Scalable Asymmetric Synthesis of Beclabuvir Intermediate for Global API Manufacturing
The global demand for direct-acting antivirals (DAAs) targeting Hepatitis C Virus (HCV) continues to drive innovation in process chemistry, particularly for non-nucleoside NS5B RNA polymerase inhibitors like Beclabuvir. Patent CN109942584B discloses a groundbreaking synthetic methodology for the key chiral intermediate of Beclabuvir, addressing critical bottlenecks in efficiency and stereocontrol. This technical disclosure outlines a robust four-step sequence starting from readily available beta-bromopropionic acid methyl ester, leveraging advanced asymmetric catalysis to bypass the limitations of classical resolution techniques. For pharmaceutical manufacturers, this represents a pivotal shift towards more sustainable and cost-effective API production, ensuring a stable supply chain for next-generation HCV therapies.
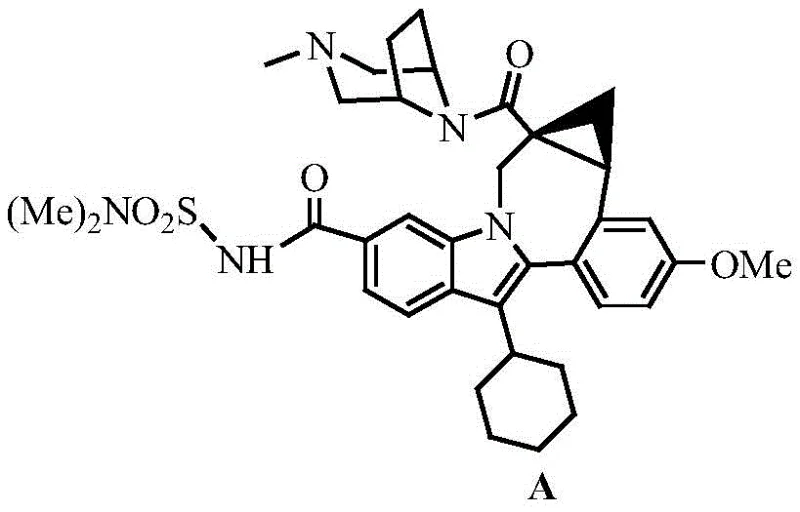
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical cyclopropane-fused indole core found in Beclabuvir has been plagued by inefficiencies inherent to classical organic synthesis strategies. As illustrated in the prior art routes, the conventional pathway typically initiates from a substituted indole ester, proceeding through bromination, amidation, and a Michael addition cyclization to form a precursor. A major drawback of this legacy approach is the reliance on a Corey-Chaykovsky reaction to generate the cyclopropane ring, which yields a racemic mixture requiring subsequent chiral resolution. This resolution step is fundamentally flawed for large-scale manufacturing because it theoretically caps the maximum yield at 50%, discarding half of the valuable material as unwanted isomer waste. Furthermore, the multi-step nature of the traditional route, often exceeding six linear steps with a cumulative yield of merely 16%, exacerbates raw material costs and generates substantial chemical waste, creating significant environmental and economic burdens for production facilities.
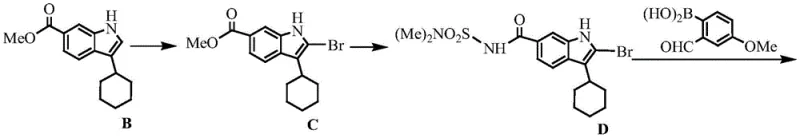
The Novel Approach
In stark contrast, the innovative methodology detailed in the patent introduces a convergent and highly efficient strategy that重构 s the synthetic logic entirely. By utilizing a chiral copper-catalyzed asymmetric cyclopropanation early in the sequence, the process establishes the critical stereocenters with high fidelity before constructing the complex molecular architecture. This new route condenses the synthesis into just four strategic steps, dramatically improving the overall throughput. The elimination of the resolution step alone theoretically doubles the material efficiency compared to the old method. Moreover, the process avoids the use of unstable phosphorus ylides (like phosphate F in the old route) which are prone to polymerization and safety hazards. Instead, it employs mild reaction conditions and stable intermediates, facilitating a smoother transition from laboratory bench to kilogram-scale production while maintaining rigorous quality standards.
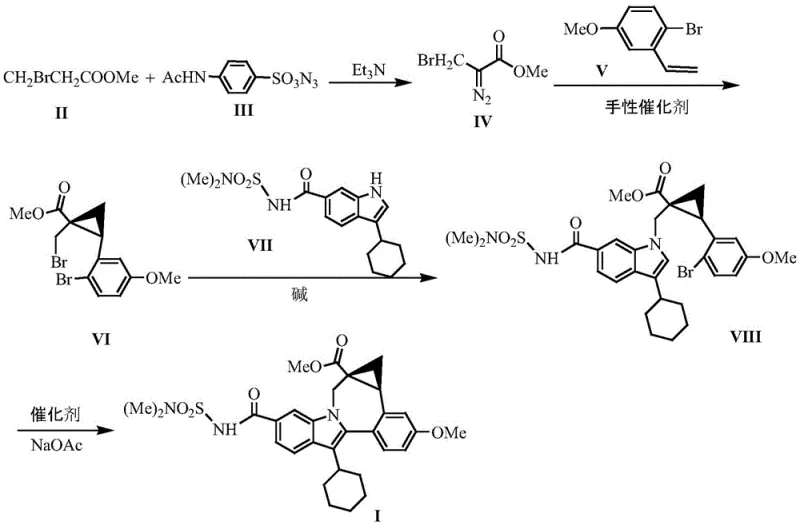
Mechanistic Insights into Chiral Copper-Catalyzed Cyclopropanation
The cornerstone of this technological advancement lies in the deployment of a specialized chiral spiroalkene diketone copper complex, which serves as the engine for stereochemical control. Unlike simple chiral ligands, this catalyst features a unique spiro-enone structure that creates a rigid, seven-membered chiral environment upon coordination with the copper metal center. This specific geometric arrangement is crucial for differentiating the enantiofaces of the diazo compound during the carbene transfer process. The surface chirality of the ligand ensures that the cyclopropanation reaction proceeds with exceptional selectivity, consistently delivering enantiomeric excess (ee) values exceeding 94% in the early intermediate stages and culminating in >99% ee in the final product. The robustness of this catalytic system allows it to function effectively at moderate temperatures between 40°C and 60°C, reducing energy consumption and minimizing thermal degradation of sensitive functional groups.
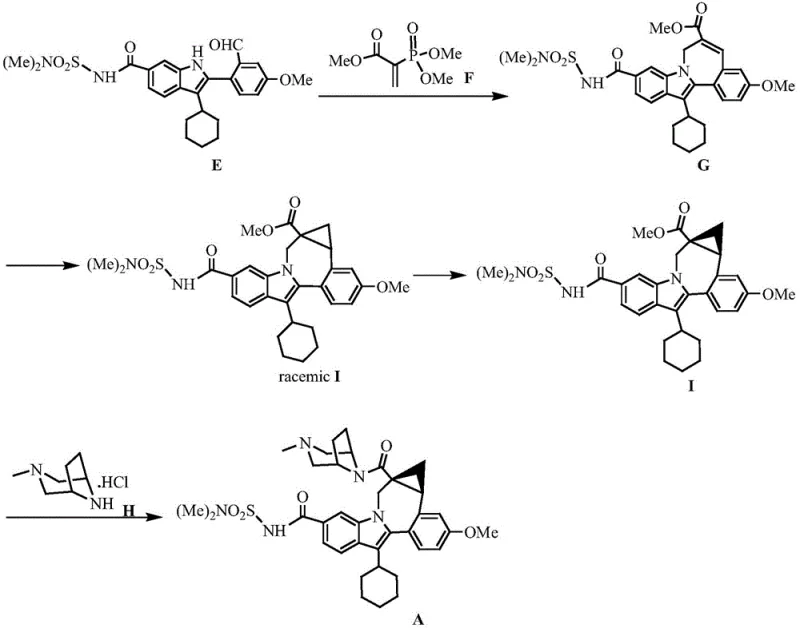
Beyond stereocontrol, the mechanistic design also inherently suppresses the formation of difficult-to-remove impurities. In traditional racemic syntheses, the presence of the wrong enantiomer often leads to downstream diastereomeric impurities that are notoriously difficult to separate via crystallization or chromatography. By enforcing high enantioselectivity at the bond-forming step, the new process prevents the generation of these isomeric impurities at the source. Additionally, the subsequent palladium-catalyzed arylation step utilizes a specific ligand system (Pcy3.HBF4) that promotes efficient C-N or C-C bond formation without compromising the integrity of the newly formed cyclopropane ring. This orthogonal reactivity ensures that the complex polycyclic structure is assembled cleanly, resulting in a crude product profile that is far easier to purify, thereby reducing the load on downstream purification units and solvent usage.
How to Synthesize Beclabuvir Intermediate Efficiently
The implementation of this synthesis requires precise control over reaction parameters to maximize the benefits of the asymmetric catalysis. The process begins with the in situ generation of a diazo species, followed immediately by the enantioselective cyclopropanation which sets the tone for the entire sequence. Operators must maintain strict anhydrous conditions during the catalytic step to prevent catalyst deactivation. Following the formation of the chiral cyclopropane scaffold, the subsequent alkylation and arylation steps proceed under basic and palladium-catalyzed conditions respectively, each optimized to preserve the optical purity established in the first stage. For a detailed breakdown of the specific reagent quantities, temperature profiles, and workup procedures required to replicate this high-efficiency pathway, please refer to the standardized protocol below.
- Perform diazotization of beta-bromomethyl propionate with p-acetamidobenzenesulfonyl azide to form the diazo compound (IV).
- Execute asymmetric cyclopropanation using a chiral spiroalkene diketone copper catalyst to establish stereochemistry with >94% ee.
- Conduct amine alkylation with the indole derivative followed by palladium-catalyzed arylation to finalize the core structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthetic route offers transformative advantages that extend well beyond simple yield improvements. The shift from a resolution-based process to an asymmetric catalytic one fundamentally alters the cost structure of the intermediate. By theoretically doubling the yield from the chiral step alone, the demand for expensive starting materials is drastically reduced, leading to significant raw material cost savings. Furthermore, the reduction in step count from six to four shortens the manufacturing cycle time, allowing for faster inventory turnover and improved responsiveness to market demand fluctuations. This efficiency gain is compounded by the elimination of the need to purchase and dispose of chiral resolving agents, which are often costly specialty chemicals.
- Cost Reduction in Manufacturing: The economic impact of eliminating the chiral resolution step cannot be overstated. In the conventional process, nearly half of the synthesized material is discarded as the unwanted enantiomer, representing a direct loss of all upstream processing costs associated with that fraction. The new asymmetric route converts the majority of the starting material into the desired isomer, effectively maximizing atom economy. Additionally, the use of a low-loading copper catalyst (1-5% molar ratio) instead of stoichiometric chiral auxiliaries significantly lowers the cost of goods sold (COGS). The simplified purification requirements due to higher crude purity further reduce solvent consumption and waste disposal fees, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses where a failure in any single step can halt the entire production line. The new four-step route reduces the number of potential failure points and utilizes robust, commercially available starting materials like beta-bromopropionic acid methyl ester. The mild reaction conditions (40-60°C) reduce the risk of thermal runaways or equipment stress, ensuring consistent batch-to-batch reproducibility. This stability is critical for long-term supply agreements, as it minimizes the risk of production delays caused by process deviations or the need for extensive re-processing of off-spec material.
- Scalability and Environmental Compliance: As regulatory pressure on pharmaceutical waste increases, the environmental profile of a synthesis route becomes a key factor in vendor selection. This new method generates substantially less chemical waste by avoiding the discard of the wrong enantiomer and reducing the number of unit operations. The avoidance of unstable and hazardous reagents like the phosphorus ylide used in the old route enhances operational safety and simplifies regulatory compliance. The process is explicitly designed for industrial production, with demonstrated scalability that supports the transition from pilot plant trials to multi-ton commercial manufacturing without the need for complex engineering modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing a clear picture of what partners can expect when adopting this methodology. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your existing API supply chain.
Q: What is the optical purity achieved in this new synthetic route?
A: The novel asymmetric catalytic method achieves an optical purity (ee value) of greater than 99% for the final Compound I, significantly surpassing traditional resolution methods.
Q: How does this process improve overall yield compared to literature methods?
A: By eliminating the inefficient chiral resolution step and reducing the step count from six to four, the total yield increases from approximately 16% to over 48%.
Q: Is the chiral catalyst used in this process commercially viable for scale-up?
A: Yes, the chiral spiroenedione ligand is easy to prepare and is used in low loading (1-5% relative to substrate), making it cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beclabuvir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antiviral agents depends on the reliability and quality of the supply chain. Our technical team has thoroughly analyzed the pathway described in CN109942584B and is fully prepared to execute this advanced asymmetric synthesis at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical chiral purity metrics required for Beclabuvir intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your HCV drug development programs. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this asymmetric route for your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can optimize your supply chain for the future.
