ensures that the consumption of the primary starting material, 1,2,3,4-tetrahydroquinoline, is minimized, directly contributing to a lower cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the purification workflow. In traditional fine chemical manufacturing, purification can account for a significant portion of total production costs. By replacing column chromatography with vacuum distillation and recrystallization, the process eliminates the recurring cost of chromatographic media and the associated solvent waste disposal fees. Additionally, the solvents used, such as dichloromethane, toluene, and ethanol, are commodity chemicals with established supply chains and low price volatility. The ability to recover and recycle these solvents further enhances the economic efficiency of the process, making the production of Aspernigerin commercially viable for the first time.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, non-proprietary starting materials. Unlike complex natural product extractions that are subject to seasonal and biological variability, this synthetic route relies on stable petrochemical derivatives. The robustness of the reaction conditions, which do not require exotic catalysts or sensitive reagents, reduces the risk of batch failures due to reagent quality issues. Moreover, the simplified workflow shortens the overall production cycle time, allowing manufacturers to respond more rapidly to fluctuations in market demand. This agility is crucial for maintaining inventory levels of critical pharmaceutical intermediates in a dynamic global market.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a greener alternative to previous techniques. The reduction in 'three wastes'—specifically the avoidance of silica waste from chromatography and the minimization of aqueous waste from pH adjustments—aligns with modern sustainability goals. The process is inherently scalable; the unit operations involved (stirring, heating, distillation, filtration) are standard in chemical plants ranging from pilot scale to multi-ton production. This scalability ensures that as the clinical demand for Aspernigerin grows, the supply chain can expand capacity without requiring fundamental changes to the manufacturing technology, thereby guaranteeing long-term supply security for downstream drug developers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Aspernigerin using the patented methodology. These insights are derived directly from the experimental data and beneficial effects reported in CN102285915B, providing clarity for stakeholders evaluating this technology for integration into their supply chains. Understanding these details is vital for assessing the feasibility of large-scale adoption and ensuring that quality expectations are met.
Q: How does the new synthesis method improve yield compared to previous literature?
A: The patented method significantly improves overall yield by eliminating complex purification steps. While previous methods reported yields around 47.3%, this optimized route achieves step yields of over 90% for the intermediate and up to 89% for the final product through efficient distillation and recrystallization.
Q: What are the primary advantages regarding impurity control and purification?
A: Unlike conventional routes requiring tedious column chromatography, this process utilizes vacuum distillation for the intermediate and recrystallization for the final product. This not only simplifies operations but also ensures high purity (>99%) suitable for pharmaceutical applications without heavy metal or silica contamination.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is highly scalable. It uses common industrial solvents like dichloromethane and toluene, avoids extreme conditions, and relies on standard unit operations like filtration and distillation, making it ideal for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aspernigerin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of optimized synthetic routes like the one described in CN102285915B for the advancement of oncology therapeutics. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate such laboratory innovations into commercial reality. Our team is equipped to handle the scale-up of complex pharmaceutical intermediates, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that the transition from gram-scale synthesis to industrial manufacturing requires meticulous attention to detail, particularly regarding the stringent purity specifications and rigorous QC labs necessary for API intermediates. Our state-of-the-art facilities are designed to implement the high-yield, low-waste protocols detailed in this report, ensuring that every batch of Aspernigerin meets the highest international quality standards.
We invite pharmaceutical companies and research institutions to collaborate with us to secure a stable, cost-effective supply of this critical anticancer intermediate. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your overall procurement spend. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your drug development timeline with a reliable, high-quality supply of Aspernigerin, backed by our commitment to excellence in process chemistry and customer service.
