Advanced Synthesis of 4,5-Dimethyl-3-Aminoisoxazole: Technical Upgrade and Commercial Scalability
Advanced Synthesis of 4,5-Dimethyl-3-Aminoisoxazole: Technical Upgrade and Commercial Scalability
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for critical heterocyclic intermediates. A recent breakthrough detailed in patent CN115010676A introduces a novel preparation method for 4,5-dimethyl-3-aminoisoxazole, a key building block for antibacterial drugs such as sulfisoxazole. This technology represents a significant paradigm shift from traditional synthetic routes, addressing long-standing challenges related to raw material availability, safety hazards, and overall process efficiency. By leveraging a streamlined three-step sequence involving condensation, copper-catalyzed oxidation, and cyclization, this method achieves superior yields and operational simplicity. For global procurement and R&D teams, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials with consistent supply continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4,5-dimethyl-3-aminoisoxazole has been plagued by significant technical and economic hurdles. Prior art, such as the route reported in Chinese patent CN110357830A, relies on a mixture of 2-methyl-2-butenenitrile and 2-methyl-3-butenenitrile as starting materials. These precursors are not only difficult to source commercially but also require complex purification before use. Furthermore, this legacy process necessitates the use of liquid bromine, a highly corrosive and hazardous reagent that demands specialized equipment and rigorous safety protocols, thereby inflating capital expenditure and operational risks. Another traditional approach involves the use of metallic sodium to synthesize sodium propionitrile, introducing severe safety liabilities due to the pyrophoric nature of the metal. Additionally, routes utilizing 3-chlorobutanone and sodium cyanide suffer from low yields and the handling of extremely toxic cyanide salts, making them unsuitable for modern, environmentally conscious manufacturing facilities.
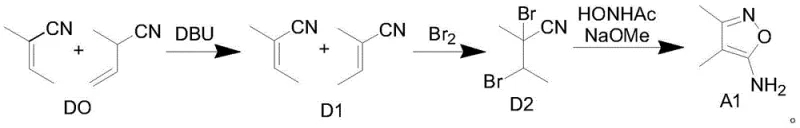
The Novel Approach
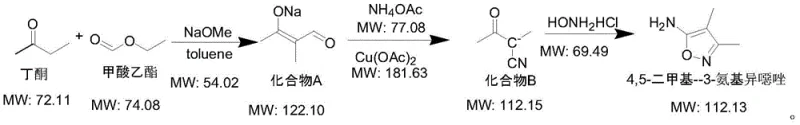
In stark contrast, the methodology disclosed in CN115010676A offers a streamlined and economically viable alternative. The new process initiates with the condensation of readily available and inexpensive bulk chemicals—butanone and ethyl formate—under basic conditions to form an aldehyde-type intermediate. This is followed by a sophisticated copper-catalyzed oxidation using ammonium acetate to generate a cyano intermediate, finally culminating in a cyclization reaction with hydroxylamine hydrochloride. This route completely eliminates the need for hazardous bromine, metallic sodium, or toxic cyanides. The overall yield of this novel process reaches up to 73% across three steps, significantly outperforming the 66% yield of the comparative example. The simplicity of the operation, combined with the use of common solvents like toluene, DMSO, and water, drastically reduces the barrier to entry for commercial scale-up of complex heterocycles.
Mechanistic Insights into Copper-Catalyzed Oxidation and Cyclization
The core innovation of this synthesis lies in the second step: the transformation of the aldehyde intermediate (Compound A) into the cyano intermediate (Compound B). This step employs a copper salt catalyst, such as copper acetate, in the presence of ammonium acetate within a polar aprotic solvent like DMSO. Mechanistically, this likely involves the formation of an imine or enamine species facilitated by ammonium acetate, followed by a copper-mediated oxidative dehydrogenation or functional group interconversion that installs the nitrile moiety. The choice of copper acetate is critical; it acts as a mild Lewis acid and redox mediator, enabling the reaction to proceed efficiently at moderate temperatures of 60-70°C. This温和 condition is pivotal for minimizing thermal degradation of the sensitive aldehyde precursor, thereby preserving the integrity of the carbon skeleton and preventing the formation of polymeric byproducts that often plague high-temperature condensations.
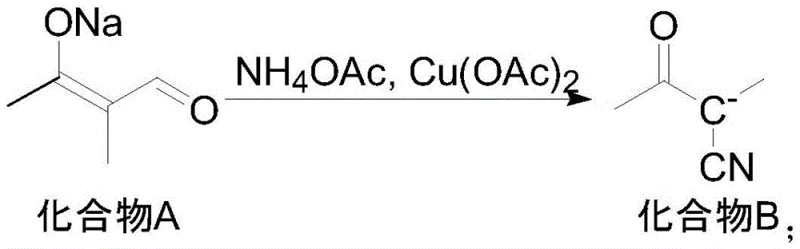
Furthermore, the final cyclization step demonstrates excellent chemoselectivity. The reaction of the cyano intermediate with hydroxylamine hydrochloride in an aqueous medium at 20-30°C ensures the precise formation of the isoxazole ring without affecting the methyl substituents. The use of water as a solvent in this final stage is particularly advantageous from a green chemistry perspective, eliminating the need for volatile organic solvents during the product isolation phase. The precipitation of the solid product directly from the aqueous reaction mixture simplifies the work-up procedure to a mere filtration and drying step. This inherent ease of purification contributes significantly to the high purity of the final API intermediate, reducing the burden on downstream quality control laboratories and ensuring that the material meets the stringent specifications required for antibiotic synthesis.
How to Synthesize 4,5-Dimethyl-3-Aminoisoxazole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process is divided into three distinct stages, each optimized for maximum conversion and minimal waste generation. The initial condensation requires careful control of the addition rate of the ketone and ester mixture to the alkoxide base to manage exothermicity. Subsequent steps leverage the stability of the intermediates to allow for flexible processing windows. For detailed operational parameters, stoichiometry, and specific work-up procedures, please refer to the standardized guide below.
- Condense butanone and ethyl formate using sodium methoxide in toluene at 60-70°C to obtain aldehyde intermediate A.
- React intermediate A with ammonium acetate and copper acetate in DMSO at 60-70°C to synthesize cyano intermediate B.
- Cyclize intermediate B with hydroxylamine hydrochloride in water at 20-30°C to isolate the final 4,5-dimethyl-3-aminoisoxazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits beyond mere chemical elegance. The shift away from specialty reagents like liquid bromine and difficult-to-source nitriles towards commodity chemicals like butanone and ethyl formate fundamentally alters the cost structure of the supply chain. This transition mitigates the risk of supply disruptions caused by the limited availability of niche starting materials, ensuring a more resilient and continuous flow of goods. Moreover, the elimination of hazardous reagents reduces the regulatory burden and insurance costs associated with storage and transport, contributing to a leaner operational model.
- Cost Reduction in Manufacturing: The new process achieves cost reduction in API manufacturing through multiple vectors. Firstly, the raw materials (butanone, ethyl formate) are commodity chemicals with stable pricing and abundant global supply, unlike the proprietary or scarce mixtures required by older methods. Secondly, the removal of liquid bromine eliminates the need for expensive corrosion-resistant reactors and complex scrubbing systems for bromine vapors. Thirdly, the higher overall yield (up to 73%) means less raw material is wasted per kilogram of final product, directly improving the cost of goods sold (COGS). Finally, the simplified work-up procedures, particularly the aqueous crystallization in the final step, reduce solvent consumption and energy usage for distillation, further driving down production expenses.
- Enhanced Supply Chain Reliability: Supply chain stability is paramount for pharmaceutical production. By relying on widely available feedstocks, this method reduces lead time for high-purity intermediates by removing dependencies on single-source suppliers of exotic reagents. The robustness of the reaction conditions—operating at atmospheric pressure and moderate temperatures—means the process can be easily replicated across different manufacturing sites globally without requiring highly specialized infrastructure. This geographic flexibility allows for diversified sourcing strategies, protecting the supply chain against regional disruptions or logistical bottlenecks. Additionally, the absence of pyrophoric metals like sodium removes a major safety bottleneck that often delays production scheduling and increases insurance premiums.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the green credentials of a synthesis route become a critical factor for long-term viability. This process generates significantly less hazardous waste compared to cyanide-based or bromination routes. The use of water in the final step and the ability to recover solvents like toluene and DMSO aligns with modern sustainability goals. The process is inherently safer, reducing the risk of catastrophic accidents, which in turn facilitates smoother permitting and faster scale-up from pilot plants to multi-ton commercial production. This environmental and safety compliance ensures uninterrupted operations and protects the brand reputation of downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety profiles, and scalability potential for decision-makers evaluating this route for their supply chains.
Q: What are the primary advantages of the new synthesis route over traditional methods?
A: The new route described in patent CN115010676A eliminates the use of hazardous liquid bromine and expensive, hard-to-source starting materials like 2-methyl-2-butenenitrile. It utilizes readily available butanone and ethyl formate, significantly improving safety profiles and reducing raw material costs while achieving higher overall yields (up to 73% vs 66%).
Q: How does the copper-catalyzed step impact product purity?
A: The use of copper acetate as a catalyst in the oxidation step allows for mild reaction conditions (60-70°C) compared to harsher traditional methods. This controlled environment minimizes side reactions and decomposition, leading to a cleaner reaction profile and facilitating easier downstream purification to meet stringent pharmaceutical specifications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids pyrophoric reagents like metallic sodium and toxic cyanides found in prior art. The use of common solvents like toluene, DMSO, and water, combined with moderate temperature ranges, makes the transition from laboratory to multi-ton commercial production straightforward and compliant with modern environmental standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Dimethyl-3-Aminoisoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antibiotics. Our technical team has thoroughly analyzed the pathway described in CN115010676A and is fully prepared to implement this advanced synthesis method. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from R&D to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4,5-dimethyl-3-aminoisoxazole meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for sulfisoxazole and related antibacterial agents. By leveraging our expertise in this novel copper-catalyzed route, we can offer you a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us be your partner in delivering efficient, safe, and cost-effective chemical solutions.
