Advanced One-Pot Synthesis of Cefcapene Key Intermediate for Scalable Pharmaceutical Manufacturing
Introduction to Advanced Cefcapene Intermediate Manufacturing
The pharmaceutical landscape for cephalosporin antibiotics demands increasingly sophisticated synthetic routes that balance high purity with economic viability. Patent CN102775425A introduces a groundbreaking one-pot preparation method for S-1006 diisopropylamine salt (III), a pivotal intermediate in the synthesis of Cefcapene Pivoxil (S-1108). This third-generation oral cephalosporin is renowned for its extensive antimicrobial activity against both gram-positive and gram-negative bacteria. The structural integrity of the cephem nucleus is paramount, yet traditional methods often compromise this core through harsh chemical environments. By shifting from unstable free acid or inorganic salt forms to a robust organic diisopropylamine salt, this technology ensures the preservation of the beta-lactam ring during critical processing stages.
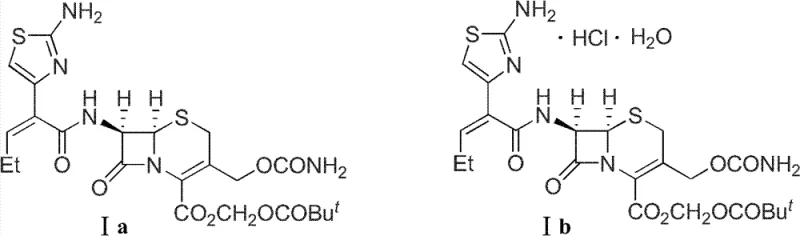
This innovation addresses the specific vulnerability of the 3-hydroxymethyl and 4-carboxyl groups on the cephem ring, which are prone to dehydration and lactone formation under alkaline conditions. The strategic implementation of a one-pot boiling technique not only streamlines the workflow but also drastically reduces the exposure of the sensitive molecule to potentially degrading conditions. For R&D directors and process chemists, this represents a significant leap forward in impurity control, offering a pathway to high-purity intermediates that meet the stringent specifications required for global regulatory approval. The ability to synthesize this key intermediate with high yield and purity directly impacts the downstream efficiency of producing the final API, making it a critical focus for supply chain optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefcapene intermediates has been plagued by complex multi-step sequences that rely heavily on inorganic bases and protecting group strategies that are difficult to manage on a large scale. As illustrated in prior art such as Scheme 1, conventional routes often involve the formation of inorganic salts like sodium or potassium salts using reagents such as potassium carbonate. While effective for solubility, these strong inorganic bases pose a severe threat to the stability of the cephem carboxylic parent nucleus. The alkaline environment can trigger destructive side reactions, including the dehydration of the 3-hydroxymethyl and 4-carboxyl groups to form lactones, or even migration of the double bond in the main ring.
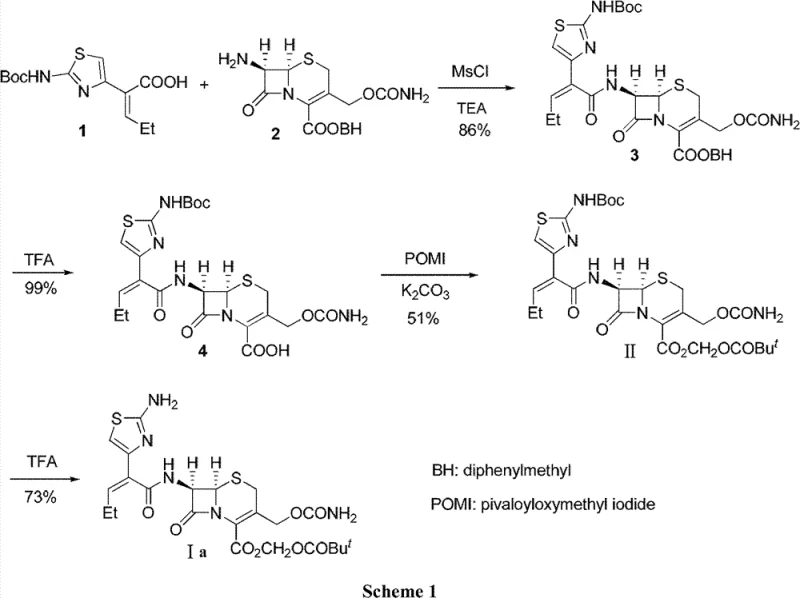
Furthermore, existing literature describes routes utilizing silylating agents like BSA (N,O-bis(trimethylsilyl)acetamide) to protect hydroxyl and carboxyl groups. While chemically sound, these methods introduce significant operational burdens, including the need for rigorous moisture control and the subsequent removal of silicon-containing byproducts. The cumulative effect of these inefficiencies is often reflected in suboptimal yields; for instance, certain esterification steps in traditional free acid pathways have been reported to yield as low as 51%. These limitations create bottlenecks in manufacturing, increasing both the cost of goods sold and the lead time for reliable pharmaceutical intermediate supplier deliveries.
The Novel Approach
The methodology disclosed in CN102775425A fundamentally reengineers this synthetic landscape by adopting a telescoped, one-pot strategy that bypasses the pitfalls of inorganic base usage. Instead of isolating the free acid or converting it to an inorganic salt, the process directly generates the diisopropylamine salt (III) through a carefully orchestrated sequence of activation, amidation, and carbamoylation. This approach leverages the steric hindrance and basicity of diisopropylamine to facilitate the reaction without compromising the delicate beta-lactam structure. By eliminating the isolation of intermediates, the process minimizes material loss and reduces the consumption of solvents and reagents associated with multiple work-up procedures.
This novel route is characterized by its rational design, which integrates four distinct reaction steps into a single continuous operation. The use of methanesulfonyl chloride (MsCl) for activation followed by direct coupling with 7-HACA allows for high conversion rates under mild conditions. Subsequent carbamoylation with chlorosulfonyl isocyanate (CSI) and final salt formation occur in the same reactor, ensuring that the intermediate never encounters the harsh conditions that lead to degradation. For procurement managers, this translates to a more robust supply chain with reduced risk of batch failure, while R&D teams benefit from a cleaner impurity profile that simplifies downstream purification.
Mechanistic Insights into Mixed Anhydride Activation and One-Pot Telescoping
The core of this technological advancement lies in the precise control of reactivity through mixed anhydride activation. The process initiates with the reaction of Boc-protected (Z)-2-(2-aminothiazol-4-yl)-pentenoic acid (1) with methanesulfonyl chloride (MsCl) in an organic solvent such as ethyl acetate. This step generates the highly reactive mixed acid anhydride (10) in situ. Unlike traditional carbodiimide couplings which can generate urea byproducts that are difficult to remove, the mesylate byproduct here is manageable and does not interfere with the subsequent nucleophilic attack. The reaction is conducted at low temperatures, typically between -5°C and -15°C, to suppress potential racemization and ensure the kinetic control necessary for high stereochemical purity.
Following activation, the introduction of 7-HACA (3-hydroxymethyl-7-aminocephalosporanic acid) in the presence of diisopropylamine drives the amidation reaction to form compound (11). The choice of diisopropylamine is critical; it acts as a proton scavenger to neutralize the HCl generated during the reaction, maintaining a pH environment that is sufficiently basic to promote amidation but not so alkaline as to open the beta-lactam ring. The subsequent addition of chlorosulfonyl isocyanate (CSI) targets the 3-hydroxymethyl group specifically, converting it into the carbamate moiety essential for the biological activity of the final drug. This sequence culminates in the addition of excess diisopropylamine to precipitate the product as a stable organic salt.
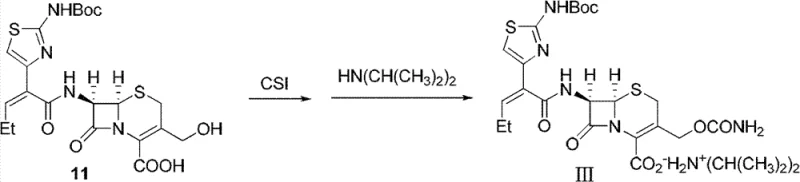
From a mechanistic standpoint, the avoidance of aqueous workups between steps is a masterstroke in process chemistry. Each intermediate remains in solution, protected by the solvent matrix and the controlled stoichiometry of reagents. This "one-pot boiling" technique ensures that the concentration of reactive species is optimized for the next transformation immediately upon completion of the previous one. For technical teams evaluating scale-up feasibility, this mechanism offers a clear advantage: it reduces the number of unit operations, minimizes the hold time of unstable intermediates, and provides a consistent trajectory for impurity rejection. The result is a process that is not only chemically elegant but also industrially robust, capable of delivering high-purity pharmaceutical intermediates with minimal variability.
How to Synthesize Cefcapene Diisopropylamine Salt Efficiently
Implementing this synthesis requires strict adherence to temperature controls and reagent addition rates to maintain the integrity of the one-pot system. The process begins with the activation of the side chain acid, followed by the sequential addition of the cephem nucleus and carbamoylating agent without intermediate isolation. This streamlined approach is designed to maximize throughput while minimizing the footprint of the manufacturing facility. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and stirring speeds are critical for reproducibility and are outlined in the technical documentation below.
- Activate Boc-protected pentenoic acid (1) with Methanesulfonyl chloride (MsCl) in ethyl acetate at low temperature to form mixed anhydride (10).
- Add 7-HACA and diisopropylamine to the reaction mixture for direct amidation to form compound (11) without isolation.
- React with Chlorosulfonyl isocyanate (CSI) for carbamoylation, followed by salt formation with diisopropylamine to precipitate product III.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the shift to this one-pot organic salt synthesis offers profound economic and logistical benefits. Traditional methods that rely on inorganic salts or complex silyl protection groups inherently carry higher operational costs due to the need for additional reagents, extended reaction times, and complex waste streams. By consolidating the synthesis into a single vessel operation, manufacturers can significantly reduce the consumption of utilities such as heating, cooling, and agitation power. Furthermore, the elimination of intermediate isolation steps means that less solvent is required for crystallization and washing, directly lowering the variable costs associated with raw material procurement and solvent recovery.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of expensive and hazardous reagents typically used in alternative routes. For instance, avoiding the use of silylating agents like BSA eliminates a significant cost center, as these reagents are often pricey and require careful handling. Additionally, the high yield of the reaction ensures that the expensive 7-HACA starting material is utilized with maximum efficiency, reducing the cost per kilogram of the final intermediate. The simplified workflow also reduces labor hours and equipment occupancy time, allowing for greater throughput within existing infrastructure without the need for capital-intensive expansion.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals such as methanesulfonyl chloride and diisopropylamine, which are readily available from multiple global suppliers. This contrasts with specialized reagents that might have single-source dependencies or long lead times. The robustness of the one-pot method also means that the process is less susceptible to minor fluctuations in reaction conditions, leading to more consistent batch-to-batch quality. For procurement managers, this reliability translates to fewer production delays and a more predictable inventory flow, ensuring that downstream API manufacturing schedules are met without interruption.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with modern green chemistry principles. The reduction in solvent usage and the avoidance of heavy metal catalysts or toxic inorganic byproducts simplify the effluent treatment process. The organic salt form of the intermediate is generally more stable and easier to handle than free acids, reducing the risk of degradation during storage and transport. This stability facilitates commercial scale-up, allowing producers to move from pilot plant quantities to multi-ton annual production with confidence. The streamlined waste profile further aids in meeting stringent environmental regulations, reducing the liability and cost associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for partners looking to integrate this intermediate into their supply chain. The following questions address common concerns regarding the stability, purity, and scalability of the diisopropylamine salt form. These insights are derived directly from the patent data and practical experience in scaling similar cephalosporin pathways, providing a transparent view of the technology's capabilities and limitations.
Q: Why is the diisopropylamine salt form preferred over inorganic salts for Cefcapene intermediates?
A: Inorganic strong bases used to form sodium or potassium salts can destroy the sensitive cephem carboxylic parent nucleus, leading to lactone formation or double-bond migration. The organic diisopropylamine salt avoids this degradation, ensuring higher purity and yield for subsequent esterification.
Q: What are the advantages of the one-pot synthesis method described in CN102775425A?
A: The one-pot method eliminates the need for isolating intermediate compounds, significantly simplifying operation steps. It avoids the use of expensive silylating agents and harsh inorganic conditions, resulting in a more reasonable process design that is easier to scale up for industrial production.
Q: How does this process improve the yield compared to conventional routes?
A: Conventional routes often suffer from low yields (as low as 51% in some esterification steps) due to side reactions caused by inorganic bases. This novel route protects the beta-lactam ring integrity throughout the sequence, leading to high reaction yields and superior product quality suitable for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefcapene Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trials to full market launch. We understand the critical nature of beta-lactam stability and employ stringent purity specifications and rigorous QC labs to guarantee that every batch of Cefcapene intermediate meets the highest international standards. Our commitment to quality assurance means that you receive a product that is not only chemically pure but also consistent in its physical properties, facilitating smooth downstream processing.
We invite you to collaborate with us to leverage this advanced one-pot synthesis technology for your pharmaceutical projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. By partnering with us, you gain access to a secure supply of high-quality intermediates that can significantly enhance your manufacturing efficiency. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering excellence can drive value for your organization.
