Advanced Synthesis of Chiral Trinuclear Copper Complexes for Magneto-Optical Applications
The landscape of molecule-based chiral functional materials is undergoing a significant transformation with the introduction of advanced coordination complexes that merge optical activity with magnetic properties. Patent CN109180709B details a groundbreaking preparation method for a (-)-2-(4'-pyridyl)-4,5-pinene-pyridine hexafluoroacetylacetonate copper complex, specifically a chiral trinuclear copper species with the molecular formula Cu3(hfac)6(L)2. This innovation addresses critical challenges in the field of magneto-optical spintronic devices by providing a material that simultaneously possesses chirality and ferromagnetism. For R&D directors and procurement specialists in the electronic materials sector, this patent represents a pivotal shift away from hazardous high-pressure synthesis towards a more manageable, atmospheric reflux protocol. The ability to produce such sophisticated trinuclear structures with high yield and purity opens new avenues for applications in chiral asymmetric catalysis and enantiomer separation, positioning this chemistry at the forefront of next-generation functional material development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of polynuclear metal complexes, particularly those requiring precise chiral induction and specific coordination geometries, has relied heavily on solvothermal methods. These conventional techniques often necessitate the use of sealed autoclaves operating under elevated temperatures and substantial internal pressures to force the reaction equilibrium towards the desired product. Such conditions introduce inherent safety hazards, including the risk of vessel failure and explosive decompression, which pose significant liabilities for large-scale manufacturing facilities. Furthermore, the post-processing of solvothermal reactions is frequently cumbersome, involving complex filtration steps to remove insoluble byproducts and difficult solvent exchange procedures to isolate the target crystals. The energy consumption associated with maintaining high-pressure environments also drives up operational costs, making the commercial scale-up of complex chiral catalysts economically challenging for many suppliers.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a mild atmospheric reflux strategy that dramatically simplifies the production workflow while enhancing safety profiles. By employing n-octane as the reaction solvent and heating the mixture to a moderate 130°C, the process avoids the need for specialized high-pressure equipment entirely. This approach not only mitigates the safety risks associated with solvothermal synthesis but also facilitates easier post-reaction handling, as the product crystallizes directly from the cooling solution over a period of 2 to 3 days. The simplicity of this route allows for straightforward filtration and vacuum drying, resulting in high-purity blue-green rhombic transparent crystals with yields reaching approximately 89% calculated as copper. This transition to ambient pressure chemistry represents a substantial cost reduction in electronic chemical manufacturing, enabling more reliable supply chains and faster turnaround times for high-value chiral intermediates.
Mechanistic Insights into Trinuclear Copper Coordination and Ferromagnetism
The structural integrity and functional performance of the (-)-2-(4'-pyridyl)-4,5-pinene-pyridine hexafluoroacetylacetonate copper complex are derived from its unique trinuclear architecture, which orchestrates a delicate balance between steric hindrance and electronic communication. As illustrated in the molecular structure below, the complex features three copper(II) ions arranged in a specific geometry where two of the ions exhibit a five-coordinated CuNO4 triangular bipyramidal configuration, while the central or third ion adopts a four-coordinated CuO4 geometry. The hexafluoroacetylacetone (hfac) ligands act as bridging agents, providing oxygen atoms that chelate to the copper centers, while the chiral monodentate nitrogen-containing organic ligand L supplies the coordinating nitrogen atoms necessary to complete the coordination sphere. This precise arrangement is critical for inducing the observed ferromagnetic coupling, as the orbital overlap between the copper centers is mediated by the specific orientation of the bridging ligands.
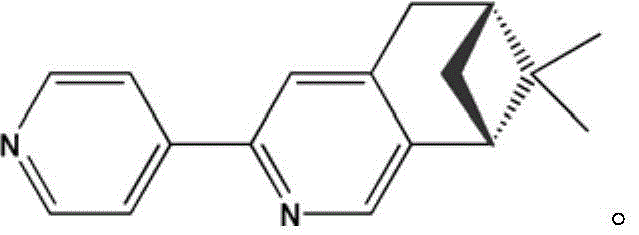
Beyond its structural novelty, the complex exhibits profound chiroptical and magnetic properties that are essential for advanced technological applications. Circular dichroism spectroscopy reveals strong exciton coupling signals at wavelengths of 310 nm and 330 nm, corresponding to negative and positive Cotton effects respectively, which confirms the successful transfer of chirality from the organic ligand to the metal center. Furthermore, magnetic susceptibility measurements indicate a slow increase in the product of molar magnetic susceptibility and temperature as the system cools from 300 K down to 2.0 K, a definitive signature of ferromagnetic interaction between the Cu(II) ions. For researchers developing magneto-optical devices, this dual functionality offers a rare opportunity to manipulate both light and magnetic fields within a single molecular entity, potentially leading to breakthroughs in data storage and spintronic logic gates.
How to Synthesize (-)-2-(4'-pyridyl)-4,5-pinene-pyridine Hexafluoroacetylacetonate Copper Complex Efficiently
The synthesis of this high-performance material is designed to be robust and reproducible, leveraging standard laboratory equipment to achieve industrial-grade results. The process begins with the preparation of the copper precursor, Cu(hfac)2·2H2O, which is synthesized by reacting hexafluoroacetylacetone with aqueous ammonia and copper acetate under mild stirring conditions. This intermediate is then combined with the chiral ligand L, which is separately prepared via the condensation of 4-(pyridylacetyl) pyridine iodide salt with myrtenal in formamide. The final complexation step involves refluxing these components in n-octane, a solvent choice that optimizes solubility and crystal growth kinetics. Detailed standardized synthesis steps follow below to ensure consistent quality and batch-to-batch reproducibility.
- Prepare the intermediate Cu(hfac)2·2H2O by reacting hexafluoroacetylacetone with aqueous ammonia and copper acetate, followed by filtration and vacuum drying.
- Synthesize the chiral ligand L [(-)-2-(4'-pyridyl)-4,5-pinene-pyridine] by condensing 4-(pyridylacetyl) pyridine iodide salt with myrtenal and ammonium acetate in formamide.
- React the Cu(hfac)2·2H2O intermediate with ligand L in n-octane under reflux at 130°C for 3 hours, then cool and crystallize to obtain the blue-green complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond mere technical performance. The elimination of high-pressure reactors significantly lowers the barrier to entry for manufacturing, allowing for deployment in a wider range of chemical facilities without the need for expensive safety certifications and specialized infrastructure. This flexibility translates directly into enhanced supply chain reliability, as production can be scaled up or shifted between sites with minimal retooling requirements. Moreover, the use of commercially available starting materials such as hexafluoroacetylacetone and myrtenal ensures a stable raw material supply, reducing the risk of bottlenecks that often plague the production of exotic fine chemicals.
- Cost Reduction in Manufacturing: The shift from solvothermal to atmospheric reflux processing eliminates the capital expenditure associated with high-pressure autoclaves and reduces the ongoing maintenance costs for such specialized equipment. Additionally, the simplified workup procedure, which relies on natural cooling and crystallization rather than complex extraction or chromatography, drastically reduces solvent consumption and labor hours per batch. By removing the need for energy-intensive high-pressure conditions, the overall utility costs for heating and pressure management are substantially lowered, contributing to a more competitive pricing structure for the final chiral complex.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent output even when scaling from laboratory to pilot plant volumes, minimizing the risk of batch failures that can disrupt downstream production schedules. Since the reaction conditions are mild and the reagents are stable, the process is less susceptible to variations in environmental factors or minor fluctuations in raw material quality. This stability allows suppliers to offer more reliable lead times for high-purity chiral functional materials, giving pharmaceutical and electronic manufacturers greater confidence in their inventory planning and product launch timelines.
- Scalability and Environmental Compliance: The use of n-octane as a solvent, combined with the absence of toxic heavy metal catalysts or harsh reagents, simplifies waste treatment and aligns with increasingly stringent environmental regulations. The high yield of the reaction minimizes the generation of chemical waste, reducing the burden on effluent treatment systems and lowering disposal costs. Furthermore, the ease of scaling this reflux-based process means that production capacity can be expanded incrementally to meet growing market demand without the long lead times typically required for installing high-pressure reactor trains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this chiral trinuclear copper complex. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the material's capabilities and limitations. Understanding these details is crucial for integrating this advanced material into your specific R&D pipeline or manufacturing process.
Q: What are the magnetic properties of this copper complex?
A: The complex exhibits significant ferromagnetic coupling between the Cu(II) centers, as evidenced by the increase in the product of molar magnetic susceptibility and temperature as temperature decreases from 300K to 2.0K.
Q: How does this synthesis method improve safety compared to traditional methods?
A: Unlike conventional solvothermal methods that require high temperature and high pressure in sealed vessels, this process utilizes atmospheric reflux in n-octane, significantly reducing explosion risks and equipment costs.
Q: What is the optical activity of the synthesized complex?
A: The complex demonstrates strong chiral optical activity, showing distinct exciton coupling signals with negative and positive Cotton effects at wavelengths of 310 nm and 330 nm respectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-2-(4'-pyridyl)-4,5-pinene-pyridine Hexafluoroacetylacetonate Copper Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating cutting-edge academic research into commercially viable products that drive innovation in the electronic and pharmaceutical sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral functional material meets the exacting standards required for magneto-optical and catalytic applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and accelerate your time to market.
