Advanced Synthesis and Purification of Etodolac Photodegradation Impurities for Global Quality Control
The pharmaceutical industry faces constant challenges in maintaining the stability and safety of active pharmaceutical ingredients (APIs) throughout their shelf life. A critical aspect of this quality assurance involves the rigorous identification and quantification of degradation products that may form under various stress conditions. Patent CN109485616B addresses a significant gap in the analytical control of Etodolac, a widely used non-steroidal anti-inflammatory drug (NSAID). This patent discloses a robust method for preparing specific photodegradation impurities, particularly the compound designated as Formula I, which serves as a vital reference standard for stability testing. By leveraging advanced supercritical fluid chromatography (SFC) techniques, the inventors have overcome the historical difficulties associated with isolating these labile compounds, providing a reliable etodolac impurity supplier pathway for global quality control laboratories.
Etodolac is known to undergo complex degradation pathways when exposed to light, heat, and oxidative environments. Traditional methods for generating these impurities often resulted in complex mixtures where the target degradation product was present in trace amounts, making isolation nearly impossible. The breakthrough described in this patent lies in the optimization of the photolytic conditions combined with a gentle yet highly efficient purification strategy. This ensures that the resulting reference standards possess the high purity required for regulatory submissions and method validation, directly supporting cost reduction in pharmaceutical intermediate manufacturing by reducing the need for repetitive synthesis attempts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of Etodolac degradation impurities relied heavily on forced degradation studies followed by preparative high-performance liquid chromatography (HPLC). However, this approach suffers from severe limitations when applied to photo-labile compounds. The target photodegradation product, Formula I, is chemically unstable and prone to further decomposition when subjected to the prolonged retention times and aqueous mobile phases typical of reverse-phase HPLC. Furthermore, the use of traditional silica gel column chromatography often leads to acid-catalyzed degradation due to the acidic nature of silica, resulting in low recovery yields and poor purity profiles. These inefficiencies create a bottleneck in the supply chain for reference standards, forcing quality control teams to rely on less characterized materials or endure long lead times for custom synthesis.
The Novel Approach
The patented methodology introduces a paradigm shift by utilizing Supercritical Fluid Chromatography (SFC) as the primary purification tool. Unlike liquid chromatography, SFC uses supercritical carbon dioxide as the main mobile phase component, which offers distinct advantages such as low viscosity and high diffusivity. This allows for rapid separation at lower temperatures, significantly minimizing the thermal and chemical stress on the sensitive photodegradation impurity. By optimizing the modifier and additive system—specifically using alcohols and trace acids—the process achieves exceptional selectivity. This novel approach not only prevents the secondary degradation observed in conventional methods but also simplifies the post-processing workflow, as the volatile CO2 evaporates easily, leaving behind the pure product without extensive solvent removal steps.
Mechanistic Insights into Photo-oxidative Degradation and SFC Separation
The formation of the Formula I impurity involves a complex photo-oxidative rearrangement of the Etodolac molecule. Under aerobic conditions and light irradiation, the electron-rich indole ring system of Etodolac is susceptible to singlet oxygen attack or radical-mediated oxidation. This initial oxidative stress triggers a ring-expansion or cleavage sequence that ultimately yields the pyran-carboxylic acid derivative structure seen in Formula I. The addition of hydrogen peroxide acts as a catalyst in this radical chain reaction, accelerating the conversion rate and driving the equilibrium towards the desired degradation product. Understanding this mechanism is crucial for scaling the reaction, as controlling the oxygen flux and light intensity determines the ratio of the target impurity versus other unknown byproducts.
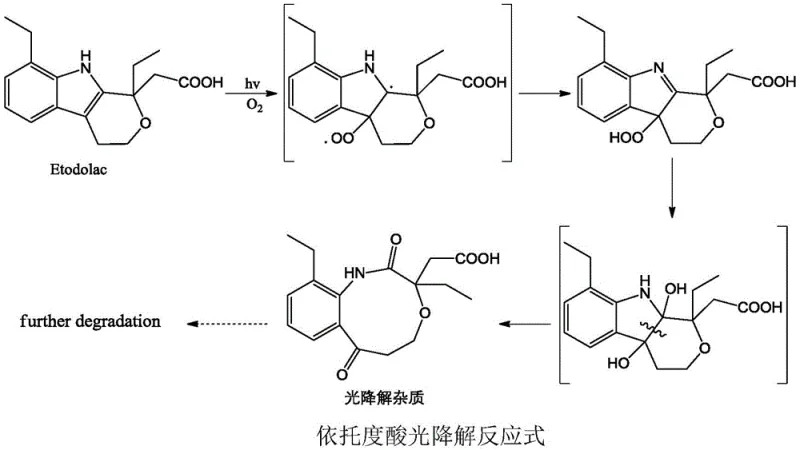
Once the crude mixture is generated, the separation mechanism relies on the subtle differences in polarity and steric interaction between the target impurity and the remaining Etodolac. In the SFC system, the supercritical CO2 acts as a non-polar solvent, while the organic modifier adjusts the solvating power. The target impurity, possessing a more polar carboxylic acid moiety and a modified ring structure compared to the parent drug, interacts differently with the stationary phase (such as diol-silica or amino-silica). This differential adsorption allows for the precise elution of the impurity peak. The use of additives like formic acid helps to suppress peak tailing caused by the acidic protons on the analyte, ensuring sharp resolution and high collection purity. This mechanistic control is what enables the process to achieve purification yields exceeding 90%.
How to Synthesize Etodolac Photodegradation Impurity Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing high-purity Formula I. It begins with the dissolution of Etodolac in a suitable organic solvent such as acetonitrile or tetrahydrofuran. To enhance the degradation efficiency, a controlled amount of hydrogen peroxide is introduced, followed by exposure to high-intensity light sources (3000-6000 Lx) for a duration of 10 to 20 hours. This step converts a significant portion of the starting material into the target impurity. The subsequent purification via SFC requires careful tuning of the back pressure and temperature to maintain the supercritical state of the mobile phase. Detailed standardized synthesis steps for this process are provided in the guide below.
- Dissolve Etodolac in a polar aprotic solvent or alkanol, optionally adding hydrogen peroxide to accelerate oxidative degradation under aerobic conditions.
- Subject the solution to controlled light irradiation (3000-6000 Lx) for 10-20 hours to induce specific photodegradation into the target Formula I structure.
- Purify the resulting mixture using Supercritical Fluid Chromatography (SFC) with a CO2-based mobile phase to isolate the impurity with over 90% recovery yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this SFC-based purification technology offers tangible strategic benefits beyond mere technical superiority. The primary advantage lies in the drastic simplification of the downstream processing. Traditional purification methods often require multiple recrystallizations or sequential column chromatography runs to achieve acceptable purity, each step incurring material loss and labor costs. By contrast, the single-pass efficiency of the SFC method significantly reduces the operational footprint and solvent consumption. This streamlined workflow translates directly into a more predictable production schedule and reduced dependency on scarce chromatographic resins that degrade quickly under harsh acidic conditions.
- Cost Reduction in Manufacturing: The elimination of extensive solvent evaporation steps and the high recovery yield of the SFC process contribute to substantial cost savings. Since supercritical CO2 is recycled within the system, the consumption of expensive organic solvents is minimized compared to large-scale preparative HPLC. Furthermore, the prevention of product degradation during purification means that less starting material is wasted, optimizing the overall atom economy of the reference standard production. This efficiency allows for a more competitive pricing structure for high-purity specialty chemicals without compromising on quality margins.
- Enhanced Supply Chain Reliability: The robustness of the photo-degradation reaction, assisted by hydrogen peroxide, ensures consistent batch-to-batch reproducibility. In the context of supplying reference standards, consistency is paramount. The ability to generate the impurity in high abundance (>50% in crude) reduces the risk of supply shortages caused by low-yielding synthesis routes. Additionally, the raw materials required—Etodolac API, common solvents, and hydrogen peroxide—are commodity chemicals with stable global supply chains, mitigating the risk of raw material bottlenecks that often plague complex synthetic intermediates.
- Scalability and Environmental Compliance: Supercritical fluid technology is inherently greener than traditional liquid chromatography, aligning with modern environmental, social, and governance (ESG) goals. The reduction in hazardous waste generation, particularly halogenated solvents often used in HPLC, simplifies waste disposal compliance. From a scalability perspective, SFC systems can be scaled up from analytical to preparative scales with minimal method re-development. This scalability ensures that the commercial scale-up of complex pharmaceutical impurities can be executed rapidly to meet surging demand from regulatory bodies requiring larger quantities of qualified reference materials.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability and handling of Etodolac photodegradation impurities. These insights are derived directly from the experimental data and optimization studies detailed in the patent literature. Understanding these nuances is essential for laboratory personnel responsible for the storage and utilization of these sensitive reference standards in stability-indicating methods.
Q: Why is Supercritical Fluid Chromatography preferred over HPLC for this impurity?
A: Traditional reverse-phase HPLC often leads to further degradation of the unstable photodegradation product during the lengthy separation process. SFC offers faster separation with lower thermal stress and easier solvent removal, preserving the integrity of the labile Formula I compound.
Q: What is the achieved purity of the Etodolac photodegradation impurity?
A: Using the patented SFC method, the purity of the isolated Etodolac photodegradation impurity can reach as high as 99.01%, making it suitable for use as a certified reference standard in analytical testing.
Q: How does the addition of hydrogen peroxide affect the reaction?
A: Adding a small amount of hydrogen peroxide (1-5% mass concentration) acts as an oxidant assistant, significantly enhancing the photodegradation efficiency and increasing the content of the target Formula I impurity to over 50% in the crude mixture.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Etodolac Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize that the availability of high-quality impurity standards is the cornerstone of pharmaceutical safety. Our technical team has extensively analyzed the patented SFC purification route and integrated similar green chemistry principles into our own manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need milligrams for method validation or grams for stability studies, our supply is uninterrupted. Our facilities are equipped with rigorous QC labs and stringent purity specifications that match or exceed the 99.01% purity benchmark set by the latest industry patents.
We invite global partners to collaborate with us to secure their supply chains for critical reference materials. By leveraging our expertise in photochemistry and supercritical fluid technologies, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for Etodolac impurities and other complex pharmaceutical intermediates.
