Advanced Synthesis of Astilbin Derivatives for Enhanced Bioavailability and Commercial Scalability
Advanced Synthesis of Astilbin Derivatives for Enhanced Bioavailability and Commercial Scalability
The pharmaceutical industry continuously seeks solutions to overcome the bioavailability limitations of potent natural products, and patent CN101638424A presents a groundbreaking approach to modifying astilbin, a dihydroflavonol with significant therapeutic potential. While astilbin is known for its robust immunomodulatory, anti-inflammatory, and analgesic properties, its clinical utility has historically been constrained by extremely poor water solubility. This comprehensive technical insight analyzes the novel synthetic pathways disclosed in the patent, which systematically modify the phenolic hydroxyl groups, phenyl rings, and carbonyl functionalities of the astilbin scaffold. By transforming the molecular architecture through esterification, phosphorylation, and sulfonation, these derivatives offer a viable pathway to create high-purity pharmaceutical intermediates that maintain biological efficacy while achieving the solubility profiles required for modern drug delivery systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional utilization of native astilbin in drug formulation faces severe thermodynamic and kinetic barriers due to its hydrophobic nature. In conventional processing, the inability of astilbin to dissolve adequately in aqueous media leads to inconsistent dosing, poor absorption rates, and the necessity for complex solubilizing excipients that may introduce toxicity or stability issues. Furthermore, direct isolation from plant sources often yields material with variable purity and batch-to-batch inconsistency, complicating the regulatory approval process for new drug applications. The lack of functional handles on the unmodified molecule limits the ability of formulators to create prodrugs or salt forms that could otherwise enhance pharmacokinetic profiles, effectively capping the commercial value of this promising bioactive compound in the competitive landscape of pharmaceutical intermediates.
The Novel Approach
The methodology outlined in patent CN101638424A circumvents these solubility bottlenecks by introducing hydrophilic moieties directly onto the flavonoid backbone through robust organic synthesis techniques. Instead of relying on physical mixture methods, this chemical approach covalently attaches groups such as carboxymethyl esters, phosphate groups, and sulfonate salts to the astilbin core. This strategic modification not only drastically improves water solubility but also opens new avenues for structure-activity relationship (SAR) studies, allowing medicinal chemists to optimize potency and metabolic stability. The versatility of this approach means that a single starting material can be diversified into a library of candidates, providing a reliable astilbin derivative supplier with the flexibility to meet diverse client specifications for both preclinical research and commercial API manufacturing without the need for developing entirely new synthetic routes for each analog.
Mechanistic Insights into Functional Group Modification Strategies
The core of this technological advancement lies in the selective nucleophilic substitution and electrophilic aromatic substitution reactions targeting specific sites on the astilbin molecule. For phenolic hydroxyl modification, the process typically involves deprotonation of the hydroxyl groups using bases like sodium hydroxide or potassium carbonate, generating a nucleophilic phenoxide ion that attacks electrophiles such as ethyl chloroacetate or alkyl halides. This O-alkylation or O-acylation is highly efficient, often proceeding to completion within hours at moderate temperatures, yielding stable ester or ether linkages that can be further hydrolyzed to free acids if needed. The precision of these reactions ensures that the stereochemistry at the chiral centers of the flavonoid skeleton remains intact, preserving the biological activity inherent to the natural product while altering its physicochemical properties.
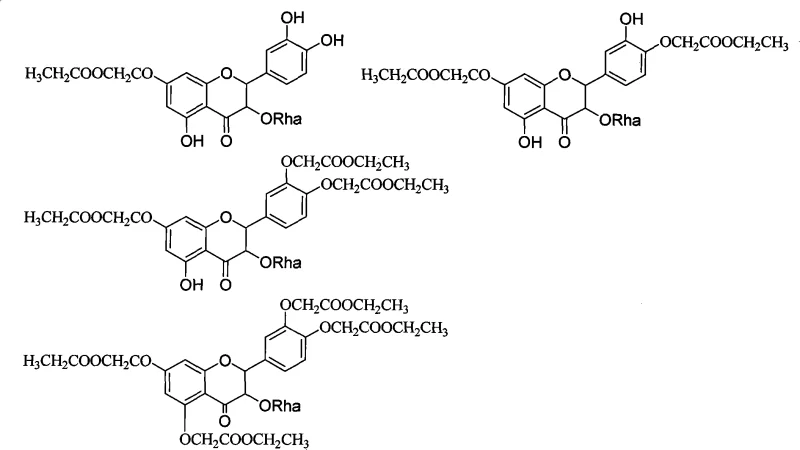
In addition to esterification, the patent details sophisticated phosphorylation and sulfonation protocols that introduce strong anionic character to the molecule. Phosphorylation is achieved by reacting astilbin with phosphorus oxychloride in acetonitrile at low temperatures, followed by hydrolysis to generate phosphate monoesters or salts. Similarly, sulfonation utilizes concentrated sulfuric acid to install sulfonic acid groups on the aromatic rings, which are subsequently neutralized to sodium salts. These transformations are critical for creating water-soluble salts suitable for injectable formulations. The mechanistic diversity allows for the creation of prodrugs that may undergo enzymatic cleavage in vivo to release the active parent compound, thereby combining improved solubility with targeted release mechanisms.
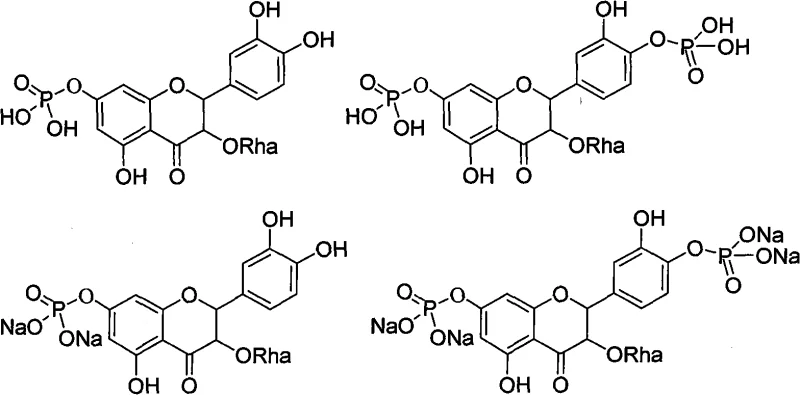
Furthermore, the modification of the phenyl ring through Friedel-Crafts reactions and chloromethylation expands the chemical space available for conjugation. By introducing chloromethyl or acyl groups onto the B-ring of the flavonoid, the technology enables subsequent coupling with amines or other nucleophiles, facilitating the creation of complex conjugates. The control over reaction conditions, such as maintaining temperatures between -5°C to 10°C for phosphorylation or using specific catalysts like zinc chloride for Friedel-Crafts alkylation, demonstrates a deep understanding of reaction kinetics to minimize side products. This level of control is essential for ensuring high purity standards, as impurities in pharmaceutical intermediates can have significant downstream effects on drug safety and efficacy profiles.
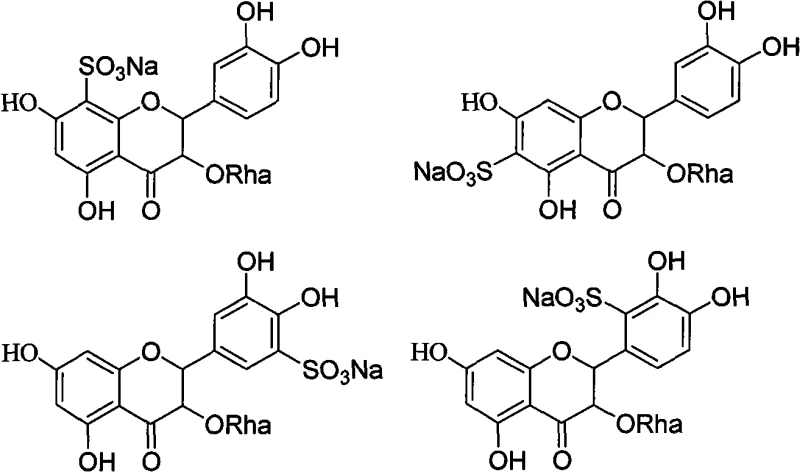
How to Synthesize Astilbin Derivatives Efficiently
The synthesis of these high-value derivatives follows a logical progression of dissolution, reaction, and purification that is amenable to both laboratory and pilot-scale operations. The process begins with the careful selection of solvent systems—ranging from polar aprotic solvents like DMF for esterification to aqueous alkaline solutions for etherification—to ensure complete solubilization of the starting astilbin. Following the addition of reagents, reaction progress is meticulously monitored using thin-layer chromatography (TLC) to determine the exact endpoint, preventing over-reaction or degradation of the sensitive flavonoid core. Detailed standardized synthesis steps see the guide below for specific operational parameters.
- Dissolve astilbin in a suitable solvent system such as sodium hydroxide solution, DMF, or pyridine depending on the target functional group modification.
- Introduce the modifying reagent (e.g., ethyl chloroacetate, phosphorus oxychloride, or chlorosulfonic acid) under controlled temperature conditions ranging from ice bath to reflux.
- Quench the reaction, isolate the crude product via precipitation or extraction, and purify using recrystallization or column chromatography to obtain high-purity derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of these synthetic routes offers substantial strategic advantages primarily driven by the availability and cost of raw materials. The reagents required, such as ethyl chloroacetate, succinic anhydride, and phosphorus oxychloride, are commodity chemicals produced on a massive global scale, ensuring a stable supply chain with minimal risk of shortage or price volatility. This reliance on established industrial chemicals rather than exotic or proprietary catalysts significantly lowers the barrier to entry for manufacturing, allowing for cost reduction in pharmaceutical intermediate manufacturing by eliminating the need for expensive transition metal catalysts and the associated heavy metal removal steps that often complicate downstream processing and regulatory compliance.
- Cost Reduction in Manufacturing: The synthetic pathways described eliminate the need for complex multi-step total synthesis, instead utilizing semi-synthetic modification of naturally derived or fermentation-derived astilbin. This approach leverages the existing chiral pool of the natural product, avoiding the high costs associated with asymmetric synthesis or chiral resolution. Furthermore, the workup procedures often involve simple precipitation or crystallization rather than energy-intensive distillation or complex chromatographic separations, leading to substantial cost savings in utility consumption and waste treatment. The ability to produce water-soluble derivatives also reduces formulation costs for downstream customers, creating additional value across the supply chain.
- Enhanced Supply Chain Reliability: By diversifying the product portfolio to include various esters, phosphates, and sulfonates, suppliers can mitigate risks associated with demand fluctuations for any single specific compound. The robustness of the chemical reactions, which tolerate standard industrial equipment materials like glass-lined steel reactors, ensures that production can be scaled up rapidly without requiring specialized infrastructure. This flexibility allows for responsive manufacturing capabilities, reducing lead time for high-purity astilbin derivatives and enabling just-in-time delivery models that align with the lean inventory strategies of modern pharmaceutical companies.
- Scalability and Environmental Compliance: The processes described generally operate at atmospheric pressure and moderate temperatures, reducing the safety risks and energy costs associated with high-pressure hydrogenation or cryogenic reactions. Waste streams are primarily composed of aqueous salts and organic solvents that can be recovered and recycled using standard distillation units, supporting sustainability goals and environmental compliance. The absence of toxic heavy metals in the catalytic systems simplifies the environmental impact assessment and facilitates easier permitting for new manufacturing facilities, ensuring long-term operational continuity in increasingly regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized flavonoid derivatives. Understanding the nuances of solubility enhancement and synthetic scalability is crucial for stakeholders evaluating these materials for drug development pipelines. The answers provided are grounded in the specific technical disclosures of the patent, ensuring accuracy and relevance for decision-makers.
Q: Why is chemical modification necessary for Astilbin?
A: Native astilbin exhibits potent pharmacological activities including immunomodulation and anti-inflammatory effects, but its clinical application is severely restricted by poor water solubility. Chemical derivatization, such as phosphorylation or esterification, introduces polar groups that significantly enhance aqueous solubility and bioavailability.
Q: Are the reagents used in this synthesis scalable for industrial production?
A: Yes, the synthesis routes described in patent CN101638424A utilize common industrial reagents such as ethyl chloroacetate, phosphorus oxychloride, and sulfuric acid. These materials are readily available in bulk quantities, ensuring supply chain stability and cost-effectiveness for large-scale manufacturing.
Q: What types of derivatives can be produced using this technology?
A: The technology enables the production of a diverse library of derivatives including phenolic hydroxyl ethers and esters, phenyl ring sulfonates and chloromethylates, and carbonyl-substituted enamines. This versatility allows for fine-tuning physicochemical properties to meet specific formulation requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Astilbin Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex academic patents into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to handle the nuanced chemistry of flavonoid modification, ensuring that every batch meets stringent purity specifications through our rigorous QC labs equipped with state-of-the-art analytical instrumentation. We understand that consistency is key in the pharmaceutical supply chain, and our robust quality management systems guarantee that the physicochemical properties of our astilbin derivatives remain consistent from pilot batches to full commercial scale, providing our partners with the confidence needed to advance their clinical programs.
We invite global pharmaceutical and agrochemical companies to collaborate with us to unlock the full potential of these soluble astilbin analogs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can accelerate your project timelines while maximizing value efficiency.
