Advanced Synthesis of Matrine Pyrimidine Derivatives for Oncology Drug Development
The pharmaceutical industry is constantly seeking novel scaffolds that combine established safety profiles with enhanced therapeutic efficacy. Patent CN113563340A introduces a significant breakthrough in this domain by disclosing a series of matrine pyrimidine derivatives. These compounds represent a strategic fusion of the matrine alkaloid skeleton, known for its broad biological activities including anticancer and antiviral effects, with the pharmacologically privileged pyrimidine heterocycle. This hybridization aims to overcome the moderate selectivity of native matrine by introducing specific binding interactions through the pyrimidine ring. The patent details two distinct general formulas, Formula (I) and Formula (II), which encompass a wide range of substituents designed to optimize lipophilicity and electronic properties for targeting human tumor cells such as DU-145, A549, HCT15, and HeLa. 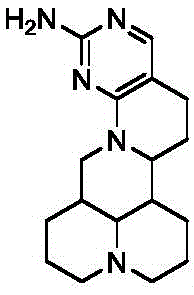
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional modification of the matrine molecule often suffers from a lack of regioselectivity due to the complexity of its tetracyclic quinolizidine structure. Conventional methods may involve harsh oxidative conditions or non-specific alkylation that can degrade the sensitive lactam ring or produce difficult-to-separate isomeric mixtures. Furthermore, direct coupling of heterocycles to the matrine core without a dedicated activation step often results in low yields and poor reproducibility. The absence of a reactive handle at the C14 or C15 positions typically necessitates multi-step protection and deprotection strategies, increasing both the cost of goods sold (COGS) and the environmental footprint of the manufacturing process. These inefficiencies create significant bottlenecks for procurement teams looking to source high-quality intermediates for preclinical development.
The Novel Approach
The methodology outlined in the patent offers a streamlined and highly controllable alternative. By employing a Vilsmeier-Haack reaction as the initial activation step, the process introduces a reactive formyl-chloro motif specifically at the C14-C15 junction. This intermediate serves as a robust platform for subsequent cyclization with guanidine salts, effectively constructing the pyrimidine ring in a single pot. This approach eliminates the need for protecting groups and minimizes side reactions. Additionally, the second pathway utilizing LDA-mediated deprotonation allows for direct nucleophilic attack on nitriles, providing access to a different structural diversity (Formula II) that is inaccessible via the acylation route. This dual-pathway strategy ensures that R&D teams have access to a comprehensive library of analogs for structure-activity relationship (SAR) studies without being constrained by a single synthetic limitation.
Mechanistic Insights into Vilsmeier-Haack Formylation and Cyclization
The core of this synthesis lies in the precise functionalization of the matrine backbone. As illustrated in the structural numbering scheme, the reaction targets the carbon atoms adjacent to the nitrogen in the D-ring. 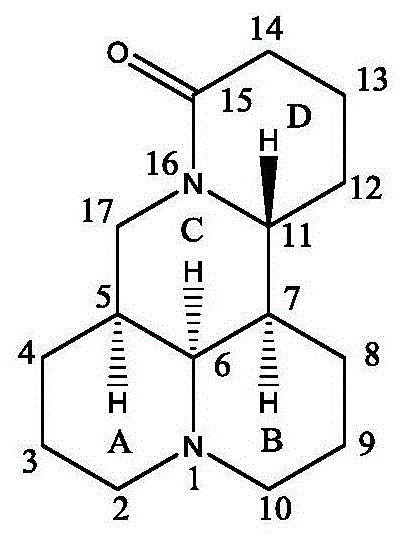 The Vilsmeier-Haack reagent, generated in situ from DMF and phosphorus oxychloride (POCl3), acts as a potent electrophile. It attacks the electron-rich enamine-like system within the matrine lactam ring, leading to the formation of an iminium intermediate which upon hydrolysis yields the 14-formyl-15-chloro derivative. This step is critical because it installs both an electrophilic carbonyl and a leaving group (chloride) in close proximity, setting the stage for intramolecular or intermolecular cyclization. The subsequent reaction with guanidine hydrochloride involves a nucleophilic attack by the amine nitrogen on the formyl carbon, followed by displacement of the chloride or elimination of water to aromatize the pyrimidine ring. This mechanism ensures high atom economy and generates the stable heteroaromatic system observed in compound WJ-01.
The Vilsmeier-Haack reagent, generated in situ from DMF and phosphorus oxychloride (POCl3), acts as a potent electrophile. It attacks the electron-rich enamine-like system within the matrine lactam ring, leading to the formation of an iminium intermediate which upon hydrolysis yields the 14-formyl-15-chloro derivative. This step is critical because it installs both an electrophilic carbonyl and a leaving group (chloride) in close proximity, setting the stage for intramolecular or intermolecular cyclization. The subsequent reaction with guanidine hydrochloride involves a nucleophilic attack by the amine nitrogen on the formyl carbon, followed by displacement of the chloride or elimination of water to aromatize the pyrimidine ring. This mechanism ensures high atom economy and generates the stable heteroaromatic system observed in compound WJ-01.
For the derivatives represented by Formula (I), the final diversification is achieved through acylation. The primary amine on the pyrimidine ring of WJ-01 acts as a nucleophile, attacking various substituted benzoyl chlorides. This step is remarkably tolerant of different functional groups on the aromatic ring, including halogens, alkyls, and trifluoromethyl groups. The reaction proceeds smoothly in dichloromethane with triethylamine as a base, indicating a low activation energy barrier. This mechanistic simplicity is advantageous for impurity control, as the byproduct is merely triethylamine hydrochloride, which is easily removed during aqueous workup. The result is a final product with extremely high purity, often exceeding 99%, which is essential for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Matrine Pyrimidine Derivatives Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing these high-value intermediates. The process begins with the activation of raw matrine, followed by cyclization to form the core scaffold, and concludes with functionalization to generate the specific target compounds (WJ-02 to WJ-35). The detailed operational parameters, including temperature controls and stoichiometry, are optimized to maximize yield and minimize waste. For a complete breakdown of the standardized operating procedures required for GMP-compliant production, please refer to the technical guide below.
- Perform Vilsmeier-Haack reaction on matrine using DMF and POCl3 to generate the 14-formyl-15-chloro intermediate.
- Execute cyclization with guanidine hydrochloride under reflux to form the core aminopyrimidine scaffold (WJ-01).
- Conduct final functionalization via acylation with acid chlorides or LDA-mediated addition with benzonitriles to obtain target derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits. The starting material, matrine, is a naturally occurring alkaloid extracted from Sophora flavescens, ensuring a renewable and stable supply chain that is not subject to the volatility of petrochemical feedstocks. The reagents used throughout the synthesis, such as DMF, POCl3, and various acid chlorides, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. This redundancy is crucial for maintaining business continuity in the face of market fluctuations or geopolitical disruptions. Furthermore, the high yields reported for the intermediate steps (up to 91% for the formyl-chloro intermediate) translate directly into reduced raw material consumption per kilogram of final product, driving down the overall variable costs of manufacturing.
- Cost Reduction in Manufacturing: The synthetic route is designed for efficiency, utilizing one-pot reactions where possible and avoiding expensive transition metal catalysts. The elimination of precious metals like palladium or platinum not only lowers the direct material cost but also removes the need for costly metal scavenging and residual metal testing, which are significant expense drivers in API production. The use of standard solvents like dichloromethane and methanol allows for efficient recovery and recycling through distillation, further enhancing the economic viability of the process on a commercial scale.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch quality. The reactions are performed under relatively mild conditions (often room temperature or gentle reflux), which reduces the stress on reactor equipment and lowers maintenance costs. The ability to synthesize a wide array of derivatives (WJ-01 to WJ-35) from a common intermediate means that inventory can be held at the semi-finished stage, allowing for rapid response to specific customer demands for different analogs without restarting the entire synthesis from raw matrine. This flexibility significantly reduces lead times for custom synthesis projects.
- Scalability and Environmental Compliance: The process avoids the generation of hazardous heavy metal waste, aligning with modern green chemistry principles. The primary waste streams consist of aqueous salts and organic solvents, which are well-understood and manageable through standard wastewater treatment protocols. The high purity of the crude products reduces the burden on purification columns, saving silica gel and solvent volumes. This streamlined downstream processing facilitates easier scale-up from laboratory grams to multi-ton production, ensuring that the supply can grow in tandem with clinical demand without requiring fundamental process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these matrine pyrimidine derivatives. The answers are derived directly from the experimental data and process descriptions found in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of the Vilsmeier-Haack route for matrine modification?
A: The Vilsmeier-Haack reaction allows for highly regioselective formylation and chlorination at the C14 and C15 positions of the matrine skeleton, creating a versatile intermediate for further heterocyclic construction without degrading the complex alkaloid core.
Q: How does the purity of these derivatives compare to standard matrine extracts?
A: The synthetic derivatives described in patent CN113563340A achieve constituent purity exceeding 99%, significantly higher than crude natural extracts, which simplifies downstream purification and ensures consistent biological assay results.
Q: Are the reagents used in this synthesis commercially scalable?
A: Yes, the process utilizes commodity chemicals such as phosphorus oxychloride, DMF, guanidine hydrochloride, and various substituted acid chlorides, all of which are readily available in bulk quantities for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Matrine Pyrimidine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of securing a reliable supply of high-quality pharmaceutical intermediates for your oncology pipeline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in early-stage discovery or late-stage clinical trials. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of matrine pyrimidine derivatives meets the highest standards of quality and consistency required for drug development.
We invite you to contact our technical procurement team to discuss your specific project needs. We are prepared to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating how our optimized synthetic routes can reduce your overall COGS. Reach out today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate the development of next-generation anticancer therapeutics.
