Advanced Synthesis of Mixed Quinoline Iridium(III) Complexes for Oncology Applications
The pharmaceutical industry is currently witnessing a paradigm shift in the development of metallodrugs, moving beyond the limitations of traditional platinum-based therapies. Patent CN112608341A discloses a groundbreaking class of mixed quinoline derivative iridium(III) complexes that demonstrate exceptional potential in oncology. This specific innovation addresses the critical need for agents that can bypass multidrug resistance mechanisms often encountered in clinical settings. The disclosed technology focuses on a novel cationic iridium complex, specifically hexafluorophosphorylation·pyrido[3,2-a]pyrido[1',2':1,2]imidazo[4,5-c]phenazine·bis-benzoquinolinone Iridium(III), which combines the pharmacophore of 8-hydroxyquinoline with rigid benzoquinoline ligands. As a leading entity in fine chemical manufacturing, we recognize that the transition from bench-scale discovery to commercial viability requires not just potent bioactivity, but also a robust, scalable, and economically feasible synthetic pathway. This report analyzes the technical merits of this patent to provide strategic insights for R&D directors, procurement managers, and supply chain heads looking to secure reliable pharmaceutical intermediates supplier partnerships for next-generation anticancer agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional cancer chemotherapy has long been dominated by platinum-based drugs such as cisplatin, carboplatin, and oxaliplatin, which function primarily through DNA cross-linking. However, the clinical utility of these agents is severely hampered by their narrow therapeutic index, leading to serious toxic side effects including nephrotoxicity, neurotoxicity, and ototoxicity. Furthermore, the emergence of tumor resistance, particularly in ovarian cancer cell lines like SK-OV-3/DDP, renders these conventional treatments ineffective over time. The synthesis of earlier generation metal complexes often involved harsh reaction conditions, unstable intermediates, or ligands that lacked the necessary structural rigidity to prevent rapid metabolic degradation in vivo. Additionally, purification processes for many organometallic drugs were fraught with difficulties, often requiring extensive chromatography that drove up manufacturing costs and reduced overall yield, making cost reduction in anticancer drug manufacturing a significant challenge for generic producers.
The Novel Approach
The novel approach detailed in the patent utilizes a strategic ligand design that incorporates a fused pyrido-imidazo-phenazine system coordinated to an iridium center. This structural architecture provides enhanced stability and allows for interaction with biological targets through mechanisms distinct from simple DNA binding, such as mitochondrial disruption or enzyme inhibition. The synthetic route described avoids the use of extremely sensitive reagents that require cryogenic temperatures, instead relying on controlled reflux conditions in common solvents like ethylene glycol and ethanol. By employing a stepwise assembly of the ligand system prior to metal coordination, the process ensures high regioselectivity and minimizes the formation of difficult-to-separate isomers. This methodological advancement translates directly into a more streamlined production workflow, offering substantial cost savings by reducing the number of purification cycles required to achieve high-purity OLED material or pharmaceutical grade standards, although in this specific case, the focus remains on high-purity pharmaceutical intermediates.
Mechanistic Insights into Iridium(III) Coordination and Ligand Assembly
The core of this technology lies in the precise construction of the heteroleptic iridium(III) complex. The synthesis begins with the oxidative modification of 8-hydroxyquinoline using sodium chlorate in hydrochloric acid to generate a quinone intermediate, which is subsequently condensed with 2-aminopyridine and o-phenylenediamine to form the large planar ligand (Compound 3a). This ligand acts as a strong chelator, stabilizing the iridium center in the +3 oxidation state. The second ligand component, derived from 7,8-benzoquinoline, forms a cyclometalated bond with the iridium, creating a rigid coordination sphere that protects the metal center from premature ligand exchange in biological fluids. The final complex exists as a cation balanced by a hexafluorophosphate anion, which enhances lipophilicity and cellular uptake compared to neutral analogues.
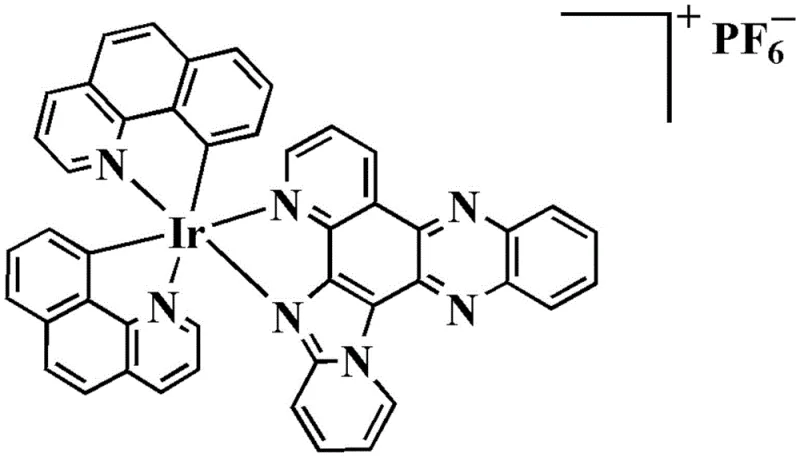
From an impurity control perspective, the mechanism relies on the thermodynamic stability of the final cyclometalated product. The reflux conditions in ethylene glycol drive the equilibrium towards the formation of the kinetically inert Ir-C and Ir-N bonds. Any unreacted starting materials or partially coordinated species are effectively removed during the workup phase, where the addition of saturated ammonium hexafluorophosphate induces selective precipitation of the target cationic complex. This precipitation step serves as a crude purification that significantly reduces the load on the subsequent alumina column chromatography. The use of neutral alumina rather than acidic silica prevents the decomposition of the acid-sensitive imidazo-phenazine moiety, ensuring that the final product retains its structural integrity and biological potency. This careful selection of stationary phase is critical for maintaining the commercial scale-up of complex organometallics without compromising quality.
How to Synthesize Mixed Quinoline Iridium Complex Efficiently
The synthesis of this high-value anticancer candidate involves a convergent strategy where the organic ligand and the metal precursor are prepared independently before being coupled in the final step. This modular approach allows for parallel processing and quality control of intermediates, which is essential for GMP compliance. The process utilizes standard laboratory glassware and heating mantles, avoiding the need for specialized high-pressure reactors. The key to success lies in the strict control of stoichiometry during the ligand condensation steps and the maintenance of an inert atmosphere during the final metal coordination to prevent oxidation of the iridium center. Detailed standardized synthesis steps follow below, outlining the precise temperatures, molar ratios, and solvent systems required to replicate the high yields reported in the patent literature.
- Synthesize the quinoline derivative ligand (Compound 3a) by reacting 8-hydroxyquinoline with sodium chlorate, followed by condensation with 2-aminopyridine and o-phenylenediamine.
- Prepare the iridium precursor (Compound 4) by refluxing 7,8-benzoquinoline with iridium trichloride hydrate in a mixture of ethylene glycol ethyl ether and water.
- Coordinate the ligand and precursor by refluxing Compound 3a and Compound 4 in ethylene glycol under argon, followed by precipitation with ammonium hexafluorophosphate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers several tangible benefits regarding cost efficiency and operational reliability. The primary advantage stems from the use of commercially available starting materials such as 8-hydroxyquinoline and iridium trichloride, which are sourced from established global supply chains, thereby reducing lead time for high-purity pharmaceutical intermediates. Unlike some exotic ligand systems that require custom synthesis from scratch, the intermediates in this pathway are generated through robust, well-understood organic transformations like condensation and oxidation. This familiarity reduces the risk of batch failure and ensures a consistent supply of raw materials. Furthermore, the elimination of cryogenic steps and the use of atmospheric pressure reflux simplify the engineering requirements for production facilities, allowing for easier technology transfer between sites.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to maximize atom economy and minimize waste generation. By utilizing a precipitation step with ammonium hexafluorophosphate to isolate the crude product, the process significantly reduces the volume of solvent required for initial isolation compared to methods that rely solely on extraction. Although column chromatography is used for final polishing, the efficiency of the preceding steps means the column load is manageable, extending the life of the stationary phase and reducing consumable costs. The avoidance of expensive palladium catalysts or rare earth promoters further contributes to substantial cost savings, making the final active ingredient more economically viable for large-scale production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like ethanol, acetic acid, and ethylene glycol ensures that the process is not vulnerable to shortages of niche reagents. These solvents are produced in massive quantities globally, providing a buffer against market volatility. Additionally, the synthetic intermediates, such as the quinone derivative and the benzoquinoline-iridium dimer, are stable solids that can be stored for extended periods without significant degradation. This stability allows manufacturers to build strategic inventory buffers, ensuring continuity of supply even in the face of logistical disruptions or sudden spikes in demand for oncology therapeutics.
- Scalability and Environmental Compliance: The process operates at moderate temperatures (40-150°C) and does not generate hazardous gaseous byproducts, simplifying废气 treatment requirements. The aqueous workup steps allow for the easy separation of inorganic salts, which can be treated via standard wastewater protocols. The use of ethylene glycol, a high-boiling solvent, facilitates energy recovery through distillation, aligning with green chemistry principles. This environmental compatibility reduces the regulatory burden associated with waste disposal permits and lowers the overall carbon footprint of the manufacturing process, which is increasingly important for meeting corporate sustainability goals in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this iridium complex. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this molecule into their existing drug development pipelines or manufacturing portfolios. The responses cover aspects ranging from biological efficacy to process scalability, providing a holistic view of the technology's value proposition.
Q: Why choose Iridium(III) complexes over traditional Platinum drugs?
A: Iridium(III) complexes offer distinct advantages including diverse mechanisms of action that can overcome cisplatin resistance, as well as potentially lower toxicity profiles towards normal human cells while maintaining high potency against resistant tumor strains.
Q: What is the solubility profile of the final complex?
A: The final hexafluorophosphate salt exhibits solubility in polar organic solvents such as acetonitrile and dichloromethane mixtures, facilitating purification via column chromatography and formulation in biological assays using DMSO stock solutions.
Q: How does the synthetic route ensure high purity?
A: The process utilizes specific recrystallization steps for intermediates and a final neutral alumina column chromatography separation, ensuring the removal of unreacted precursors and side products to achieve pharmaceutical-grade purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mixed Quinoline Iridium Complex Supplier
The development of novel metallodrugs represents a frontier in modern medicine, and the mixed quinoline derivative iridium(III) complex stands out as a promising candidate for overcoming cisplatin resistance. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from preclinical research to clinical trials is supported by a robust supply chain. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of characterizing complex organometallic structures using advanced spectroscopic techniques. We understand the critical nature of timeline adherence in drug development and are committed to delivering materials that meet the highest international quality standards.
We invite you to collaborate with us to optimize your supply chain for this high-potency anticancer agent. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with us, you gain access to our deep expertise in organometallic synthesis and process optimization. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us help you accelerate the delivery of life-saving therapies to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
