Scaling High-Efficiency P-N-S-Br Synergistic Flame Retardants for Industrial Polymer Applications
Scaling High-Efficiency P-N-S-Br Synergistic Flame Retardants for Industrial Polymer Applications
The global demand for high-performance polymeric materials has necessitated the development of advanced flame retardant technologies that balance fire safety with environmental compliance and material integrity. Patent CN101967162B introduces a groundbreaking synergistic flame retardant compound, phenylthiophosphonobis(2,4,6-tribromophenyl)amine, which uniquely integrates Phosphorus, Nitrogen, Sulfur, and Bromine elements into a single molecular architecture. This tetra-element synergy addresses the critical limitations of traditional halogenated additives by enhancing thermal stability and char formation while maintaining excellent compatibility with polymer matrices. The structural complexity of this molecule, featuring a central thiophosphonyl group bridged to two heavily brominated aniline moieties, allows it to function through multiple flame-retardant mechanisms simultaneously, including gas-phase radical quenching and condensed-phase char promotion.
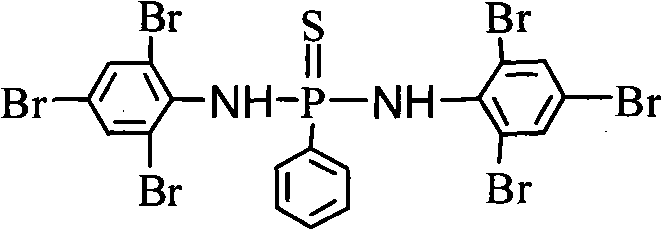
For R&D directors and formulation scientists, the significance of this patent lies in its ability to overcome the migration and volatility issues often associated with smaller brominated molecules. The high molecular weight and symmetrical structure of phenylthiophosphonobis(2,4,6-tribromophenyl)amine ensure that it remains embedded within the polymer host during processing and service life. Furthermore, the presence of the P-N bond creates a stable amide-like linkage that resists hydrolysis, thereby extending the lifespan of the final plastic or resin product. As a reliable flame retardant intermediate supplier, understanding the precise synthesis and scale-up potential of such complex molecules is paramount for securing a competitive edge in the specialty chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing organophosphorus flame retardants often suffer from significant drawbacks regarding reaction efficiency and product purity. Conventional methods typically rely on harsh reaction conditions or expensive transition metal catalysts that leave behind toxic residues, necessitating costly purification steps to meet stringent regulatory standards for electronic or automotive applications. Moreover, standard bromination processes frequently result in mixtures of isomers or incomplete substitution, leading to inconsistent flame retardant performance and unpredictable effects on the mechanical properties of the base polymer. The lack of synergistic elements in older generations of additives means that higher loading levels are required to achieve the same UL-94 rating, which can degrade the tensile strength and impact resistance of the final material.
The Novel Approach
The methodology described in CN101967162B represents a paradigm shift by utilizing a direct condensation reaction between phenylphosphonothioic dichloride and 2,4,6-tribromoaniline. This novel approach leverages the inherent reactivity of the acid chloride functionality while mitigating the deactivating effects of the bromine substituents through the use of a specialized catalyst system. By employing low hydrated zinc borate as a catalyst, the process achieves high conversion rates without the need for precious metals, significantly simplifying the downstream purification workflow. The reaction is conducted in common industrial solvents such as xylene or toluene, which facilitates easy solvent recovery and recycling, aligning with green chemistry principles. This streamlined synthetic route not only reduces the overall production cost but also ensures a consistent supply of high-purity intermediates essential for manufacturing high-performance engineering plastics.
Mechanistic Insights into Catalytic Amidation with Zinc Borate
The core chemical transformation in this patent is a nucleophilic substitution reaction where the amino group of 2,4,6-tribromoaniline attacks the phosphorus center of phenylphosphonothioic dichloride. However, the presence of three electronegative bromine atoms on the aniline ring exerts a powerful electron-withdrawing inductive effect, which drastically reduces the electron density on the nitrogen lone pair. This deactivation makes the aniline a poor nucleophile, typically rendering the amidation reaction sluggish or requiring prohibitively high temperatures that could lead to decomposition. The innovation here lies in the strategic use of low hydrated zinc borate (2ZnO·3B2O3·3.5H2O) as a Lewis acid catalyst. The zinc centers coordinate with the sulfur or chlorine atoms on the phosphorus reagent, increasing the electrophilicity of the phosphorus atom and lowering the activation energy for the nucleophilic attack.
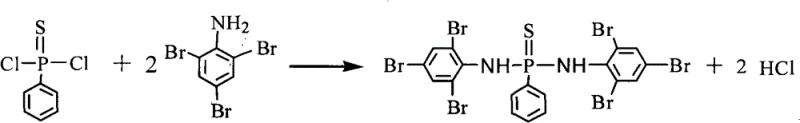
Furthermore, the reaction mechanism involves the simultaneous neutralization of the hydrogen chloride byproduct by an acid-binding agent such as pyridine or triethylamine. This scavenging action prevents the protonation of the aniline reactant, which would otherwise completely shut down the nucleophilic pathway. The synergy between the zinc borate catalyst and the organic base creates a balanced reaction environment that promotes the formation of the P-N bond while suppressing side reactions like hydrolysis of the acid chloride. Detailed kinetic studies suggest that maintaining the reaction temperature between 110°C and 146°C is critical; lower temperatures result in incomplete conversion, while excessive heat may promote degradation of the sensitive P-S bond. This precise control over the reaction parameters ensures the formation of the desired symmetrical bis-amide structure with minimal impurities.
How to Synthesize Phenylthiophosphonobis(2,4,6-tribromophenyl)amine Efficiently
Implementing this synthesis on a commercial scale requires strict adherence to the optimized parameters outlined in the patent examples to ensure reproducibility and safety. The process begins with the preparation of a reaction mixture containing the tribromoaniline substrate, the acid binder, and the zinc borate catalyst suspended in a high-boiling aromatic solvent. Under a protective nitrogen atmosphere to prevent oxidation and moisture ingress, the phenylphosphonothioic dichloride is introduced slowly to manage the exotherm and control the evolution of HCl gas. Following the addition, the system is heated to reflux for an extended period, typically ranging from 6 to 14 hours, to drive the equilibrium towards the product. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the technical guide below.
- Charge a reactor with 2,4,6-tribromoaniline, acid-binding agent (e.g., pyridine), low hydrated zinc borate catalyst, and organic solvent (xylene or toluene) under nitrogen atmosphere.
- Heat the mixture to 100°C and slowly add phenylphosphonothioic dichloride dropwise over approximately 1 hour while maintaining stirring.
- Raise temperature to reflux (110-146°C depending on solvent) for 6-14 hours, then purify via distillation, crystallization, and ethanol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthesis route offers substantial strategic benefits beyond mere technical performance. The reliance on commodity chemicals such as 2,4,6-tribromoaniline and phenylphosphonothioic dichloride ensures a robust and resilient supply chain, minimizing the risk of raw material shortages that often plague specialty chemical manufacturing. Additionally, the use of recyclable solvents like xylene and toluene allows for significant operational expenditure reductions through solvent recovery loops, directly impacting the bottom line without compromising product quality. The simplicity of the equipment requirements—standard glass-lined reactors equipped with reflux condensers and dropping funnels—means that existing manufacturing infrastructure can be utilized with minimal capital investment, accelerating the time-to-market for new flame retardant formulations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the ability to recycle solvents directly contribute to a leaner cost structure. By avoiding complex purification steps associated with heavy metal removal, manufacturers can reduce utility consumption and waste disposal fees. The high yield range of 66% to 83% reported in the patent examples indicates a material-efficient process that maximizes the output per batch, further driving down the unit cost of the active ingredient. This economic efficiency makes the final polymer additive more competitive against legacy brominated solutions.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis are widely available from established chemical suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic failure, ensures consistent production output even in large-scale operations. This reliability is crucial for maintaining continuous supply to downstream polymer compounders who operate on just-in-time inventory models. Furthermore, the solid nature of the final product simplifies logistics and storage compared to liquid additives that may require specialized containment.
- Scalability and Environmental Compliance: The process generates hydrogen chloride as the primary byproduct, which is easily captured and neutralized by the acid-binding agent, preventing corrosive emissions. The absence of persistent organic pollutants or heavy metal residues in the final product simplifies regulatory compliance for REACH and RoHS directives. The scalability of the batch process allows for seamless transition from pilot plant trials to multi-ton annual production capacities, supporting the growing demand for eco-friendly yet effective flame retardants in the electronics and automotive sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this synergistic flame retardant. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these nuances is essential for integrating this intermediate into your specific polymer matrix effectively.
Q: Why is low hydrated zinc borate used as a catalyst in this synthesis?
A: The strong electron-withdrawing effect of the three bromine atoms on the aniline ring reduces the nucleophilicity of the nitrogen lone pair, making the amidation reaction inherently slow. Low hydrated zinc borate acts as a Lewis acid catalyst to activate the phosphorus center and facilitate the substitution reaction.
Q: What are the optimal reaction conditions for maximum yield?
A: Experimental data indicates that using xylene as a solvent with pyridine as the acid binder, reacting at 142-146°C for 12-14 hours, yields the highest conversion rates, achieving up to 83% isolated yield of the white powder product.
Q: How does this compound improve upon traditional brominated flame retardants?
A: Unlike simple brominated compounds, this molecule integrates Phosphorus, Nitrogen, Sulfur, and Bromine. This multi-element synergy provides a char-forming mechanism (P-N) alongside radical scavenging (Br), resulting in higher thermal stability and better compatibility with polymer matrices without compromising mechanical properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylthiophosphonobis(2,4,6-tribromophenyl)amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced intermediates play in the development of next-generation flame retardant materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of phenylthiophosphonobis(2,4,6-tribromophenyl)amine meets the exacting standards required for high-performance polymer applications. Our commitment to quality assurance means that you can rely on us as a long-term partner for your specialty chemical sourcing.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your current supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this P-N-S-Br synergistic system. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique manufacturing requirements, ensuring a seamless integration of this high-value intermediate into your production workflow.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
