Advanced Manufacturing of High-Purity Mono-Aryl Phosphates for Pharmaceutical Intermediates
Advanced Manufacturing of High-Purity Mono-Aryl Phosphates for Pharmaceutical Intermediates
The synthesis of aryl phosphate esters represents a critical challenge in the production of high-value pharmaceutical intermediates and fine chemicals, primarily due to the difficulty in controlling selectivity during the phosphorylation of phenols. Patent CN101274942A introduces a groundbreaking methodology that fundamentally alters the reaction environment to favor the exclusive formation of mono-substituted products. By shifting from traditional homogeneous systems to a carefully engineered heterogeneous suspension in non-polar solvents, this technology effectively suppresses the formation of unwanted di- and tri-substituted byproducts. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this innovation offers a pathway to significantly higher purity and reduced downstream processing costs. The following analysis details the mechanistic advantages and commercial viability of this selective phosphorylation technique.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the phosphorylation of phenols using phosphorus oxychloride has been plagued by poor selectivity, resulting in complex mixtures that are difficult and expensive to separate. Conventional techniques often involve dissolving both the phenol and the phosphorylating agent in a mutual solvent, such as pyridine, or converting the phenol into a salt prior to reaction. In these homogeneous environments, the high reactivity of the three chlorine atoms in phosphorus oxychloride leads to uncontrolled successive substitutions. As illustrated in the structural diagrams below, the reaction inevitably produces a mixture of mono-substituted compounds (A), di-substituted compounds (B), and tri-substituted compounds (C). This lack of selectivity not only consumes excessive amounts of raw materials but also generates substantial chemical waste, complicating the purification process and driving up the overall cost of goods sold for the final active ingredient.
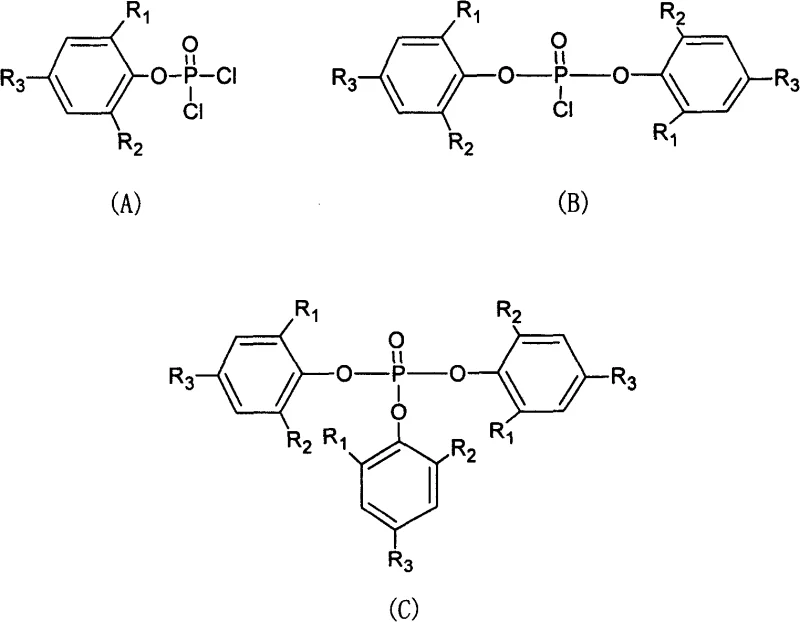
The Novel Approach
The innovative strategy disclosed in the patent overcomes these limitations by exploiting differences in solubility to kinetically control the reaction progression. Instead of forcing all components into a single phase, the method suspends the phenolic starting material in a non-polar solvent, such as toluene, benzene, or xylene, which contains a molar excess of phosphorus oxychloride. Crucially, the phenol is chosen or the conditions are set such that it has very low solubility in this non-polar medium. As the acid binding agent is added dropwise at low temperatures, the reaction occurs primarily at the interface or in the limited dissolved phase. Once the mono-substituted phosphate ester is formed, its low solubility in the non-polar solvent causes it to precipitate or remain suspended, physically removing it from the reactive pool of phosphorus oxychloride. This self-limiting mechanism ensures that the reaction terminates predominantly at the mono-substitution stage, yielding a product of exceptional purity without the need for extensive chromatographic purification.
Mechanistic Insights into Selective Phosphorylation in Non-Polar Media
The core of this technology lies in the thermodynamic and kinetic control exerted by the solvent system. In a standard polar or protic solvent, the transition states for the second and third substitutions are energetically accessible, leading to the statistical distribution of products seen in traditional methods. However, in the non-polar solvent system described, the solvation energy of the intermediate mono-ester is insufficient to keep it in solution, especially in the presence of the non-polar bulk solvent. This precipitation effect acts as a physical barrier against further reaction. Furthermore, the strict temperature control, maintained between 0°C and 30°C, serves to dampen the kinetic energy of the molecules, further suppressing the activation energy required for the less favorable di- and tri-substitution pathways. The slow addition of the acid binding agent, such as pyridine or triethylamine, ensures that the local concentration of the activated phenoxide species remains low, preventing localized hotspots of high reactivity that could lead to over-phosphorylation.
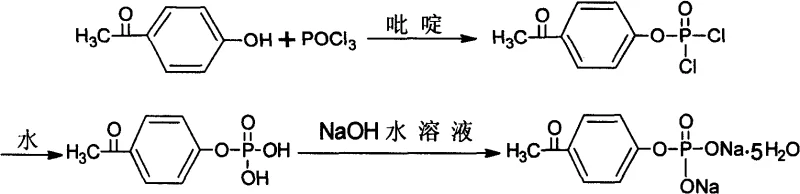
From an impurity control perspective, this mechanism provides a robust defense against the formation of structurally similar byproducts that are notoriously difficult to remove. In the synthesis of sensitive pharmaceutical intermediates, even trace amounts of di-substituted phosphates can act as genotoxic impurities or interfere with downstream coupling reactions. By preventing their formation at the source rather than attempting to remove them later, this process aligns perfectly with Quality by Design (QbD) principles. The hydrolysis step, performed after the initial phosphorylation, converts the chloro-phosphate intermediate into the stable phosphoric acid monoester, which can then be easily isolated as a salt, such as the disodium salt shown in the specific embodiment. This sequence ensures that the final product stream is chemically clean, simplifying the regulatory filing process for drug manufacturers who require rigorous characterization of their supply chain inputs.
How to Synthesize Mono-Aryl Phosphate Efficiently
The operational protocol for this synthesis is designed for reproducibility and safety, utilizing standard equipment found in most fine chemical manufacturing facilities. The process begins with the preparation of a suspension of the phenolic substrate in a non-polar solvent like toluene, followed by the addition of phosphorus oxychloride. The key operational parameter is the maintenance of a low-temperature regime, typically between 10°C and 20°C, during the dropwise addition of the acid scavenger. Detailed standardized synthetic procedures, including specific stoichiometric ratios and workup protocols for various substituted phenols, are outlined in the technical guide below.
- Suspend the phenolic compound in a non-polar solvent (such as toluene or benzene) containing an excess of phosphorus oxychloride, ensuring the phenol remains largely insoluble.
- Maintain the reaction temperature between 0°C and 30°C while slowly dropping in an acid binding agent (like pyridine or triethylamine) to control the reaction rate.
- Hydrolyze the reaction mixture with water, separate the layers, and neutralize with alkali to isolate the high-purity mono-substituted phosphate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates directly into enhanced operational efficiency and cost stability. Traditional methods that yield complex mixtures require energy-intensive distillation or costly chromatography to achieve pharmaceutical-grade purity, creating bottlenecks in production schedules. By contrast, this novel approach simplifies the isolation process to basic filtration and crystallization steps, drastically reducing the cycle time per batch. The ability to achieve high crude yields means that less raw material is wasted on byproducts, leading to a more sustainable and economically viable manufacturing process. This efficiency gain is particularly critical for cost reduction in pharmaceutical intermediate manufacturing, where margin pressures are constant.
- Cost Reduction in Manufacturing: The elimination of complex separation processes significantly lowers the operational expenditure associated with purification. Since the reaction inherently suppresses the formation of di- and tri-substituted impurities, there is no need for expensive preparative HPLC or multiple recrystallization cycles to meet purity specifications. The use of common, inexpensive solvents like toluene and readily available reagents further contributes to a lower bill of materials. Additionally, the high atom economy achieved by minimizing waste generation reduces the costs associated with waste disposal and environmental compliance, providing a comprehensive financial advantage over legacy synthetic routes.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures consistent output quality, which is vital for maintaining uninterrupted supply chains for downstream drug production. Because the reaction conditions are mild and the selectivity is high, the risk of batch failure due to runaway reactions or impurity spikes is minimized. This reliability allows for more accurate forecasting and inventory planning. Furthermore, the raw materials required, such as phosphorus oxychloride and substituted phenols, are commodity chemicals with stable global availability, reducing the risk of supply disruptions caused by niche reagent shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop experiments to multi-ton commercial production without significant re-engineering. The use of non-polar solvents facilitates easier solvent recovery and recycling compared to high-boiling polar solvents like pyridine, which are difficult to remove and recycle. This aligns with modern green chemistry initiatives by reducing the overall solvent footprint and energy consumption of the manufacturing process. The simplified workup also means less aqueous waste is generated, easing the burden on wastewater treatment facilities and ensuring compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aryl phosphate synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process feasibility. Understanding these details is essential for technical teams assessing the integration of this method into existing production lines.
Q: How does this method prevent the formation of di- and tri-substituted byproducts?
A: The method utilizes a heterogeneous system where the phenol and the resulting mono-substituted product have low solubility in the non-polar solvent. This physical state limits the contact between the product and the phosphorylating agent, effectively stopping the reaction at the mono-substitution stage.
Q: What are the typical yields and purity levels achievable with this process?
A: Experimental data from the patent indicates crude yields reaching up to 95.8% with recrystallization yields around 80%. The process inherently minimizes impurities, reducing the need for complex purification steps.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process uses common industrial solvents like toluene and standard reagents like phosphorus oxychloride and pyridine. The low-temperature requirement (0-30°C) is easily manageable in standard jacketed reactors, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mono-Aryl Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of advanced synthetic methods like the one described in CN101274942A are fully realized at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of mono-aryl phosphate meets the exacting standards required by global regulatory bodies. Our commitment to quality ensures that your supply chain remains robust and compliant.
We invite you to collaborate with us to optimize your sourcing strategy for these essential building blocks. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
