Scalable Synthesis of N,N'-Bis(2,2,6,6-Tetramethyl-4-Piperidyl)-1,3-Benzenedicarboxamide Using Covalent Organic Frameworks
The global demand for high-performance polymer additives continues to surge, driven by the expanding applications of outdoor plastic products that require exceptional durability against UV degradation. In this context, the synthesis of N,N'-bis(2,2,6,6-tetramethyl-4-piperidyl)-1,3-benzenedicarboxamide represents a critical advancement in the field of hindered amine light stabilizers (HALS). A groundbreaking approach detailed in patent CN109280179B introduces a novel covalent organic framework (COF) material that serves as a highly efficient, metal-free heterogeneous catalyst for this transformation. This technology addresses long-standing challenges in the industry by replacing toxic acyl chloride routes with a greener ester aminolysis process, significantly enhancing both yield and environmental compliance. The structural integrity and functional efficiency of this target molecule are paramount for its role in capturing free radicals and quenching excited oxygen within polymer matrices.
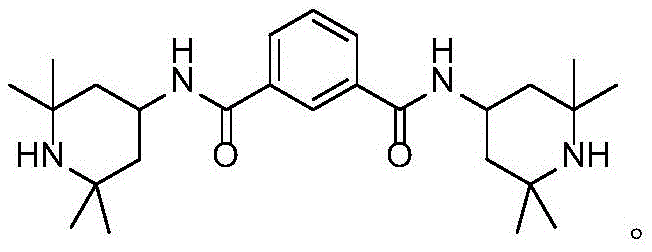
As a reliable polymer additive supplier, understanding the nuances of this synthetic pathway is essential for R&D directors aiming to optimize their supply chains. The utilization of COFs provides sufficient alkaline sites to effectively catalyze the reaction without the need for homogeneous bases that complicate downstream processing. This innovation not only improves the economic viability of producing high-purity HALS but also aligns with stringent global regulations regarding heavy metal discharge and wastewater treatment. By leveraging this patented methodology, manufacturers can achieve a robust balance between operational efficiency and sustainability, ensuring a continuous supply of critical stabilizers for the plastics industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N'-bis(2,2,6,6-tetramethyl-4-piperidyl)-1,3-benzenedicarboxamide has relied heavily on the use of m-diformyl chloride as a primary raw material, a method fraught with significant operational and environmental drawbacks. As illustrated in prior art such as CN103508938A and WO2017/024608, the high reactivity of acyl chlorides necessitates the use of stoichiometric acid-binding agents to neutralize the hydrochloric acid byproduct generated during the amidation reaction. This requirement leads to the formation of substantial quantities of salt-containing wastewater, creating a severe burden on waste treatment facilities and increasing the overall cost of production. Furthermore, the susceptibility of acyl chlorides to hydrolysis demands strictly anhydrous conditions, complicating the reaction setup and increasing the risk of side reactions that lower the final purity of the stabilizer.
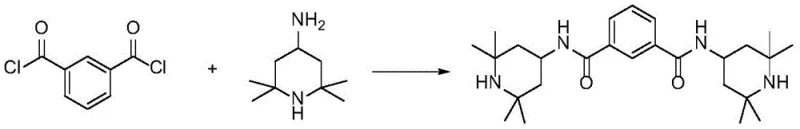
In addition to the environmental concerns, the post-treatment processes associated with acyl chloride routes are extremely complicated, often requiring multiple washing and purification steps to remove residual salts and unreacted intermediates. Alternative methods utilizing m-phthalic diesters without catalysts, as seen in CN106905225A, suffer from inherently low yields due to the poor nucleophilicity of the amine and the stability of the ester bond, rendering them commercially unviable for large-scale manufacturing. Even solid-supported catalysts containing metals, while offering some improvement in yield, introduce the risk of heavy metal contamination in the final polymer additive, which is unacceptable for high-end applications in food packaging or medical devices. These cumulative limitations highlight the urgent need for a catalytic system that combines high activity with environmental safety and ease of separation.
The Novel Approach
The novel approach presented in patent CN109280179B revolutionizes this synthesis by employing a specifically designed covalent organic framework (COF) as a heterogeneous catalyst for the aminolysis of dimethyl isophthalate. This method utilizes dimethyl isophthalate and 2,2,6,6-tetramethyl-4-aminopiperidine as starting materials, reacting them in an organic solvent under mild stirring conditions to achieve exceptional conversion rates. Unlike traditional homogeneous catalysts, the COF material possesses a rigid, porous structure with abundant alkaline sites that facilitate the nucleophilic attack of the amine on the ester carbonyl group without generating corrosive byproducts. The reaction proceeds efficiently at temperatures ranging from 40°C to 90°C, with optimal results observed at 60°C, demonstrating a significant reduction in energy consumption compared to high-temperature melt processes.
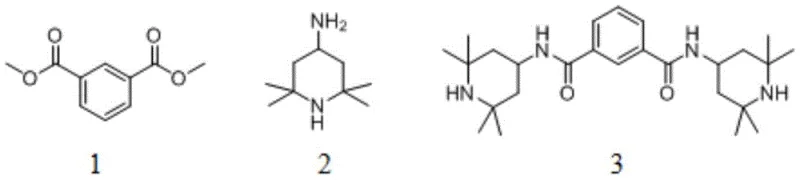
Furthermore, this innovative pathway drastically simplifies the post-treatment workflow, as the solid COF catalyst can be easily separated from the reaction mixture via simple filtration, allowing for immediate recovery and reuse. The elimination of acid-binding agents means that no salt-containing wastewater is discharged, directly addressing the environmental pain points associated with conventional acyl chloride methods. Experimental data from the patent indicates that this method achieves yields exceeding 96%, surpassing even the most optimized metal-catalyzed processes while maintaining a metal-free profile. This combination of high efficiency, operational simplicity, and environmental friendliness positions the COF-catalyzed route as the superior choice for cost reduction in plastic additive manufacturing and sustainable chemical production.
Mechanistic Insights into COF-Catalyzed Heterogeneous Aminolysis
The efficacy of the covalent organic framework catalyst lies in its unique structural composition, synthesized through the condensation of 2,5-diformylpyridine and 2,4,6-tris(4-aminophenyl)-1,3,5-triazine. This reticular chemistry results in a highly ordered, crystalline porous material that exposes a high density of nitrogen-containing basic sites within its framework. These alkaline sites act as active centers that activate the carbonyl group of the dimethyl isophthalate, making it more susceptible to nucleophilic attack by the sterically hindered 2,2,6,6-tetramethyl-4-aminopiperidine. The porous nature of the COF ensures that the reactants can diffuse freely to the active sites while preventing the aggregation of catalytic centers, which is a common issue in amorphous solid supports. This precise architectural control allows for a heterogeneous catalytic cycle that mimics the efficiency of homogeneous bases but retains the separability of a solid.
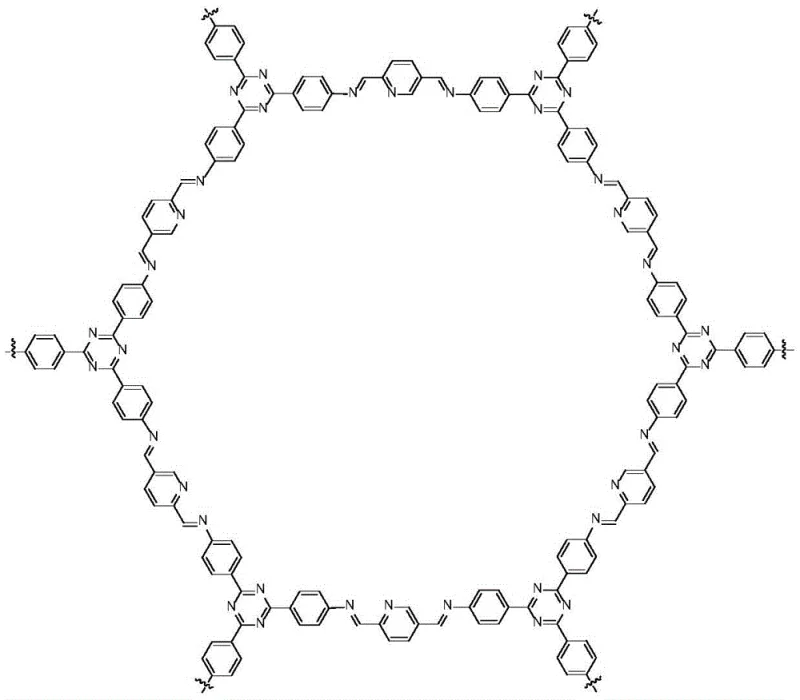
From an impurity control perspective, the metal-free nature of the COF catalyst is a decisive advantage for the production of high-purity OLED material precursors or pharmaceutical intermediates where trace metal limits are strict. Traditional metal-supported catalysts often leach trace amounts of transition metals into the product stream, necessitating expensive and time-consuming purification steps such as chelation or ion exchange. In contrast, the organic backbone of the COF ensures that no metallic contaminants are introduced, thereby simplifying the quality control process and ensuring the final product meets rigorous specification standards. Additionally, the stability of the covalent bonds within the framework prevents the degradation of the catalyst under the reaction conditions, ensuring consistent performance over multiple batches and minimizing the formation of catalyst-derived degradation byproducts.
How to Synthesize N,N'-Bis(2,2,6,6-Tetramethyl-4-Piperidyl)-1,3-Benzenedicarboxamide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology at scale, emphasizing the importance of catalyst preparation and reaction parameter optimization. The process begins with the solvothermal synthesis of the COF catalyst, followed by its application in the aminolysis reaction using ethanol as a preferred green solvent. Detailed standard operating procedures regarding solvent ratios, temperature gradients, and workup techniques are critical for reproducing the high yields reported in the examples. For a comprehensive guide on the exact stoichiometry and purification steps required to achieve commercial-grade purity, please refer to the standardized synthesis instructions below.
- Prepare the COF catalyst by reacting 2,5-diformylpyridine and 2,4,6-tris(4-aminophenyl)-1,3,5-triazine in a mixed solvent system at 120°C for 5 days.
- Mix dimethyl isophthalate and 2,2,6,6-tetramethyl-4-aminopiperidine in ethanol with the COF catalyst at a mass ratio of 58: 1.
- Stir the reaction mixture at 60°C for 12 hours, then filter to recover the catalyst and recrystallize the filtrate to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this COF-catalyzed synthesis route offers transformative benefits that extend far beyond simple yield improvements. The shift from acyl chloride chemistry to ester aminolysis fundamentally alters the cost structure of production by eliminating the need for expensive acid scavengers and the associated waste disposal costs. The ability to recycle the catalyst multiple times without significant loss of activity translates directly into a drastic reduction in raw material consumption per kilogram of finished product. Moreover, the simplified post-treatment process reduces the turnaround time for reactor vessels, allowing for higher throughput and better asset utilization within existing manufacturing facilities. These factors collectively contribute to a more resilient and cost-effective supply chain for critical polymer additives.
- Cost Reduction in Manufacturing: The elimination of acid-binding agents and the subsequent removal of salt-containing wastewater treatment steps result in substantial cost savings throughout the production lifecycle. By avoiding the use of corrosive acyl chlorides, the process also reduces equipment maintenance costs and extends the lifespan of reactor linings and piping systems. The high yield achieved under mild conditions minimizes the loss of valuable starting materials, ensuring that the cost of goods sold is optimized for maximum profitability. Furthermore, the recovery and reuse of the COF catalyst over at least 10 cycles significantly lowers the catalyst cost contribution per batch, providing a long-term economic advantage over single-use homogeneous catalysts.
- Enhanced Supply Chain Reliability: The use of stable ester starting materials like dimethyl isophthalate, which are less sensitive to moisture than acyl chlorides, reduces the risk of batch failures due to raw material degradation during storage or transport. The robustness of the COF catalyst ensures consistent reaction performance, minimizing the variability in production schedules and ensuring on-time delivery to customers. This reliability is crucial for maintaining uninterrupted production lines in the downstream plastics industry, where delays in stabilizer supply can halt the manufacturing of final consumer goods. The simplified logistics of handling non-hazardous esters compared to hazardous acid chlorides also streamline the procurement process and reduce regulatory compliance burdens.
- Scalability and Environmental Compliance: The heterogeneous nature of the reaction facilitates easy scale-up from laboratory to commercial production without the mixing and heat transfer limitations often encountered with viscous homogeneous systems. The absence of heavy metals and the reduction in saline wastewater discharge align perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing process against tighter emission standards. This green chemistry approach enhances the corporate sustainability profile of the manufacturer, appealing to eco-conscious clients and potentially qualifying for green manufacturing incentives. The ability to operate at lower temperatures also reduces the carbon footprint of the process, contributing to broader corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this COF-catalyzed technology, derived directly from the patent's background and beneficial effect sections. These insights are intended to clarify the operational advantages and technical feasibility for potential partners looking to integrate this method into their production portfolios. Understanding these details is key to evaluating the strategic fit of this technology within your existing manufacturing capabilities.
Q: Why is the COF catalyst superior to traditional acyl chloride methods for HALS synthesis?
A: Traditional acyl chloride methods require acid-binding agents which generate large amounts of salt-containing wastewater and complicate purification. The COF-catalyzed ester aminolysis avoids hydrolysis issues, eliminates heavy metal pollution, and simplifies post-treatment.
Q: Can the Covalent Organic Framework catalyst be reused in industrial production?
A: Yes, the heterogeneous nature of the COF catalyst allows for easy separation via filtration. Patent data indicates the catalyst maintains high activity with no significant reduction after at least 10 cycles of repeated use.
Q: What are the optimal reaction conditions for maximizing yield in this process?
A: The optimal conditions involve using ethanol as the solvent, a molar ratio of ester to amine of 1:2.2, and a reaction temperature of 60°C for 12 to 15 hours, achieving yields exceeding 96%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-Bis(2,2,6,6-Tetramethyl-4-Piperidyl)-1,3-Benzenedicarboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced catalytic technologies to maintain competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless and efficient. We are committed to delivering high-purity N,N'-bis(2,2,6,6-tetramethyl-4-piperidyl)-1,3-benzenedicarboxamide that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in heterogeneous catalysis and green chemistry allows us to optimize this COF-based route for maximum yield and minimal environmental impact, providing our clients with a sustainable and reliable source of high-performance light stabilizers.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of patent CN109280179B can drive value and efficiency in your supply chain. Let us be your partner in advancing the quality and sustainability of your polymer additive portfolio.
